Acute limb ischemia (ALI) is a vascular emergency defined by a sudden loss of blood flow to a limb, potentially resulting in amputation or death. The most common cause of ALI, accounting for approximately 30% of cases, is embolic occlusion, frequently occurring at the femoral bifurcation. This condition is typically associated with atrial fibrillation or originates from a cardiac source, such as myocardial infarction, a prosthetic heart valve, or mitral stenosis [1, 2]. It may also arise from severe left ventricular dysfunction and aortic stenosis [3].
A 55-year-old man was admitted to the emergency department with acute ischemia of the left lower limb (Figure 1 A). The patient was hemodynamically and respiratory stable. He presented with pallor of the extremity with no palpable pulse in its distal part. Laboratory tests revealed slightly impaired kidney function (eGFR 58 ml/min/1.73 m2) and increased white blood cell levels (12.19 × 103/µl).
Figure 1
A – An amputation of SFA in computed tomographic angiography (green arrow). B – A heavily calcified aortic tricuspid valve looking like bicuspid observed on transthoracic echocardiogram (green arrow). C – A mobile thrombus measuring 0.9 ȕ 1 cm located in the apical segment of the left ventricle (green arrow). D – A Ňsmoke-likeň appearance noted in both the left atrium and left ventricle (green arrow)
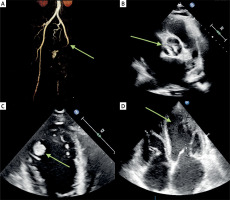
The transthoracic echocardiography (TTE) revealed global cardiac enlargement, dilation of the ascending aorta and main pulmonary artery, and generalized contractility disorders, including impaired right ventricular systolic function. The examination also showed a heavily calcific aortic bicuspid valve (Figure 1 B) with severe low-flow, low-gradient aortic stenosis (AVA of 0.8 cm2, grad.: 37/22 mm Hg, LVEF 20%) and moderate regurgitation. It also indicated a high probability of pulmonary hypertension (RVSP 58 mm Hg), systolic hepatic vein flow reversal, and a mobile mass (0.9 × 1 cm) in the apical segment of the left ventricle, suggestive of a thrombus (Figure 1 C). Moreover, spontaneous echo contrast was observed in the left atrium and left ventricle (Figure 1 D).
A thromboembolectomy was performed under local anesthesia of the left groin. Large amounts of thromboembolic material were removed from the common femoral artery (CFA), superficial femoral artery (SFA), and profunda femoris artery (PFA). The extracted elements were sent for bacteriological examination. The pulse on the limb was restored.
An urgently performed coronarography was unremarkable. The patient was qualified for a surgical aortic valve replacement. After opening the ascending aorta, the anatomically tricuspid stenotic valve was removed and replaced by an Avalus 23A bioprosthesis. The left ventricle was examined during the procedure, but the thrombus was not found, and intraoperative transesophageal echocardiography did not reveal its presence either.
The postoperative course was stable, with no adverse neurological events.
In low-flow, low-gradient (LFLG) conditions, severe aortic stenosis often arises due to the progressive onset of left ventricular systolic dysfunction characterized by poor contractility, hemodynamic disturbances, and blood stasis. As heart failure progresses, various abnormalities in the vessel wall and blood constituents, including elevated levels of von Willebrand factor, increase the risk of thromboembolism. Some guidelines recommend anticoagulation therapy in heart failure associated with certain specific conditions, such as venous thromboembolism, atrial fibrillation, prior thromboembolic events, or a cardioembolic source [4]. However, severe aortic stenosis can lead to a counterintuitive phenomenon known as Heyde’s syndrome, where high shear stress around the valve alters the structure of von Willebrand factor, resulting in its degradation and subsequent disruption of hemostasis [5]. Therefore, complex cases involving aortic valve stenosis and ventricular dysfunction must be carefully monitored for the risk of thrombosis, which can result in intracardiac thrombus formation and subsequent thromboembolism, such as ALI.







