Introduction
As a consequence of the development of early prognostic models stratifying patients with brain metastases into groups with distinct survival outcomes [1–3], discussions emerged around the preferred management strategies for patients in defined prognostic classes. For example, the recursive partitioning analysis (RPA) model from 1997, which was derived from three Radiation Therapy Oncology Group clinical trials, showed that median survival was limited to 2.3 months in class III, i.e. patients with Karnofsky performance status (KPS) < 70 [1]. In disease-specific prognostic models, e.g. for lung cancer [3], it was also obvious that actively treated patients in the worst prognostic categories had short median survival, which was only marginally longer than that of patients managed with steroids and other supportive measures, so-called best supportive care (BSC). These findings questioned the role of palliative whole-brain irradiation (WBRT), a historical standard of care for most patients with multiple brain metastases, which on the one hand may improve neurological symptoms but on the other hand carries a risk of fatigue and neurocognitive decline, to name some potential side effects [4].
Given that cross-study comparisons may suffer from numerous shortcomings, eventually a well-designed prospective randomised study was performed, the QUARTZ trial [5]. It focused on patients with non-small cell lung cancer (NSCLC) and required a multidisciplinary team that included both neurosurgeons and radiation oncologists to have concluded that the patients were unsuitable for either surgery or stereotactic radiotherapy (SRT). Trial participants were randomly assigned (1 : 1) to receive either BSC (notably, 35% received additional anticancer therapy including but not limited to systemic therapy) plus WBRT or BSC (termed optimal supportive care by the investigators) alone. The latter included oral dexamethasone (median daily dose 8 mg) given with a proton pump inhibitor, with the dose of steroid determined by the patients’ symptoms and titrated downwards if symptoms improved, as well as support from a specialist nurse and immediate access to specialized clinicians and pal- liative care teams. Given that patients with limited survival expectation were included, WBRT consisted of 20 Gy in 5 daily fractions rather than 30 Gy in 10 daily fractions. In 2007–2014, 538 patients from the UK and Australia were randomly assigned to BSC plus WBRT (n = 269), or BSC alone (n = 269). Their median age was 66 years (range 38–85 years), and 203 (38%) had a KPS < 70. In the majority, the primary tumour was uncontrolled and extracranial metastases were present too. The median survival was similar (9.2 weeks in the WBRT arm and 8.5 weeks in the other arm), and so was the outcome for the primary endpoint of quality-adjusted life-years (QALY).
Due to the increasing implementation of BSC across additional primary tumour types for which no specific prospective clinical studies are available, attention should be given to potential differences in survival expectation, and prognostic heterogeneity, in order to provide sound and realistic counselling to patients, their families and caregivers. The purpose of the present study was to analyse the impact of numerous patient- and disease-related baseline parameters on survival in a consecutive cohort managed in a geographically well-defined health care region, Nordland county in northern Norway. We also included blood test results, such as serum albumin and C-reactive protein (CRP).
Material and methods
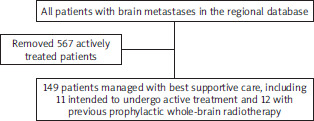
All oncology care in the Nordland county is overseen and guided by clinical oncologists and multidisciplinary tumour boards located at the Nordland Hospital in Bodø. The driving distance to the nearest hospitals with oncology departments in Tromsø and Trondheim exceeds 500 km, meaning that strong attempts are made to avoid unnecessary travel [6]. Especially, palliative treatment of patients with metastatic disease is conducted close to their place of living and guided by the Nordland Hospital in Bodø. Therefore, the hospital’s electronic patient records provide excellent coverage of regional oncology care. High-quality studies comparable to those of large cancer registries are feasible and also regularly performed to monitor quality of care in our rural region with approximately 240,000 inhabitants [7]. One of our regularly updated quality-of-care databases includes all patients who developed brain metastases from solid primary tumours [8]. We employed this database, which covers the time period of 2007–2024, to extract all adult patients managed with BSC rather than WBRT, SRT, surgery or systemic therapy for their brain metastases (Figure 1). Before proceeding with detailed analyses, we verified the appropriateness of including patients who were scheduled for active treatment but did not receive it (n = 11, surgery in 1 case, radiotherapy in 10) and those managed with BSC after previous prophylactic WBRT for small cell lung cancer (SCLC) (n = 12). We hypothesized in the beginning that these patients may represent prognostically different subgroups. However, survival was similar to that of all other BSC patients, and therefore the study eventually included all 149 patients. The reasons for conversion from initially recommended active therapy to BSC in 11 patients included rapid clinical deterioration during treatment preparation, and patient decision. Given that all patients were covered by the public national health care system, which provides all types of anti-cancer treatment and supportive care including travel and housing expenses, financial reasons were not causative when choosing between different management options.
The main purpose was to stratify the patients by primary tumour type, because the pivotal previous study (QUARTZ) only included NSCLC. Secondary purposes included stratified outcomes for other baseline parameters such as age, sex and blood test results. The latter included all components of the brain-metastases-specific LabBM score, i.e. serum albumin, haemoglobin, platelets, CRP and lactate dehydrogenase (LDH) [9]. The Glasgow prognostic score was assigned, too (normal CRP and albumin: 0, abnormal CRP and albumin: 2, others: 1) [10]. The primary outcome of interest was overall survival from the date of imaging diagnosis of brain metastases. At the time of analysis, all patients had died (no censored observations). Imaging and laboratory data refer to the closest time point before detection of brain metastases, or the same day. Data older than 4 weeks were not considered. Institutional upper and lower limits of normal were employed to stratify blood test results, in line with the LabBM methodology [9]. Extracranial staging consisted of compu- ted tomography. If necessary for oncologic decision-making, further modalities were added to clarify computed tomography findings, e.g. isotope bone scan, ultrasound, positron- emission tomography. The number of brain metastases was derived from magnetic resonance imaging reports. Overall survival (time to death) from radiological diagnosis of brain metastases was calculated employing the Kaplan-Meier method, and different groups were compared using the log-rank test (SPSS 29.0.1.0; IBM Corp., Armonk, NY, USA). For continuous variables, such as age, univariate Cox regression was employed. A multivariate forward conditional Cox regression analysis was also performed. A p-value < 0.05 was considered statistically significant.
Results
The study included 30 patients (20%) with SCLC, 25 (17%) with NSCLC adenocarcinoma (in one case with actionable target, EGFR mutation), 16 (11%) with malignant melanoma and numerous other cancers with maximum 11 patients per group. Non-small cell lung cancer programmed death- ligand 1 (PD-L1) status was available in 6 cases, 5 of whom were positive. Malignant melanomas were BRAF mutated in 5 cases. The median age of our cohort was 72 years and the median KPS 60, meaning that most patients belonged to RPA class III. In 83% of cases, brain metastases developed after a certain latency period from first cancer diagnosis, typically 1–2 years, and at that time a high burden of extracranial disease was present. The latter was reflected in high proportions of patients with abnormal blood test results, especially CRP, LDH and haemoglobin (Table 1).
Table 1
Patient characteristics (N = 149)
Patients who had received prophylactic WBRT for SCLC had a median survival of 1.0 months, those who remained untreated despite other plans (BSC conversion) of 1.2 months, and those with intended BSC of 1.4 months (all pairwise comparisons of survival curves had p-values > 0.15). Median survival was 1.3 months (95% CI 1.08–1.52) for all 149 patients combined. The 3- and 6-month survival rates were 20% and 1%, respectively. Maximum survival was 7.5 months and observed in a 73-year old male patient with newly diagnosed NSCLC adenocarcinoma without extracranial metastases, but with malignant pleural effusion, who had 2 brain metastases measuring 7 mm each and poor KPS. At the time of diagnosis in 2010, such patients were not tested for PD-L1 status or uncommon targets. In 2024, next generation sequencing would have been standard at our hospital and, depending on the findings, may be systemic therapy.
Information about steroid treatment was recorded in 50 of 149 patients. Of these, 31 responded to steroids, while 10 did not. The remaining 9 patients had imaging-detected asymptomatic lesions and did not receive any steroids. Survival was significantly different between the 3 groups: median 1.6 months (improved symptoms), 0.8 months (non-responders), and 1.1 months (asymptomatic), p = 0.004 pooled over all strata. Due to missing information in 99 patients, multivariate analysis did not include steroid treatment.
The primary tumour type was significantly associated with survival. Median values 1.0–1.5 months were observed for all types, except renal cell cancer (1.7 months) and malignant melanoma (2.3 months). Figure 2 shows the survi-val curves. Presence of extracranial metastases was significantly associated with survival (Figure 3) (median 1.3 and 1.8 months, respectively), but the number of involved organs or presence of specific sites such as the liver or bones was not. Karnofsky performance status was significantly associated with survival (Figure 4) (median 0.8, 1.1 and 2.3 months, respectively). Because the p-value for the comparison between the curves of patients with KPS 30–40 and 50–60 was 0.33, only 2 categories (< 70 and 70–90) were carried forward to multivariate analysis. An unexpected, counterintuitive correlation between age and survival was observed in univariate Cox regression (p = 0.013), which also persisted in a log-rank test of survival curves dichotomized by median age. Patients aged 72 years or older survived significantly longer than their younger counterparts (median 1.5 and 1.1 months, respectively; p = 0.02). Three of five blood tests were also significantly associated with survival: LDH (median 1.0 and 1.7 months, p = 0.002), CRP (median 1.2 vs. 1.5 months, p < 0.001), and haemoglobin (median 1.1 and 1.5 months, p = 0.01). The Glasgow prognostic score did not reach significance (median 1.3, 1.2 and 1.0 months, respectively; p = 0.07). Numerous other baseline variables were not significantly associated with survival: synchronous brain metastases, time interval to development of brain metastases, size and number of brain metastases, setting (symptoms vs. imaging finding), time of BSC (2007–2012 vs. 2013–2018 vs. 2019–2024), sex, primary tumour status (controlled vs. uncontrolled), serum albumin and platelet count.
Figure 2
Kaplan-Meier survival curves for patients with renal cell cancer/malignant melanoma vs. all other cancer types p = 0.006 (log-rank test)
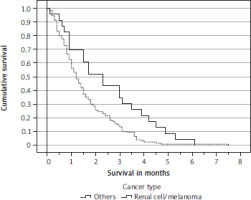
Figure 3
Kaplan-Meier survival curves for patients with or without extracranial metastases p = 0.029 (log-rank test)
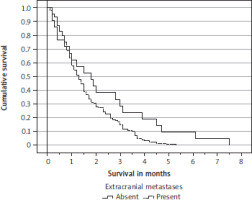
Figure 4
Kaplan-Meier survival curves for patients with different Karnofsky performance status categories p = 0.007 (log-rank test, pooled over all strata)
Patient selection criteria changed over time. While median age and proportion of patients with extracranial metastases were similar in all three strata (2007–2012, 2013–2018, 2019–2024), median KPS was 50 in contemporary patients and 60 in the two previously managed strata (p = 0.008). Fewer contemporary patients had malignant melanoma or renal cell cancer (5% compared to 15% in 2013–2018 and 29% in 2007–2012, p = 0.003).
The parameters with significant p-values in univariate survival analyses were included in the multivariate Cox regression analysis. An exploratory initial version also included the three blood test results (CRP, LDH, haemoglobin). We were concerned that loosing 45 patients with unknown LDH results would lead to undesirable loss of statistical power. On the other hand, the fact that dichotomized CRP (normal vs. elevated) emerged as a significant predictor of survival in the exploratory Cox analysis is worth mentioning and relevant for the design of future studies. Based on the full dataset in 149 patients (without blood tests), the subsequent Cox model suggested that survival mainly depends on the dichotomized variables of KPS, extracranial metastases, and primary tumour type (Table 2). In principle, a prognostic model or score may be clinically desirable. However, all prognostic strata in our study had median survival times < 2.5 months, indicating an inevitable poor outcome. A better prognostic group based on KPS 70–90 plus additional factors would contain only 10 patients, i.e. 7% of the cohort, which is not very helpful. The same would be true for primary tumour type as the main decision criterion.
Discussion
This study was performed to analyse the impact of numerous patient- and disease-related baseline parameters, including blood test results, on survival in a contemporary BSC cohort from a single institution, yet at the same time a geographically large region where all oncologists are located at the main hospital. The latter fact is relevant when judging the generalizability of the results, which resemble those derived from cancer registries. Notable differences exist between the publicly funded Norwegian health care system and others, where the threat of financial toxicity may impact on patients’ choices [11, 12]. Therefore, regional studies from other parts of the world are warranted. In total, 21% of the brain metastases patients overseen by our region’s oncology department were managed with BSC, largely in the setting of intentionally recommended BSC and sometimes due to circumstances interfering with originally envisioned active treatment. The median age of our cohort was 72 years and the median KPS 60, meaning that most patients belonged to RPA class III. Shared decision-making was always individualized, accounting for the complete oncological picture rather than basing recommendations on specific prognostic scores. It is not surprising that age and KPS were among the factors our oncologists considered before recommending BSC. Over time, several baseline characteristics of BSC patients changed, e.g., lower KPS in contemporary patients. Possibly, patients with KPS 60–70 who were considered poor candidates for chemotherapy in 2007–2012, i.e., before modern tyrosine kinase inhibitors and immune checkpoint inhibitors became available, were more likely to try active systemic therapy with the improved toxicity profile and administration route in recent years. Such drugs were introduced for patients with malignant melanoma and renal cell cancer, i.e. subgroups less commonly managed with BSC in 2019–2024 in our region. In typical studies of active treatment for brain metastases, median age was lower and median KPS higher than in our cohort [2, 9, 13, 14]. Median survival was 1.3 months in this study and temporally stable over time, but notably poor (0.8 months) in steroid non-responders. Median values of 1.0–1.5 months were observed for all primary tumour types, except renal cell cancer (1.7 months) and malignant melanoma (2.3 months). A large Australian series included 327 melanoma patients with brain metastases managed with BSC in 1952–1984 and another 210 patients from the era of 1985–2000 [15]. Median survival was 1.7 months in patients treated in the earlier part of the study and 2.1 months in those treated after 1984. During the time- period 1996–2000, 26% of patients were managed with BSC. We are not aware of comparable research data for patients with primary renal cell cancer.
When interpreting our findings, the typical limitations of retrospective studies spanning over more than a decade should be considered, such as temporal changes in management approaches and molecular characterization of tumours. In addition, the subgroups were smaller than originally expected, even for common cancer types such as breast cancer. This resulted in limited statistical power for several comparisons. The group with other primary tumours (n = 21) may have included subsets with different prognoses, which only a large study can address in sufficient detail. Furthermore, some patients had incomplete blood tests and steroid data available. We were therefore unable to perform a complete multivariate analysis of all potential prognostic factors in all 149 patients. It appears possible that both CRP and steroid response may emerge as significant prognostic factors in multivariate Cox regression, in addition to KPS, extracranial metastases and primary tumour type. Larger studies are needed to resolve this issue. On the other hand, given the inevitable poor outcome, the clinical impact of optimized, complex survival prediction models appears limited in the BSC setting with its focus on other aspects of care. Statistically significant scores may not always impact on clinical end-of-life care practice [16]. If the oncology care providers are aware of the fact that classical prognostic factors such as KPS and extracranial metastases also determine survival in the BSC setting, and that the natural history of renal cell cancer and malignant melanoma differs from that of other cancer types, they are equipped with relevant basic knowledge, which however should be complemented by additional clinical variables and, if involved, the invaluable input of the palliative care team. Such a comprehensive team effort may provide the best available patient counselling and symptom management.
A notable difference must be considered when comparing QUARTZ, a trial providing high-evidence outcome data, to other BSC data, namely that 35% of trial patients received additional anticancer therapy including but not limited to systemic therapy. The latter may exert beneficial effects also on brain metastases, at least in a proportion of patients [17]. Median survival was similar in the QUARTZ trial arms (9.2 weeks with WBRT and 8.5 weeks in the other arm), and so was the outcome for the primary endpoint of QALY. The equivalent number of 8.5-week survival is 2 months, a slightly longer result than the 1.3 months observed in our all-comers study. In QUARTZ, 38% had a KPS < 70, compared to 81% in the present cohort. Given the prognostic role of KPS, this difference likely explains the survival discrepancy. In the literature, BSC data can be derived mainly from heterogeneous studies that employed different management approaches, rather than studies specifically addressing BSC. In a different part of Norway, a multicentre, prospective, observational study was performed, which also included 50 patients managed with BSC [18]. Their median age was 74 years (72 in our study), 66% had Eastern Cooperative Oncology Group (ECOG) PS 3–4, 48% lung cancer, 74% symptomatic brain metastases, and 88% extracranial metastases. The median survival was 1.2 months, and all patients died within 8 months, similar to our study. In an older Dutch study, 118 patients received BSC [2]. Reasons for withholding additional radiotherapy were poor KPS, patient refusal, and prior prophylactic WBRT. The median survival was 1.3 months, with 4% alive after 6 months (1% in our study). Median survival of 1.3 months was also reported in a study from the USA (2017–2019), where 110 of 862 patients (13%) received BSC [19].
In patients with brain metastases, short survival or early mortality, defined as 30-day mortality, is largely a consequence of uncontrolled extracranial disease [20]. Therefore, the extent of extracranial disease, its impact on organ function expressed by blood test results, and availability of further systemic therapy addressing these metastases should always be assessed before planning active brain-directed treatment. The American Society for Radiation Oncology clinical practice guideline states that for patients with brain metastases and poor prognosis, early introduction of palliative care for symptom management and caregiver support are recommended [21]. In addition, supportive care only (with omission of WBRT) should be considered. If WBRT is used, brief schedules (e.g., 5 fractions) are preferred. Estimates of patient prognosis can be derived from the RPA or the newer diagnosis-specific graded prognostic assessment, which is a validated prognostic score based on the histologic cancer subtype [22]. The American Society for Radiation Oncology provided a treatment algorithm in Figure 2 of the practice guideline, where supportive care was suggested for patients with extensive brain involvement, if they also were classified as ECOG PS 3–4 with systemic disease and poor systemic therapy options, and if the brain metastases symptoms were controlled with steroids. A broader discussion of supportive needs and components of palliative care is beyond the scope of our study. Several recent publications have covered these aspects and provided excellent overviews [23–25]. Patient-centred care has become more relevant than ever before, throughout the disease trajectory [26]. Especially in the terminal phase, choosing wisely and avoiding futile treatment efforts saves patients from unnecessary toxicity, travelling and time spent in hospitals or oncology practices [27]. The present results inform decision-making and planning towards the end-of-life (hospice enrolment and other aspects) by underlining the short survival of patients with KPS < 70 and/or steroid unresponsiveness.
Conclusions
All prognostic strata in our study had median survival times < 2.5 months, indicating an inevitable poor outcome, despite presence of statistically significant differences, e.g. for primary tumour type. The clinical impact of prognostic scores would thus be very limited. Median survival was similar in historical studies of BSC. Best supportive care is a reasonable choice in patients with brain metastases and very short life expectancy, as also evident from prospective research.