Summary
Our study results indicate that fibroblast growth factor 23 (FGF23) and NOD-like receptor protein 3 (NLRP3) levels are associated with the occurrence and progression of atherosclerotic cardiovascular disease (ASCVD) in elderly diabetic patients. Elevated levels of NLRP3 and FGF23 may be influential factors for atherosclerotic cardiovascular disease (ASCVD) in elderly diabetic patients. This provides new ideas and methods for risk assessment of ASCVD in elderly diabetic patients. It should be noted that the potential additional value of NLRP3 is limited and requires further validation in future longitudinal studies. Looking ahead, prospective cohort studies are needed to validate the causal relationships between NLRP3 and FGF23 and ASCVD, and systematically control confounding factors such as renal function (e.g., eGFR) and medication use in the studies. Expanding the sample size facilitates subgroup analyses to assess the heterogeneity of markers across different populations. Meanwhile, metrics such as NRI/IDI should be utilized to quantify the incremental predictive value of these biomarkers in traditional risk models and further investigate their interactive mechanisms in the development of ASCVD, in order to clarify the potential roles and intervention values of FGF23 and NLRP3 levels in cardiovascular risk management in elderly diabetic patients.
Introduction
Individuals diagnosed with diabetes demonstrate increased susceptibility to cardiovascular disease development [1]. Atherosclerotic cardiovascular disease (ASCVD) is a major cause of mortality and disability in elderly patients with diabetes [2, 3], with clinical manifestations including coronary artery disease, stroke, peripheral artery disease, and other macrovascular complications, contributing to reduced life expectancy and increased healthcare burden in the group [4]. Despite advancements in glycemic control, lipid-lowering therapies [5], and lifestyle interventions, residual cardiovascular risk remains high. This highlights the urgent need for novel and reliable biomarkers that can facilitate early identification of high-risk individuals and enable timely intervention to prevent adverse outcomes.
Inflammation and metabolic disorders jointly constitute the core pathological basis of diabetes-related ASCVD [6, 7]. The NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome, a key regulator of innate immunity, is abnormally activated in metabolic inflammatory diseases such as diabetes, promoting the maturation of cytokines like IL-1b and exacerbating endothelial dysfunction and atherosclerotic plaque formation [8–10]. Elevated expression levels of the NLRP3 inflammasome have been found in the aortas of patients with coronary atherosclerosis [11]. Existing evidence suggests that the NLRP3 inflammasome promotes atherosclerosis development by influencing various pathogenic events, such as oxidative stress, mitochondrial dysfunction, endoplasmic reticulum stress, and lysosomal disruption [11–13]. Diabetes and atherosclerosis are linked through multiple pathological pathways, including endothelial dysfunction, dyslipidemia, and oxidative stress. As a sensor of metabolic stress, NLRP3 links metabolic disorders to inflammation [11] and holds potential as a therapeutic target for atherosclerosis.
In addition to regulating phosphorus and vitamin D metabolism, fibroblast growth factor 23 (FGF23) has been shown to be independently associated with arterial calcification, left ventricular hypertrophy, and cardiovascular mortality risk [14–16]. In type 2 diabetes mellitus (T2DM), high FGF23 levels may further impair vascular function by promoting oxidative stress and fibrosis [17]. Recent studies have revealed that serum FGF23 and a-klotho (a co-receptor for FGF23) levels are associated with vascular calcification and endothelial dysfunction, both key factors in atherosclerosis [18]. Bi et al. reported that FGF23 and the FGF23/a-klotho ratio are positively correlated with carotid intima-media thickness and atherosclerosis in patients with T2DM [18].
Currently, most studies have focused solely on the independent roles of NLRP3 or FGF23 in ASCVD [19, 20], with limited evidence on their synergistic effects in the specific high-risk population of elderly patients with diabetes. There exists an interactive relationship between inflammatory pathways and disturbances in mineral metabolism during the development of atherosclerosis. NLRP3 may indirectly influence FGF23 signaling through inflammatory effects, while FGF23 may also enhance vascular inflammatory responses.
Aim
This study aimed to analyze the variation in serum NLRP3 and FGF23 levels in elderly diabetic patients with ASCVD through a cross-sectional survey, explore their associations with the risk of ASCVD, and evaluate their potential interactions, in order to provide new epidemiological evidence for a deeper understanding of the pathogenesis of ASCVD in elderly diabetic patients.
Material and methods
Study subjects
This was a hospital-based cross-sectional study conducted from February 2021 to June 2023. A total of 130 elderly diabetic patients, aged 60–89 years, were selected for the study. Patients were recruited using a consecutive sampling approach based on hospital admission records during the study period. A post hoc power analysis indicated that the sample size was sufficient to detect an area under the curve (AUC) > 0.8 with 80% power and a = 0.05.
Inclusion criteria: (1) patients who met the diagnostic criteria for T2DM in the elderly [3]: glycosylated hemoglobin > 6.50%, fasting blood glucose ≥ 7.00 mmol/l, and 2-h blood glucose > 11.10 mmol/l in the oral glucose tolerance test; (2) patients who could tolerate coronary angiography; (3) patients whose NLRP3 and FGF23 levels were measured after admission; (4) patients with complete clinical data; (5) patients without psychiatric diseases and cognitive disorders. Exclusion criteria: (1) patients with malignant tumors of the digestive system; (2) patients with failure of the heart, kidney, lung, and other vital organs; (3) patients with serious infections; (4) patients with autoimmune diseases or coagulation disorders; (5) patients with previous coronary artery stenting; (6) patients who have recently used medications that may affect NLRP3 and FGF23 levels.
Based on the results of coronary arteriography, the diabetic patients were allocated to the diabetic group (n = 54) and the ASCVD group (n = 76). The SYNTAX score was calculated online using the website https://syntaxscore.org/. Based on the SYNTAX scores, among the 76 patients with ASCVD, 28 were classified as low-risk (scores ranging from 1 to 22), 25 as intermediate-risk (scores from 23 to 32), and 23 as high-risk (scores ≥ 33). Diagnostic criteria for ASCVD: refer to the 2013 edition of the Guidelines for Cholesterol-Lowering Therapy to Reduce the Risk of Atherosclerotic Cardiovascular Disease (ASCVD) in Adults [21], i.e., ASCVD includes myocardial infarction, atherosclerotic stroke, acute coronary syndromes, transient ischemic attack, stable and unstable angina pectoris, peripheral atherosclerotic disease, and coronary or peripheral revascularization.
At the same time, 60 healthy individuals without diabetes, cardiovascular diseases, or other such conditions, who underwent physical examinations at our hospital during the same period, were selected as the control group. Their age ranged from 61 to 87 years.
Data collection
Information was collected and observed in all the study subjects, including age, gender, body mass index (BMI), smoking history, alcohol consumption history, fasting blood glucose, duration of diabetes, triglycerides (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and glycosylated hemoglobin, as well as NLRP3 and FGF23 levels.
All biomarker analyses were performed by laboratory technicians blinded to the patients’ ASCVD status to avoid assessment bias.
Measurement of NLRP3 and FGF23
After being enrolled in each group, patients underwent a 3 ml fasting peripheral elbow vein blood collection. The blood was placed in a yellow-headed tube of separating gel containing procoagulant and centrifuged using an Allegray 64R Centrifuge (Beckman Coulter, USA) at a speed of 3,000 r/min for 10 min, with the centrifugation radius of 10 cm. The obtained serum was divided into packages and stored at –4°C for spare use. The expression levels of NLRP3 and FGF23 in patients were measured using the enzyme-linked immunosorbent assay (ELISA) method with an automated ELISA analyzer (Thermo Multi-skan, FC model, USA). The ELISA detection kits were purchased from Jianglai Biotechnology Co., Ltd. in Shanghai, China. The specific operational procedures for the experiment were carried out according to the kit instructions.
Statistical analysis
All statistical analyses were conducted using SPSS v22.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism 8.0 (GraphPad Software, San Diego, CA, USA). Categorical data were expressed as the number of cases (%), and the c2 test was performed. Data distribution was assessed using the Shapiro-Wilk test to confirm normality before applying parametric statistical tests. Measurement data were expressed as mean ± standard deviation (SD); the independent sample t-test or one-way ANOVA was performed, and the LSD-t test was employed to make the pairwise comparison. A multivariate analysis was conducted using logistic regression analysis for independent variables related to the presence or absence of ASCVD. Receiver operating characteristic (ROC) curves were computed to determine the sensitivity and specificity of serum NLRP3 and FGF23 for ASCVD and help to find optimal cut-off points which were then defined by the Youden index. P < 0.05 was considered statistically significant.
Results
General information
There were no notable differences between the ASCVD, diabetic, and control groups of participants in terms of age, gender, BMI, history of smoking, history of alcohol consumption, TG, TC, LDL-C, and HDL-C (all p > 0.05). The fasting blood glucose and glycosylated hemoglobin levels were markedly higher in the ASCVD and diabetic groups than in the control group (all p < 0.001), and the ASCVD group exhibited longer diabetes duration and higher fasting blood glucose levels than the diabetes group (all p < 0.001) (Table I).
Table I
Comparison of general information of participants in the ASCVD group, diabetes group, and control group
| Factor | ASCVD group (n = 76) | Diabetes group (n = 54) | Control group (n = 60) | F/t/χ2 | P-value |
|---|---|---|---|---|---|
| Age [years] | 72.67 ±6.74 | 72.39 ±5.36 | 71.57 ±4.89 | 0.627 | 0.535 |
| Gender | |||||
| Male | 50 (66%) | 34 (63%) | 28 (47%) | 5.569 | 0.062 |
| Female | 26 (34%) | 20 (37%) | 32 (53%) | ||
| BMI [kg/m2] | 24.25 ±3.28 | 23.15 ±3.69 | 23.96 ±3.74 | 1.544 | 0.216 |
| Smoking history | 31 (41%) | 22 (41%) | 28 (47%) | 0.584 | 0.747 |
| Drinking history | 42 (55%) | 25 (46%) | 27 (45%) | 1.718 | 0.424 |
| Duration of diabetes [years] | 18.51 ±3.06# | 14.91 ±3.15 | – | 6.542 | < 0.001 |
| Fasting blood glucose [mmol/l] | 9.03 ±0.88*# | 8.08 ±0.61* | 5.14 ±1.05 | 348.088 | < 0.001 |
| Glycosylated hemoglobin (%) | 7.79 ±0.88* | 7.68 ±0.86* | 5.77 ±0.85 | 107.206 | < 0.001 |
| TG [mmol/l] | 1.72 ±0.34 | 1.68 ±0.33 | 1.62 ±0.35 | 1.446 | 0.238 |
| TC [mmol/l] | 5.55 ±0.65 | 5.43 ±0.67 | 5.36 ±0.70 | 1.424 | 0.243 |
| LDL-C [mmol/l] | 2.87 ±0.35 | 2.83 ±0.32 | 2.77 ±0.31 | 1.444 | 0.239 |
| HDL-C [mmol/l] | 1.22 ±0.27 | 1.23 ±0.28 | 1.28 ±0.25 | 0.869 | 0.421 |
ASCVD – atherosclerotic cardiovascular disease, BMI – body mass index, TG – triglycerides, TC – total cholesterol, LDL-C – low-density lipoprotein cholesterol, HDL-C – high-density lipoprotein cholesterol. Data are presented as n (%) for categorical variables and as mean ± SD for continuous variables.
Expression levels of NLRP3 and FGF23
NLRP3 and FGF23 levels in the ASCVD group were significantly higher than in both the diabetic and control groups (all p < 0.001), and those in the diabetic group were higher than in the control group (all p < 0.001) (Table II).
Table II
Comparison of serum NLRP3 and FGF23 in ASCVD group, diabetic group, and control group
| Indicators | ASCVD group (n = 76) | Diabetes group (n = 54) | Control group (n = 60) | F | P-value |
|---|---|---|---|---|---|
| NLRP3 [pg/ml] | 615.74 ±105.37*# | 532.54 ±87.92* | 356.38 ±52.26 | 152.800 | < 0.001 |
| FGF23 [pg/ml] | 954.67 ±168.96*# | 723.49 ±144.07* | 531.70 ±116.25 | 140.655 | < 0.001 |
Multifactorial logistic regression analysis of factors affecting the development of ASCVD in elderly diabetic patients
Taking the occurrence of ASCVD as the dependent variable (occurrence = 1, non-occurrence = 0), serum NLRP3 (continuous variable), FGF23 (continuous variable), fasting blood glucose (continuous variable), and diabetes duration (continuous variable) were used as independent variables and included in a binary logistic regression analysis model. The entry method was used to screen for relevant risk factors, with a significance level of 0.05 for introducing variables. The results revealed that NLRP3 (OR = 1.012; 95% CI: 1.005–1.019, p= 0.001), FGF23 (OR = 1.009; 95% CI: 1.005–1.014, p < 0.001), fasting blood glucose (OR = 1.371; 95% CI: 1.101–1.706, p= 0.005), and duration of diabetes (OR = 8.456; 95% CI: 2.830–25.271, p < 0.001) were independent risk factors for ASCVD in elderly diabetic patients (Table III).
Table III
Multifactorial logistic regression analysis of factors associated with the development of ASCVD in elderly diabetic patients
ROC curve analysis
Using NLRP3 and FGF23 as test variables and whether ASCVD occurred as a status variable, the ROC curve was analyzed. The results of the ROC curve analysis showed that the AUC was recorded as 0.732 (95% CI: 0.646–0.818), and the optimal cut-off value of the NLRP3 level for ASCVD was 609.575 pg/ml, with a Youden index of 0.378, corresponding to a sensitivity and specificity of 0.526 and 0.852, respectively. The AUC was 0.861 (95% CI: 0.791–0.932), and the optimal cut-off value of the FGF23 level for ASCVD was 812.350 pg/ml, with a Youden index of 0.694, corresponding to a sensitivity and specificity of 0.842 and 0.852, respectively. The combined (NLRP3 and FGF23) detection with optimal cut-off values (607.04 pg/ml, 723.91 pg/ml) had a sensitivity of 0.947, a specificity of 0.722, a Youden index of 0.669, and a corresponding AUC of 0.887 (95% CI: 0.827–0.947) (Table IV, Figure 1).
Table IV
Predictive value of blood levels of NLRP3 and FGF23 for the occurrence of ASCVD in elderly diabetic patients
Figure 1
ROC curves of serum NLRP3 and FGF23 levels to predict the occurrence of ASCVD in elderly diabetic patients
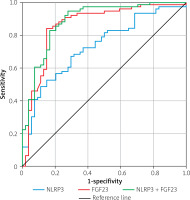
Further comparison of ROC (AUCs) using the DeLong test showed that the AUC for the combined detection of NLRP3 and FGF23 was greater than that for NLRP3 alone, with a statistically significant difference (z = 3.736, p < 0.001); the AUC for the combined detection of NLRP3 and FGF23 was slightly greater than that for FGF23 alone, but the difference was not significant (z = 1.352, p = 0.176).
Discussion
This study explored the associations of NLRP3 and FGF23 with ASCVD in elderly diabetic patients through cross-sectional analysis. The results of this study indicate that FGF23 and NLRP3 levels were associated with the occurrence and progression of ASCVD in elderly diabetic patients. Elevated levels of NLRP3 and FGF23 may be influential factors for ASCVD in elderly diabetic patients.
Specifically, both markers were elevated in patients with ASCVD compared to diabetic patients without cardiovascular complications and healthy controls. This finding suggests that inflammatory activation and disturbances in mineral metabolism may jointly participate in the pathological process of atherosclerosis in elderly diabetic patients with ASCVD. NLRP3 was significantly elevated in the ASCVD group, consistent with its role as a core component of the innate immune system, where its activation promotes the maturation of pro-inflammatory factors such as IL-1b, driving vascular inflammation and atherosclerosis progression [22, 23]. Our findings echo those of Hsu et al. [24] in animal models; their study showed that NLRP3 deficiency reduces atherosclerotic lesions in diabetic mice. The gradient elevation we observed in the human population suggests that NLRP3 inflammasome activation may serve as a mechanistic bridge linking diabetes to ASCVD. Furthermore, Bi et al.’s study revealed a positive correlation between FGF23, the FGF23/a-klotho ratio, and type 2 diabetes as well as its risk of atherosclerosis [18]. A recent study by Kurpas et al. confirmed that FGF23 levels are independently associated with major adverse cardiovascular events in diabetic cohorts, particularly in elderly populations [17]. Our data further support this association, showing that FGF23 levels in the ASCVD group were nearly double those in healthy controls, likely reflecting its dual role in mineral metabolism dysregulation and direct pro-inflammatory effects on vascular tissues.
ROC curve analysis showed that the AUC for the combined detection of NLRP3 and FGF23 was greater than that for NLRP3 alone (with statistical significance) and slightly greater than that for FGF23 alone (but without statistical significance). Although the combined model showed some discriminatory advantage, the primary association signal may stem from FGF23, while the incremental contribution of NLRP3 in this cross-sectional design was relatively limited, and its independent predictive value still requires validation in future longitudinal studies. Combined detection of these two markers aids in a more comprehensive assessment of concurrent inflammatory activation and disturbances in mineral metabolism in elderly diabetic patients (Figure 2), thereby providing references for early risk identification and intervention for ASCVD. Our findings suggest that inflammatory pathways (represented by NLRP3) and mineral metabolism imbalances (represented by FGF23) may have synergistic effects in the development of ASCVD, offering a new perspective for understanding the pathological mechanisms of cardiovascular complications in this population. Simultaneous elevation of both markers can also more comprehensively capture the multiple pathophysiological processes of ASCVD, providing a more comprehensive basis for risk stratification. This strategy also aligns with the modern concept of integrating multiple biomarkers in cardiovascular medicine [25].
Furthermore, the ASCVD group exhibited longer diabetes duration and higher fasting blood glucose compared to the diabetes-only group, consistent with prior studies linking poor glycemic control and chronic hyperglycemia to accelerated atherosclerosis [26, 27]. It has been reported that prolonged exposure to hyperglycemia likely amplifies NLRP3 activation via advanced glycation end products, which promote ROS generation and inflammasome priming [27]. It has also been reported that diabetes duration may reflect cumulative FGF23 exposure, as chronic kidney disease – a common comorbidity in long-standing diabetes – is a major stimulus for FGF23 secretion [28].
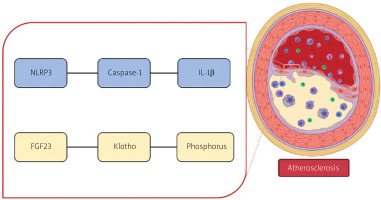
Figure 2
Schematic diagram showing the convergence of inflammatory and phosphate handling pathways in vascular outcomes. NLRP3 may participate in atherosclerosis through the caspase-1/IL-1β pathway, while FGF23 may influence atherosclerosis via the Klotho/phosphorus pathway
However, our study still has some limitations. Firstly, the cross-sectional design adopted in this study limits the establishment of causal relationships in the results. Secondly, the analysis model of this study did not include estimated glomerular filtration rate (eGFR) as a covariate, thus failing to exclude severe confounding caused by different stages of chronic kidney disease. This is a major limitation of this study. Thirdly, medications used by patients may affect FGF23 levels or inflammatory status, and this study did not systematically analyze the impact of medications, which may constitute another confounding factor. Fourthly, the sample size was relatively limited, and this study did not conduct subtype analysis to test the robustness of the research results in different subtypes of the population. This may limit the generalizability of the research results in specific subtypes of the population. In addition, this study did not include reclassification metrics such as the net reclassification improvement (NRI) index or integrated discrimination improvement (IDI) index to further verify the additional value of the dual-biomarker model compared to traditional risk assessment models.
Conclusions
Our study results indicate that FGF23 and NLRP3 levels are associated with the occurrence and progression of ASCVD in elderly diabetic patients. Elevated levels of NLRP3 and FGF23 may be influential factors for ASCVD in elderly diabetic patients. This provides new ideas and methods for risk assessment of ASCVD in elderly diabetic patients. It should be noted that the potential additional value of NLRP3 is limited and requires further validation in future longitudinal studies. Looking ahead, prospective cohort studies are needed to validate the causal relationships between NLRP3 and FGF23 and ASCVD, and systematically control confounding factors such as renal function (e.g., eGFR) and medication use in the studies. Expanding the sample size facilitates subgroup analyses to assess the heterogeneity of markers across different populations. Meanwhile, metrics such as NRI/IDI should be used to quantify the incremental predictive value of these biomarkers in traditional risk models and further investigate their interactive mechanisms in the development of ASCVD, in order to clarify the potential roles and intervention value of FGF23 and NLRP3 levels in cardiovascular risk management in elderly diabetic patients.







