Chronic pancreatitis (CP) is a disease defined by inflammation and fibrosis of the pancreas due to genetic, environmental, and other risk factors, with the development of persistent pathological responses to parenchymal injury or stress [1]. The risk factors for chronic pancreatitis include toxic-metabolic abnormalities – idiopathic, genetic, autoimmune, chronic, and obstruction [2].
A 25-year-old woman with a history of epilepsy for the last 20 years and chronic pancreatitis for the last 15 years was admitted in 2024 to the Department of Digestive Tract Diseases with nausea and severe epigastric pain radiating to the back.
At admission, blood tests showed an increase in white blood cell count of 11.9 G/l (reference range [RR]: 4.0–10.0 G/l), haemoglobin 12 g/dl (RR: 13.5–18.0 g/dl), C-reactive protein 63.1 mg/l (RR: 0.0–5.0 mg/l), procalcitonin assay 0.21 (RR: 0.12–0.36); liver enzymes: aspartate aminotransferase 13 units per litre (U/l) (RR: 0–40 U/l), alanine aminotransferase 9 U/l (RR: 0–40 U/l), serum pancreas enzymes: lipase 348 U/l (RR: 0–67 U/l), and amylase 107 U/l (RR: 22–80 U/l). Acute pancreatitis was diagnosed.
A computed tomography (CT) scan showed a dilated Wirsung duct and multiple calcifications in the pancreatic parenchyma. Due to chronic pancreatitis history, the patient was using 10,000 IU of pancreatin for main meals. The patient, in good condition, was discharged from our department after 7 days. The dose of pancreatic enzyme was increased to 32,000 UI for main meals and half dose for snacks. Also, a light diet and a healthy lifestyle were recommended. The patient is currently continuing treatment at the gastroenterology clinic at our hospital.
The history of chronic pancreatitis started with recurrent attacks of acute pancreatitis (RAP) 14 years prior. In the past, she was frequently hospitalised due to acute pancreatitis, repeatedly accompanied by epileptic seizures.
Genetic tests revealed that the patient carries SPINK1 (variation N34S) and CFTR mutations. The patient was adopted, and the family history is unknown as a result, so it is impossible to assess a hereditary predisposition. She underwent numerous endoscopic retrograde cholangiopancreatography (ERCP) procedures (2011, 2014, 2015), which caused serum amylase and lipase increase and no improvement in the course of the disease.
Due to persistent exacerbations of pancreatitis and lack of possibility for endoscopic treatment, in 2015 she underwent a Roux-en-Y anastomosis pancreatojejunostomy. One week later, the patient experienced blood in stools, so she was qualified for an urgent relaparotomy. This procedure revealed a haemorrhage from the distal loops of the small intestine and the large intestine. The source of bleeding was identified from both sections of the pancreas, which were coagulated. A separate incision was made to introduce a feeding tube, which was inserted into the pancreatic sections through a previously created Roux-en-Y loop. Despite these interventions, no reduction in the frequency of pancreatitis attacks was observed. After the operation, the patient was hospitalised due to exacerbations 8 times.
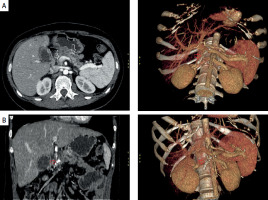
In 2017, when the patient was hospitalised in our department for the first time due to exacerbations of the disease, a CT scan was performed, which revealed a significant narrowing of the Wirsung track close to the pancreaticojejunostomy. Subsequently, in 2018, another surgical procedure was introduced using the modified Puestow method (Partington-Rochelle procedure). This is a drainage procedure in which the Roux loop is attached 5 cm distal into the previous pancreatic-intestinal anastomosis. This method causes a lower mortality and morbidity rate during operation compared to other surgical procedures [3]. After the second surgery, the number of AP attacks in this patient decreased (Figures 1 A, B).
Figure 1
Abdominal computed tomography (CT) scan. White arrows (A, B) point to the irregular dilated Wirsung duct. Red arrows (A, B) point to massive calcification in the pancreatic head. A shows a cross-section in the axial plane and 3D reconstruction. B is a cross-section in the coronal plane and 3D reconstruction. The acquisition was performed after intravenous administration of the contrast agent
The Puestow procedure decreases the severity of abdominal pain among patients with chronic pancreatitis. It was shown that during 5–10 years of observations, 70–80% of patients obtained permanent pain relief [3, 4]. In the presented patient, the second operation decreased the number of AP attacks to two between 2018 and 2024.
In the presented patient, mutations in the genes SPINK1(N34S) and CFTR(IVS8-5T) were detected. The correlation between the function of the proteins CFTR and SPINK1 and whether a single mutation in the CFTR or SPINK1 gene or both of them could cause chronic pancreatitis is a matter of debate [1, 5]. SPINK 1 is a gene that encodes a pancreatic secretory trypsin inhibitor. This inhibitor plays a vital role in preventing the premature activation of the zymogen and, therefore, protecting the pancreas from self-digestion [1, 6]. The prevalence of SPINK1 mutations in the general population is between 1% and 2% [5, 7]. Mutations in SPINK1 are frequently detected among Polish children with CP [8]. Polish research revealed that the frequency of alleles with SPIK1 mutation was represented in 8 of 52 paediatric patients with CP (7.7%) [8]. The most commonly observed variant of this mutation is N34S. The prevalence of N34S in patients with idiopathic chronic pancreatitis is between 21% and 32.5% [9–11].
With the SPINK1 mutation, the onset of pancreatitis occurs earlier (c.a. 10 years) than with other CP cases; the median age of onset of CP is 20.1 years [5]. Witt’s studies reveal that SPINK1 mutation was recognised in 22 out of 96 patients (23%), both adolescents and children (no relationship) [11]. In addition, N34S mutation was detected in 81% of those 22 patients [11]. In the control group of 279 healthy individuals, the SPINK1 mutation was found only in one person, more precisely in a person with the N34S mutation in one allele [11]. The CFTR gene encodes a protein that functions in epithelial cells and plays a vital role in driving bicarbonate secretion [1]. Either type of CTR mutation may increase the risk of pancreatitis, so not only cystic fibrosis patients are at risk of developing CP [1, 12]. Among patients who have both CFTR and SPINK 1 mutation, there is a 900-fold increased risk of pancreatitis [12].
Our patient had the IVS8-5T variant (5T polymorphism in intron 8), which causes less efficient splicing without intron 9 [13], and below normal CFTR [14]. The correlation between class V mutations (including the IVS8-5T polymorphism) of the CFTR mutation classification in CP is debatable. However, several studies claim that there is a link between the 5T allele in the CFTR gene and the occurrence of chronic pancreatitis [14]. Sharer suggests that the prevalence of IVS8-5T in patients with CP is 10.4% (14 of 134) and is two times higher than in the general population, which is 5% [14]. Still, the author did account for patients with alcoholic etiology.
In Polish research, the IVS8-5T variant was detected in 26 out of 260 children with CP (10%), which is more common compared to the general population. however, in the case of 15 children it did not reveal any other involved factors [13]. CFTR mutations like Phe 508del/- and Dele-2,3 (21 kb) were observed in 20 out of 260 (7.6%); however, having excluded other agents, there were only 10 children with the mutations mentioned above [13]. Nevertheless, a difference in the course of disease between these two groups was not noticed.
The second possible reason for the frequent exacerbations of the disease in this case is taking valproic acid for epilepsy. The patient had suffered from epilepsy since the age of 5 years and had been treated with this drug since childhood.
The study from the Children’s Hospital of Wisconsin showed that 22 children treated with valproic acid within a medium time of 32 months developed chronic pancreatitis [15]. In other multiple sources, the time between starting valproic acid treatment and developing CP was, on average, from 3 to 24 months [16]. However, it was emphasised that there is no connection between the serum level of valproic acid and pancreatitis occurrence and severity of the disease [16]. In addition, valproic acid causes the recurrence of pancreatitis [15]. The analysis of data collected from 73 published studies from 1979 to 2023 conducted in a group of 125 patients (83 children and 42 adults) with epilepsy showed that recurrence of pancreatitis occurred in 16 out of 19 patients re-exposed to valproic acid (84% of patients) [16]. Due to pancreatitis exacerbations, the patient’s treatment was modified. Instead of taking valproic acid, she was treated with levetiracetam, carbamazepine, and vigabatrin. Nevertheless, this change has not decreased the number of attack seizures or improved the patient’s condition. For this reason, the patient is still being treated with valproic acid but at a higher dose. Muller’s studies, involving a group of 209 patients with SPINK1 mutation, indicated that these individuals exhibit signs of the disease more persistently and are more likely to develop pancreatic calcifications compared to patients suffering from chronic pancreatitis with other aetiologies [5].
In addition, patients with SPINK1 mutations have a 53–87-fold higher risk of developing pancreatic cancer (PDAC) compared to the general population and a 12-fold higher compared to idiopathic chronic pancreatitis [5, 9].
According to Polish Pancreatic Club guidelines, CP patients should take pancreatic enzymes in a dose of c.a 30,000 IU for main meals and half this dose for snacks, to prevent exocrine pancreatic insufficiency [17]. Coming back to our patient, the latest CT scan revealed many calcifications of about 7 mm in the dilated Wirsung tract. ERCP treatment could be introduced to remove stones from the Wirsung track when associated with pain. Our case points out the difficulties in cases of pancreatitis in young patients with multiple aetiological factors, which should be carefully monitored and treated in high-volume pancreatic centres.









