An increase in cardiac troponin T (cTnT) values after transvenous lead extraction (TLE) occurs in nearly all patients (98%). Ischemic heart disease is not associated with the level of cTnT release during TLE. The factors most strongly associated with an increase in cTnT level during TLE were age of the extracted leads and multiple indicators of TLE complexity. The magnitude of the increase in cTnT level during TLE does not affect occurrence of major complications, peri-operative mortality, or long term survival. ΔcTnT8 can be considered a valuable retrospective indicator of the complexity and aggressiveness of the TLE procedure.
Introduction
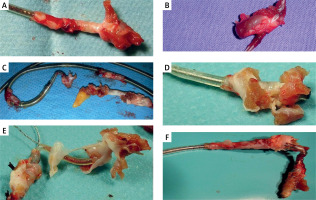
Transvenous lead extraction has a permanent place in the treatment and prevention of complications related to the presence of intracardiac leads [1–3]. After years within the cardiovascular system, the leads become encased in connective scar tissue that permanently connects them to the walls of the veins and cardiac structures [4, 5]. Removal of “ingrained” leads requires the use of special dilatation catheters that are slipped over the removed lead and free it from connective tissue growths [1–3]. The procedures are performed under the guidance of fluoroscopy and transoesophageal echocardiography (TEE) [6, 7]. We only observe the effectiveness of dilation. Accidental damage to the walls of large veins and the heart is recognized based on clinical and echocardiographic symptoms of bleeding [1–3, 6–8]. Sometimes, in addition to connective tissue, we also see fragments of muscle tissue on the removed lead [4] (Figure 1).
Figure 1
Examples of extracted leads on the operating table. Fragments of connective scar tissue and possibly fragments of endocardium are visible as fragments of muscle tissue originating from the right ventricular wall (A–F). The muscle tissue defects were so small that they were invisible in the TEE image, and none of the patients developed cardiac tamponade. In some cases, a fragment of epicardial fat tissue was observed at the tip of the lead in the subepicardial location (C)
We are unable to assess the occurrence of minor damage that does not cause visible clinical consequences and thus more fully assess the degree of invasiveness of the procedure. Although troponin level determination is mainly used for diagnosis and differentiation of chest pain and acute coronary syndromes [9], it has been reported to be a valuable marker of heart damage during percutaneous [10] and surgical [11, 12] coronary artery procedures. It has been reported that troponin level elevations may occur after transcatheter aortic valve implantation (TAVI) procedures [13] and ablations of the arrhythmia substrate [14, 15]. The observation of this phenomenon in patients after transvenous lead extraction (TLE) [16] prompted us to conduct studies on a sufficiently large population of consecutive (without any exceptions) patients undergoing TLE in order to investigate the determinants of troponin release during TLE.
Aim
The general aim of the study was to identify factors influencing the intensity of troponin release during and after TLE, taking into account patient-dependent factors (age, presence of ischemic heart disease – IHD), cardiac implantable electronic device (CIED) system-dependent factors (duration and number of leads being extracted, abandoned leads), and procedure-dependent factors (dilatation time, tools used, procedure complexity, unexpected technical difficulties, hypotension, significant traction on cardiac structures observed in TEE). An additional aim of the study was to investigate whether troponin release could be an additional factor allowing for retrospective assessment of the complexity, difficulties, and potentially damaging effect of the procedure and to investigate whether the increase in troponin levels after TLE (Tp8 levels) has any prognostic significance for medium-term survival.
Material and methods
Study population
The study population consisted of 166 consecutive patients, in whom 262 pacemaker or ICD leads were extracted. All 166 TLE procedures performed between March 2021 and December 2022 by one very experienced operator in a one high-volume centre were reviewed. The study included all consecutively operated patients, there were no exclusion criteria. In the study group an average age was 68.5 years (median: 72.00), 68.67% males and 15.66% of procedures were performed for infectious reasons.
Definitions
Indications for TLE, procedure effectiveness and complications were defined according to recent TLE recommendations (2017 Heart Rhythm Society [HRS] consensus and 2018 European Heart Rhythm Association [EHRA] guidelines) [1, 2]. The indications were: 1) mechanical lead damage (electric failure), 2) non-damaged lead dysfunction (exit/entry block, tip dislodgement or extracardiac pacing), lead perforation of cardiac wall, 3) other indications: abandoned lead, threatening/potentially threatening lead, magnetic resonance imaging (MRI) indication, cancer, pocket pain, cessation of indication for ICD/CRT [17], 4) recapture of venous access (symptomatic occlusion, SVC syndrome, lead replacement/upgrading) [18], and 5) local pocket infection or systemic infection (with or without pocket infection) [19].
Lead extraction procedure
TLE indications, procedure effectiveness and complications were estimated according to recent TLE recommendations (2017 HRS and 2018 EHRA recommendations) [1, 2, 19]. The efficacy of TLE was expressed as the percentage of procedural success, which was defined as the removal of all targeted leads and lead material from the vascular space with the absence of any permanently disabling complication or procedure-related death [1–3].
TLE procedures were performed in a hybrid room, under general anesthesia with a mandatory arterial line (radial access) for invasive blood pressure monitoring and with TEE monitoring [7, 8].
Major complications of TLE were defined as those that were life-threatening, resulted in significant or permanent disability or death, or required surgical intervention [1–3].
Procedure complexity
Procedure complexity was expressed as lead extraction time (“sheath-to-sheath time”) and average time of single lead extraction (sheath-to-sheath time/number of extracted leads). These individually calculated values do not include the fluoroscopy time for additional activities (implantation of the electrode for temporary stimulation, implantation of new electrodes), but they are similar to the fluoroscopy time estimated by other authors [20–22]. The second indicator of procedure complexity was the need for use of second-line tools and advanced tools [23–26]. The third one was occurrence of unexpected technical problems (block in venous lead entry region, extracted lead break, collapse and twist polypropylene sheath, necessity for change of venous approach) that are not complications but make the procedure more complex [25, 26]. The fourth, newly introduced measure was the Complex Indicator of Difficulty score of the TLE (CID-TLE), which comprises: global sheath-to-sheath time for extraction of all leads > 20 min (2 points); average duration of single lead sheath-to-sheath extraction time > 12 min (2 points); and need for use of a metal sheath or Evolution/TightRail, an alternative venous entry approach, or lasso or basket catheters (1 point each). The sum of points was the value of CID-TLE. A procedure was considered as difficult when CID-TLE ≥ 2 [23].
Blood pressure drop and prolonged hypotension
Arterial blood pressure was monitored continuously using an invasive method. The baseline blood pressure value was taken from the results of measurements obtained after the patient was put under general anesthesia, and the hemodynamic parameters were stabilized. A significant drop in blood pressure was defined as a sudden decrease to values under 100 mm Hg or a sudden reduction of more than 10% of the initial value. Prolonged hypotension was defined as a sustained decrease in blood pressure lasting at least 1 min or requiring additional intervention.
Dataset and statistical methods
Laboratory tests for troponin T were performed on blood samples collected before the procedure (cTnT0), at 2 h (c-TnT2, CK-MB2), and 8 h after the procedure was completed (cTnT8). The following immunoassays were used for quantitative determination of troponin and creatine kinase MB isoenzyme levels in vitro: Elecsys Troponin T hs and Elecsys CK-MB, performed on the Cobas analyzer (Roche, Switzerland, Basel). No other tests unrelated to the routine pre- and postoperative laboratory assessment were performed. Before the procedure, all patients underwent standard cardiological (including transthoracic echocardiography – TTE) and anesthetic examination. ECG was performed before and after TLE. None of the patients had an exacerbation of coronary artery disease.
Statistical analysis
Due to nonlinear distribution, continuous data are presented as median and interquartile range. Continuous data were compared with the Mann-Whitney U test. Categorical data are presented as number and percentage and were compared with the c2 test with Yates’s correction if appropriate. To determine factors influencing troponin T release during TLE, uni- and multivariable logistic regression was used. Unrelated data were included in the multivariable regression analysis. The 1-year survival impact of troponin T was determined with Cox regression. Survival curves were estimated by the Kaplan-Meier method, and the difference between the curves was evaluated using the log-rank test. To assess the association between cTnT concentration and global lead dilatation time and age of oldest extracted lead, the Spearman r correlation was used. A two-tailed p-value < 0.05 was considered statistically significant. Statistical analysis was performed using Statistica 13.1 PL (TIBCO, Krakow, Poland).
Results
Patient clinical characteristics, CIED system and history of pacing, data on targeted leads of groups with ICD lead extraction, pacemaker (PM) lead extraction and all patients/procedures are presented in Table I.
Table I
General characteristics of study group
[i] IQR – interquartile range, TLE – transvenous lead extraction, MI – myocardial infarction, ICD – implantable cardioverter defibrillator, cTnT0 – troponin T concentration before TLE, cTnT8 – troponin T concentration 8 h after TLE, ΔcTnT8 – delta cTnT; difference of concentrations cTnT8 and cTnT0, ΔcTnT [%] – delta cTnT; difference of concentrations cTnT8 and cTnT0 expressed in % [(cTnT8 - cTnT0)/cTnT0 × 100].
Table I presents information on the studied population of 166 unselected (consecutive) patients who underwent TLE. The relatively low percentage of infectious indications (15.66%) is noteworthy, which results from the adopted strategy of not leaving abandoned leads. Also of note is the significantly longer implant duration than in the reported large populations [3, 27, 28], the rare use of second-line instruments, and very good TLE outcomes (98.80% procedural success and 0.60% major complications). Half of the patients showed a troponin T level > 0.014 µg/l before the procedure, and in almost 100% it was elevated; on average (137.7 ±150.0 µg/l) ten times higher than the norm. The increase in troponin level (DcTnT8) was on average 926.9% and nearly 400% of the median.
In accordance with the objectives of the study, in the following tables we focused on trying to determine the factors predisposing to a very pronounced release of troponins during and after TLE (Tables II and III).
Table II
Potential determinants of cardiac troponin T concentration increase as a result of transvenous lead extraction procedure
| Parameter | ΔcTnT8 ≤ median (0.053 µg/l) Median (IQR) *Mean ± SD | ΔcTnT8 > median (0.053 µg/l) Median (IQR) *Mean ± SD | Mann-Whitney U test χ2 test P-value |
|---|---|---|---|
| Number of patients | 83 (100.0) | 83 (100.0) | |
| Potential patient and system risk factors | |||
| Ischemic heart disease (with or without prior MI) | 51 (61.45%) | 46 (55.42) | 0.54 |
| Number of procedures before lead extraction | 1 (1) | 2 (2) | 0.018 |
| Two or more CIED procedures before TLE | 34 (40.96%) | 53 (63.86%) | 0.003 |
| Oldest extracted lead body dwelling time in the patient [months] | 77.00 (82.00) | 144.0 (110.0) | < 0.001 |
| Global extracted lead dwell time [years] | 7.92 (9.40) | 21.25 (22.67) | < 0.001 |
| Potential procedure-related risk factors | |||
| Number of extracted leads per patient | 1 (2) | 2 (2) | 0.14 |
| Extraction of abandoned lead(s) (any) | 1 (1.20%) | 2 (2.41%) | 0.54 |
| Extraction of lead with unnecessary (large) lead loop in the heart | 18 (21.75%) | 30.00 (36.15%) | 0.031 |
| LECOM index [points] | 7.08 (7.27) | 19.24 (28.94) | < 0.001 |
| Potential procedure-related risk factors | |||
| Procedure duration (sheath-to-sheath): global lead dilatation time [min] | 5.0 (6.0) | 14.0 (20.0) | < 0.001 |
| Mean time of single lead extraction (sheath-to sheath/number of extracted leads) [min] | 4.0 (2.5) | 8.0 (13.0) | < 0.001 |
| Global lead extraction time [min] | 6.0 (7.0) | 13.0 (13.0) | < 0.001 |
| Global number of technical problems in the group of patients [number]* | 1 (1) *1.13 ±0.35 | 1 (1) *1.59±0.73 | 0.043 |
| Occurrence of two or more technical problems | 1 (1.20%) | 10 (12.00%) | 0.004 |
| Use of tools other than polypropylene sheaths (any) | 5 (6.02%) | 28 (33.73%) | 0.001 |
| CID score index [points; 0–5] | 0 (0) *0.12 ±0.45 | 0 (2) *1.07 ±1.35 | < 0.001 |
| CID index 2 or more points | 2 (2.41%) | 33 (39.76%) | 0.001 |
| Significant blood pressure drop during lead dilatation | 15 (18.29%) | 35 (42.17%) | 0.002 |
| Hypotonia during lead dilatation > 1 min | 7 (8.43%) | 26 (31.33%) | 0.001 |
| Traction of cardiac structures visible in TEE | 0 (0,00%) | 18 (21.69%) | 0.001 |
| CK-MB2 concentration [µg/l] | 3.17 (2.37) | 7.56 (7,64) | < 0.001 |
| Number of patients with CK-MB2 concentration > 5 µg/l | 12 (14.46%) | 56 (67.47%) | 0.001 |
| One-year survival | |||
| Alive at 1-year follow-up | 70 (84.33%) | 69 (83.13%) | 0.83 |
| Deceased during 1-year follow-up | 13 (15.67%) | 14 (16.87%) | 0.83 |
cTnT – troponin T, ΔcTnT8 – delta cTnT; difference of concentrations cTnT8 and cTnT0, MI – myocardial infarction, CIED – cardiac implantable electronic devices, TLE – transvenous lead extraction,
* mean ± SD – for data with the same median values, the mean with standard deviation is also presented, CID – Complex Indicator of Difficulty of TLE procedure, LECOM – Lead Extraction COMplexity index, TEE – transesophageal echocardiography, CK-MB2 – concentration of MB isoenzyme of creatine kinase in plasma 2 h after TLE.
Table III
Values of heart damage indicators (cTnT8, DcTnT8, and CK-MB2) according to selected clinical factors and the course of TLE
[i] IQR – interquartile range, IHD – presence of ischemic heart disease (with or without prior MI), cTnT0 – troponin T concentration before TLE, cTnT8 – troponin T concentration 8 h after TLE, ΔcTnT8 – delta cTnT; difference of concentrations cTnT8 and cTnT0, ΔcTnT [%] – delta cTnT; difference of concentrations of cTnT8 and cTnT0 expressed in % [(cTnT8 - cTnT0)/cTnT0 × 100], CK-MB2 – concentration of MB isoenzyme of creatine kinase in plasma 2 h after TLE, TLE – transvenous lead extraction, TEE – transesophageal echocardiography, CID – Complex Indicator of Difficulty of TLE procedure.
We assumed that the degree of impact of TLE on the myocardium is expressed by the increase in cTnT concentration. In order to draw preliminary conclusions, we compared the potential determinants of cTnT release in two groups of patients selected according to the median DcTnT8 value (0.053 µg/l). The results are presented in Table II.
The group of patients with DcTnT8 levels over the median was characterized by more CIED-rated procedures before lead extraction (medians: 2 and 1), a slightly higher but nonsignificant number of leads removed per patient, more frequent extraction of lead with unnecessary (large) lead loop in the heart (36.15% vs. 21.75%), significantly longer oldest extracted lead body dwelling time in the patient (medians: 144.0 vs. 77.00 months), and nearly three times longer global extracted leads dwell time (medians: 21.25 vs. 7.92 years). Therefore, the global lead dilatation time (sheath-to-sheath time) was nearly three times longer (medians: 14.00 vs. 5.00 min), technical problems occurred one third more often per patient (1.59 vs. 1.13), and the accumulation of technical problems occurred ten times more often (12.00% vs. 1.20%). Therefore, in patients with a higher release of troponin T, second-line tools (other than polypropylene sheaths) were used more often (33.73% vs. 6.02%) and the retrospectively calculated CID score was on average ten times higher (1.07 vs. 0.12). Significant blood pressure drops and hypotension prolonged over 1 min (due to necessary traction on the lead) occurred several times more often (42.17% vs. 18.29% and 31.33% vs. 8.43%), and the echocardiographer more often noted dangerous traction of cardiac structures in his database (21.69% vs. 0.00%). In patients with a higher DcTnT8, the CK-MB2 level was significantly (two-fold) higher (medians: 7.56 vs. 3.17 µg/l) and CK-MB2 values > 5 µg/l were observed nearly five times more often (67.47% vs. 14.46%). Annual mortality in both groups was similar, which indicates that the increase in cTnT level after TLE has no prognostic significance.
After obtaining some preliminary information, we decided to analyze in more detail the values of heart damage indicators (cTnT8, DcTnT8, and CK-MB2) in relation to some selected clinical factors and the course of the TLE procedure (Table III).
First, we investigated whether the presence of ischemic heart disease could affect the increased release of troponins during maneuvers related to lead extraction. However, the analysis showed that there is no such relationship; moreover, a higher percentage increase of cTnT release in the group without CHD was detected. In contrast, a clear trend was observed between the number of removed leads and the increase in cTnT.
Global lead dilatation time well represents the degree of complexity and laboriousness of the procedure. For the purposes of the analysis, the median value (8.00) of this indicator was used. The increases in troponin levels and CKMB2 values were several times (and significantly) higher in cases of longer lead dilatation time, with median DcTnT8 values of 0.12 vs. 0.03 µg/l and median CKMB2 values of 6.71 vs. 3.83 µg/l.
The occurrence of any technical problems – block in venous lead entry region, extracted lead break, collapse or twisting of the polypropylene sheath, of the need to change the venous approach – making the procedure longer and more complex was associated with four times higher DcTnT8 (medians: 0.17 vs. 0.04 µg/l) and almost double the CK-MB2 level (medians: 7.64 vs. 4.39 µg/l). A sudden drop of arterial blood pressure during dilatation is most often the result of too strong tension of the removed lead, resulting in a decrease in the volume of the right ventricle or occlusion of the lumen of the superior vena cava at its connection with the right atrium. Traction on cardiac structures and a sudden decrease in arterial pressure causes a temporary decrease in coronary perfusion. As can be seen from the table, it was associated with an increase in DcTnT8 (medians: 0.11 vs. 0.04 µg/l) and CK-MB2 (medians: 6.96 vs. 4.13 µg/l). Hypotension lasting more than 1 min had a similar effect.
Significant traction of cardiac structures during the necessary but sometimes excessively strong tensioning of the removed lead, not always causing sudden drops in blood pressure but preceding damage to the cardiac wall, is visible during monitoring of the procedure using TEE. In cases where the echocardiographer noted this phenomenon, the increase in troponin level (DcTnT8) was significantly higher (medians: 0.20 vs. 0.05 µg/l), as were the CK-MB2 values (medians: 12.30 vs. 4.18 µg/l). The complexity, technical difficulties, and laboriousness of the TLE procedure and in a sense its aggressiveness are reflected retrospectively by calculating the CID score. In patients with a CID score ≥ 1 point, the value of DcTnT8 was significantly higher (medians: 0.12 vs. 0.04 µg/l), similarly to CK-MB values (medians: 7.53 vs. 4.0 µg/l) (Table III).
Linear logistic regression analysis showed that the factors most responsible for cTnT release from the myocardium during TLE are age of the oldest extracted leads – with a 16.5% increase in the probability of cTnT exceeding 0.053 µg/l per year of lead age (OR = 1.165; 95% CI 1.079–1.258; p < 0.001) – and global lead dilatation time – with a 255% increase in probability per additional minute (OR = 2.552; 95% CI (1.102–5.912), p < 0.001) (Table IV).
Table IV
Uni- and multivariable analysis of factors associated with an increase above the median value (0.053 µg/l) of cTnT concentration measured 8 h after TLE (ΔcTnT8)
| Linear logistic regression analysis identifying the main determinants of cTnT release | Univariable regression | Multivariable regression | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | OR | 95% CI | P-value | |
| Ischemic heart disease [y/n] | 1.241 | 0.648–2.379 | 0.51 | 1.223 | 0.523–2.861 | 0.63 |
| Dwell time of oldest extracted lead* [by 1 year] | 1.191 | 1.106–1.281 | < 0.001 | 1.182 | 1.089–1.283 | < 0.001 |
| Global (cumulative) age of extracted leads in the patient* [by 1 year] | 1.092 | 1.052–1.134 | < 0.001 | |||
| CID score [by 1 point] | 2.170 | 1.458–1.458 | < 0.001 | 2.561 | 1.247–5.261 | 0.009 |
| Significant blood pressure drop appearance [y/n] | 3.232 | 1.684–6.208 | < 0.001 | |||
| Hypotonia > 1 min appearance [y/n] | 5.107 | 2.057–12.68 | < 0.001 | 1.844 | 0.792–4.297 | 0.15 |
| Traction of cardiac structures visible in TEE [y/n] | 21.12 | 2.697–165.4 | < 0.001 | 5.304 | 0.604–46.61 | 0.99 |
| Technical problems during TLE [y/n] | 2.785 | 1.182–6.562 | < 0.001 | 0.796 | 0.288–2.199 | 0.65 |
| Global lead dilatation time [> 8 min] | 5.607 | 2.860–10.99 | < 0.001 | |||
| Number of leads being extracted [by 1 lead] | 1.537 | 0.900–2.624 | 0.11 | 1.197 | 0.524–2.735 | 0.66 |
The associations of the above parameters (DcTnT8 vs. age of oldest extracted lead and vs. CID index together with the Spearman r correlation coefficients) are presented graphically in Figure 2 A.
Figure 2
Influence of age of oldest extracted lead and global lead dilatation time on the value of ΔcTnT8 and influence of cTnT0 and ΔcTnT8 values on survival after TLE in 1-year follow-up. A – Associations of ΔcTnT8 [µg/l] with age of oldest extracted lead and CID points. B – Kaplan-Meier survival analysis
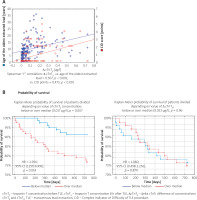
Kaplan-Meier and Cox regression survival analysis confirmed the negative prognostic role of primary value of cTnT concentrations. Patients with cTnT concentration over 0.017 µg/l had a worse prognosis expressed as nearly four times higher risk of death in 2-year follow-up (HR = 2.956; 95% CI: 1.250–6.991; p = 0.014). However, the increase in cTnT concentration during TLE had no prognostic significance (HR = 1.060; 95% CI: 0.498–2.256; p = 0.87) (Figure 2 B).
Discussion
We found that the increase in troponin level (DcTnT8), similarly to CK-MB2 values, were significantly higher after extraction of older leads, after longer global dilatation time, when unexpected technical problems occurred, there was a sudden arterial blood pressure drop during dilatation or prolonged hypotension, the echocardiographer reported traction of cardiac structures, and the Complex Indicator of Difficulty of TLE procedure score was 2 or more points. The increase in cTnT level is represented by DcTnT8, expressed as absolute values (µg/l) and as a percentage of the initial values (cTnT0).
This is the first study on the release of cardiac troponins during transvenous lead removal, and we do not have other studies to compare the results. We can only refer to the observations of an increase in troponin levels after TAVI procedures [13] and ablations of the arrhythmia substrate [14, 16]. The massive release of cardiac troponins after percutaneous [10] and surgical [11, 12] coronary artery procedures may have a slightly different (ischemic) mechanism. Cardiac troponin T is a sensitive biomarker of myocardial injury and may be released not only in the setting of overt myocardial necrosis but also as a consequence of micro- or minimal myocardial injury. In patients with non-ST-elevation myocardial infarction (NSTEMI), troponin T elevation most commonly reflects ischemic injury related to coronary plaque instability and incomplete coronary occlusion; however, small increases in troponin T may also result from micro-mechanical damage to cardiomyocytes, without extensive or transmural necrosis.
Such minimal myocardial injury may be caused by transient ischemia, microvascular dysfunction, increased myocardial wall stress, or repetitive mechanical strain, leading to reversible cell membrane disruption and leakage of cytosolic troponin pools. According to the Fourth Universal Definition of Myocardial Infarction, even small troponin elevations indicate myocardial injury, and when accompanied by clinical evidence of ischemia, fulfill the criteria for NSTEMI. Importantly, low-grade troponin T elevations associated with micro- or minimal myocardial injury have been shown to carry adverse prognostic implications, emphasizing their clinical relevance in risk stratification [9, 28–30].
All of our analyses have shown a link between the increase in cTnT levels (DcTnT8) and age of extracted lead and the degree of complexity of the procedure in the absence of any association with the existence of any form of ischemic disease. The increase in DcTnT8 value can be considered a retrospective, fairly reliable indicator of the aggressiveness of the procedure.
The practical conclusion from the analyses performed is to demonstrate and confirm that the increase in cTnT and CK-MB levels in patients after TLE is a “typical”, understandable and natural phenomenon, which at the same time is a limitation of the use of these indicators in the diagnosis of chest pain in this group for a certain unknown period.
The studies certainly require continuation: determination of marker values after a few and a dozen or so days, i.e. determination of the moment (and conditions) of their normalization.
Study limitations
This is a pilot study. We have described the circumstances of occurrence of the observed phenomenon without description of the dynamics of cTnT value normalization in the following days after TLE. We were unable to examine the association between major complications of TLE and increase of troponin levels, because they occur rarely (under 1% of procedures). In none of the patients with a significant increase of troponin levels was acute coronary syndrome suspected, and in none of them was coronary angiography performed, due to the lack of indications.
Conclusions
An increase in cTnT values after TLE occurs in nearly all patients (98%). Ischemic heart disease is not associated with the degree of cTnT release during TLE. The factors most strongly associated with the magnitude of the increase in cTnT level during TLE were the age of the extracted leads and multiple indicators of the complexity of the TLE procedure. The magnitude of the increase in cTnT level during TLE does not affect occurrence of major complications, peri-operative mortality, or long-term survival. DcTnT8 can be considered a valuable retrospective indicator of the complexity and aggressiveness of the TLE procedure.







