In this study, we examined 431 young patients (aged ≤ 50 years) who had ST-elevation myocardial infarction and multivessel coronary artery disease. We compared two treatment approaches: culprit-only percutaneous coronary intervention versus complete revascularization. We found that complete revascularization significantly reduced major adverse cardiac events and mortality. Kidney dysfunction and reduced left ventricular function were strong predictors of poor outcomes, while the use of statins and b-blockers appeared protective. These results suggest that even in younger patients, addressing all significant blockages and optimizing medical therapy can meaningfully improve survival and long-term heart health.
Introduction
Cardiovascular diseases continue to cause the highest levels of morbidity and mortality throughout the globe [1]. ST-segment elevation myocardial infarction (STEMI), which causes myocardial ischemia, is one of the best recognized forms of acute coronary syndrome (ACS) and occurs due to complete or near-complete occlusion of coronary arteries [2]. Prompt revascularization is the only viable treatment in STEMI, which improves outcomes, preserves left ventricular ejection fraction (LVEF), and extends survival [3].
Multivessel coronary artery disease (CAD) is common among patients with STEMI [4]. In this population, the standard of care has long been considered to be primary percutaneous coronary intervention (PCI) targeting only the culprit lesion [5]. This approach shortens procedures and contrast exposure, which may be particularly advantageous for critically ill patients [4, 5]. However, residual stenoses in non-culprit arteries may predispose patients to recurrent ischemic events, repeat revascularizations, or adverse long-term outcomes. As such, recent randomized controlled trials have challenged the traditional culprit-only strategy, suggesting that complete revascularization (PCI for culprit and significantly stenosed non-culprit vessels) may improve outcomes [6–9]. Studies such as the PRAMI, CvLPRIT, DANAMI-3–PRIMULTI, and most notably the COMPLETE trial, have demonstrated that staged or immediate complete revascularization reduces the composite risk of cardiovascular death and myocardial infarction in STEMI patients with multivessel disease [6–9]. As a result, the 2021 ACC/AHA/SCAI (American College of Cardiology/American Heart Association/Society for Cardiovascular Angiography and Interventions) revascularization guidelines provide a Class 1A recommendation for complete revascularization carried out in a staged fashion [10].
On the other hand, there are still uncertainties regarding revascularization strategies in younger STEMI patients, largely due to limited data in this specific population. Younger individuals often present with fewer comorbidities, different plaque morphology, and distinct clinical profiles compared to older populations [11], which might alter the outcomes of complete revascularization.
Aim
We therefore conducted a retrospective study to compare cardiovascular outcomes between culprit-only and complete revascularization strategies in young STEMI patients with multivessel CAD. Additionally, we aimed to identify clinical, procedural, and demographic factors associated with adverse outcomes in this population, which can address knowledge gaps in this specific population.
Material and methods
Study approval and design
This retrospective cohort study was approved by the Non-Interventional Clinical Research Ethics Committee of Haseki Training and Research Hospital (Approval date: 14.05.2025, no: 56-2025). The study was conducted in accordance with the principles of the Declaration of Helsinki and relevant ethical guidelines for medical research involving human subjects. All data were collected, stored, and analyzed with strict adherence to institutional confidentiality protocols and data protection policies.
The present research was designed as a single-center, retrospective cohort study conducted at the Cardiology Department of Haseki Training and Research Hospital (Istanbul, Turkey). The study included patients hospitalized with a diagnosis of STEMI between January 1, 2022, and December 31, 2023.
Inclusion criteria: Patients aged 18–50 years hospitalized with a diagnosis of STEMI and multivessel CAD (≥ 2 vessels with significant stenosis) between January 1, 2022, and December 31, 2023, who underwent successful primary PCI.
Exclusion criteria: Refusal of treatment, incomplete follow-up, single-vessel CAD, age over 50 years or under 18 years, pre-procedural death, hemodynamic instability or contraindications precluding PCI, and unsuccessful PCI. The upper age limit of 50 years was selected based on prior literature, where cutoffs of 45–50 years have been consistently employed to define young patients with acute coronary syndromes [12].
Treatment and classification
All patients underwent urgent coronary angiography and PCI targeting the infarct-related artery. Treatment decisions regarding staged revascularization of non-culprit lesions were made by the attending interventional cardiologist, based on angiographic findings, clinical judgment, and institutional protocols consistent with contemporary European Society of Cardiology (ESC) guidelines. Factors influencing the decision to defer non-culprit intervention included hemodynamic instability, unfavorable lesion characteristics (severe calcification, tortuosity, or diffuse disease), chronic total occlusions, and operator judgment. As this was a retrospective study, systematic documentation of these decision factors was not available for all patients.
Patients were categorized into two groups: those who underwent culprit-only PCI, and those who received complete revascularization, defined as additional PCI for all significantly stenotic non-culprit vessels within 30 days of the index procedure. Patients with prior CABG (n = 6) were included if they had significant stenoses in at least two vessels, including native coronary arteries or bypass grafts supplying native territories, thereby meeting criteria for multivessel disease.
STEMI-related definitions
STEMI characteristics were extracted from emergency department documentation and initial electrocardiograms. Infarct territory was defined as anterior, inferior, lateral, or posterior according to standard 12-lead electrocardiogram criteria. Culprit vessels (left anterior descending, circumflex, or right coronary artery) and the number of affected vessels were determined from coronary angiography reports, which were interpreted by two experienced cardiologists blinded to clinical outcomes.
Data collection
Patient data were collected retrospectively from the institutional electronic medical record system using a structured data abstraction form. Demographic and lifestyle characteristics were recorded, including age, sex, immigration status, and smoking status. Smoking was further classified as non-smoker, passive smoker, ex-smoker, or active smoker, based on documentation in admission and follow-up notes.
Baseline clinical comorbidities were identified through diagnoses recorded according to the International Classification of Diseases (ICD) in electronic patient files.
Procedural data included timing parameters such as symptom-onset-to-revascularization time and door-to-revascularization time (emergency arrival to device activation), number and total length of stents used, and number of vessels treated. The number of stents and total stent length (in mm) were extracted from PCI procedural reports. Anatomical complexity was quantified using the SYNTAX score, calculated from baseline coronary angiograms by two experienced interventional cardiologists. Periprocedural pharmacological therapy was also recorded, including the use of antiaggregants, anticoagulants, b-blockers, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, lipid-lowering agents, calcium channel blockers, vasodilators, and diuretics.
Laboratory and imaging data included pre- and post-procedure serum creatinine levels (drawn within 24 h of PCI) and LVEF, which was evaluated by transthoracic echocardiography within 48 h after the intervention, using the Simpson’s biplane method in accordance with ESC echocardiography guidelines. All echocardiograms were performed by certified technicians and interpreted by cardiologists blinded to treatment group allocation.
Complications and follow-up
Major adverse cardiac events (MACE) were defined as a composite of all-cause mortality, heart failure hospitalization, urgent repeat PCI, urgent coronary artery bypass grafting (CABG), cardiogenic shock, high-grade atrioventricular block requiring pacing, cerebrovascular events, ventricular tachycardia, or ventricular fibrillation. Event definitions were based on standard ACC/AHA clinical trial endpoints. Follow-up data were obtained from outpatient visit records and telephone follow-up and included duration of follow-up in months and clinical endpoints.
Endpoints
The primary aim of this study was to compare cardiovascular outcomes between patients undergoing culprit-only PCI and those receiving complete revascularization. The primary outcome was the incidence of MACE as defined above. Secondary outcomes included all-cause mortality.
Sample size
The sample size was calculated using the ‘c2 test power analysis’ module in the PASS 11 Home software (Hintze, J. (2011). PASS 11. NCSS, LLC. Kaysville, Utah, USA. www.ncss.com). Based on the descriptive statistics from the article titled “A Comparison Between Culprit Versus Complete Revascularization in Diabetic Patients With Acute Myocardial Infarction” [13], the necessary targeted effect size was determined as 0.208. The minimum required sample size for our study to be completed with 95% confidence (a = 0.05) and 90% power was determined to be a total of 292 patients.
Statistical analysis
All analyses were performed using IBM SPSS Statistics for Windows, Version 27.0 (IBM Corp., Armonk, NY, USA). The level of statistical significance was set at p < 0.05. Categorical data were summarized as frequency (percentage). Descriptive statistics are presented as mean ± standard deviation for normally distributed continuous variables, and as median (25th percentile to 75th percentile: interquartile range, IQR) for non-normally distributed continuous variables. The assumption of normality was evaluated using histograms and Q-Q plots. Between-group analysis of continuous variables was performed using Student’s t-test or the Mann-Whitney U test, based on parametric assumptions. Categorical variables were analyzed using c2 tests or Fisher’s exact test or the Fisher-Freeman-Halton test. Multivariable logistic regression analysis (forward conditional) was performed to determine independent risk factors associated with MACE and mortality. The model included variables that were significant in univariate analyses.
Results
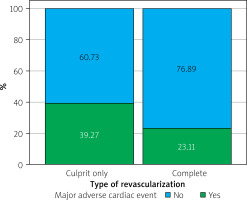
The study included a total of 431 patients with STEMI, who were divided into two revascularization groups: 219 (50.8%) patients underwent culprit-only revascularization, while 212 (49.2%) patients received complete revascularization. The median age of the overall cohort was 49 years (IQR: 46–50), with no significant difference between the two treatment groups (p = 0.06). The majority of patients were male (89.1%, n = 384), and the sex distribution was similar between the groups (p = 1.00). SYNTAX scores were comparable between the culprit-only and complete revascularization groups (p = 0.440), indicating similar anatomical complexity at baseline. Patients in the culprit-only group had a higher prevalence of prior CABG (p = 0.004). Complete revascularization was associated with significantly higher use of antilipemic drugs (p = 0.027). The culprit-only group experienced a substantially higher rate of MACE (p < 0.001) (Figure 1). The need for urgent CABG was more common in the culprit-only revascularization group (p = 0.020), whereas urgent repeat PCI rates did not differ significantly between groups (p = 0.23) (Table I).
Table I
Type of revascularization and patient characteristics
| Parameter | Type of revascularization | P-value | ||
|---|---|---|---|---|
| Total (n = 431) | Culprit-only (n = 219) | Complete (n = 212) | ||
| Age [years] | 49 (46–50) | 48 (45–50) | 49 (47–50) | 0.062‡ |
| Sex | ||||
| Female | 47 (10.90%) | 24 (10.96%) | 23 (10.85%) | 1.000§ |
| Male | 384 (89.10%) | 195 (89.04%) | 189 (89.15%) | |
| Immigrant | 34 (7.89%) | 19 (8.68%) | 15 (7.08%) | 0.662§ |
| Smoking | ||||
| Non-smoker | 39 (9.09%) | 16 (7.37%) | 23 (10.85%) | 0.526§ |
| Passive smoker | 23 (5.36%) | 11 (5.07%) | 12 (5.66%) | |
| Ex-smoker | 69 (16.08%) | 33 (15.21%) | 36 (16.98%) | |
| Active smoker | 298 (69.46%) | 157 (72.35%) | 141 (66.51%) | |
| Previous comorbidities | ||||
| Hypertension | 152 (35.27%) | 78 (35.62%) | 74 (34.91%) | 0.877§ |
| Diabetes mellitus | 151 (35.03%) | 76 (34.70%) | 75 (35.38%) | 0.883§ |
| Hyperlipidemia | 220 (51.04%) | 114 (52.05%) | 106 (50.00%) | 0.670§ |
| COPD | 17 (3.94%) | 9 (4.11%) | 8 (3.77%) | 1.000§ |
| Coronary artery disease | 78 (18.10%) | 44 (20.09%) | 34 (16.04%) | 0.274§ |
| CABG | 9 (2.09%) | 9 (4.11%) | 0 (0.00%) | 0.004# |
| Peripheral artery disease | 18 (4.18%) | 8 (3.65%) | 10 (4.72%) | 0.756§ |
| Cerebrovascular disease | 12 (2.78%) | 6 (2.74%) | 6 (2.83%) | 1.000§ |
| Pulmonary embolism | 2 (0.46%) | 1 (0.46%) | 1 (0.47%) | 1.000# |
| Deep vein thrombosis | 4 (0.93%) | 2 (0.91%) | 2 (0.94%) | 1.000# |
| Renal diseases | 12 (2.78%) | 8 (3.65%) | 4 (1.89%) | 0.411§ |
| Malignancy | 7 (1.62%) | 4 (1.83%) | 3 (1.42%) | 1.000# |
| STEMI territorya | ||||
| Anterior | 144 (33.41%) | 74 (33.79%) | 70 (33.02%) | 0.865§ |
| Lateral | 27 (6.26%) | 16 (7.31%) | 11 (5.19%) | 0.479§ |
| Inferior | 246 (57.08%) | 120 (54.79%) | 126 (59.43%) | 0.381§ |
| Posterior | 31 (7.19%) | 19 (8.68%) | 12 (5.66%) | 0.305§ |
| Culprit vessel | ||||
| LAD | 157 (36.43%) | 83 (37.90%) | 74 (34.91%) | 0.797§ |
| Cx | 78 (18.10%) | 38 (17.35%) | 40 (18.87%) | |
| RCA | 196 (45.48%) | 98 (44.75%) | 98 (46.23%) | |
| Number of affected vessels | ||||
| Two | 341 (79.12%) | 171 (78.08%) | 170 (80.19%) | 0.591§ |
| Three | 90 (20.88%) | 48 (21.92%) | 42 (19.81%) | |
| Symptom to revascularization time [h] | 2 (2–4) | 2 (2–4) | 3 (2–5) | 0.606‡ |
| Emergency to revascularization time [min] | 41 (27–87) | 39 (27–82) | 44 (28–97.5) | 0.461‡ |
| Number of stents | ||||
| One | 227 (52.67%) | 121 (55.25%) | 106 (50.00%) | 0.490§ |
| Two | 153 (35.50%) | 75 (34.25%) | 78 (36.79%) | |
| Three or more | 51 (11.83%) | 23 (10.50%) | 28 (13.21%) | |
| Total length of stents | 34 (24–54) | 33 (24–52) | 35 (25–56.5) | 0.260‡ |
| Preoperative creatinine [µmol/l] | 0.87 (0.75–1.03) | 0.87 (0.77–1.03) | 0.86 (0.74–1.04) | 0.749‡ |
| Postoperative creatinine [µmol/l] | 0.86 (0.75–0.99) | 0.86 (0.75–0.99) | 0.87 (0.77–0.99) | 0.757‡ |
| postoperative LVEF, % | 47.82 ±9.54 | 46.94 ±9.40 | 48.70 ±9.62 | 0.057† |
| Medications | ||||
| Antiaggregants | 424 (98.38%) | 213 (97.26%) | 211 (99.53%) | 0.122# |
| Anticoagulants | 3 (0.70%) | 2 (0.91%) | 1 (0.47%) | 1.000# |
| B-blockers | 313 (72.62%) | 150 (68.49%) | 163 (76.89%) | 0.051§ |
| ACE inhibitors | 135 (31.32%) | 67 (30.59%) | 68 (32.08%) | 0.740§ |
| ARBs | 16 (3.71%) | 5 (2.28%) | 11 (5.19%) | 0.180§ |
| Antilipemics | 402 (93.27%) | 198 (90.41%) | 204 (96.23%) | 0.027§ |
| Calcium channel blockers | 46 (10.67%) | 21 (9.59%) | 25 (11.79%) | 0.559§ |
| Vasodilators | 104 (24.13%) | 61 (27.85%) | 43 (20.28%) | 0.066§ |
| Diuretics | 87 (20.19%) | 49 (22.37%) | 38 (17.92%) | 0.250§ |
| Follow-up time [months] | 29.67 ±7.50 | 30.12 ±7.71 | 29.20 ±7.27 | 0.201† |
| SYNTAX score | 13 (10.5–17) | 13.5 (10.5–18) | 13 (10.5–16) | 0.440‡ |
| Major adverse cardiac eventa | 135 (31.32%) | 86 (39.27%) | 49 (23.11%) | < 0.001§ |
| Heart failure | 41 (9.51%) | 23 (10.50%) | 18 (8.49%) | 0.584§ |
| Urgent PCI | 23 (5.34%) | 15 (6.85%) | 8 (3.77%) | 0.228§ |
| Urgent CABG | 10 (2.32%) | 9 (4.11%) | 1 (0.47%) | 0.020# |
| Cardiogenic shock | 9 (2.09%) | 7 (3.20%) | 2 (0.94%) | 0.176# |
| Complete AV block | 19 (4.41%) | 14 (6.39%) | 5 (2.36%) | 0.071§ |
| Cerebrovascular disease | 5 (1.16%) | 3 (1.37%) | 2 (0.94%) | 1.000# |
| Ventricular fibrillation | 23 (5.34%) | 15 (6.85%) | 8 (3.77%) | 0.228§ |
| Ventricular tachycardia | 6 (1.39%) | 4 (1.83%) | 2 (0.94%) | 0.686# |
| Mortality | 44 (10.21%) | 28 (12.79%) | 16 (7.55%) | 0.102§ |
MACE outcomes were not observed in 296 (68.7%) of the patients, while 135 (31.3%) experienced MACE. These groups showed similar age (p = 0.55) and sex distribution (p = 0.68). The MACE group had higher frequencies of diabetes mellitus (p = 0.011), CAD (p = 0.004), renal diseases (p < 0.001), anterior STEMI (p = 0.029), and three-vessel disease (p < 0.001). Furthermore, culprit-only revascularization (p < 0.001) had been performed at a higher frequency among those with MACE, and these patients had significantly lower postoperative LVEF (p<0.001) and higher creatinine levels both preoperatively (p < 0.001) and postoperatively (p < 0.001). Antilipemic medication use was more common in the non-MACE group (p < 0.001), while diuretics were more frequently used in the MACE group (p< 0.001) (Table II).
Table II
Summary of patient characteristics with regard to major adverse cardiac events
| Parameter | Major adverse cardiac event | P-value | |
|---|---|---|---|
| No (n = 296) | Yes (n = 135) | ||
| Age [years] | 49 (46–50) | 49 (46–50) | 0.550‡ |
| Sex | |||
| Female | 34 (11.49%) | 13 (9.63%) | 0.684§ |
| Male | 262 (88.51%) | 122 (90.37%) | |
| Immigrant | 18 (6.08%) | 16 (11.85%) | 0.062§ |
| Smoking | |||
| Non-smoker | 28 (9.46%) | 11 (8.27%) | 0.451§ |
| Passive smoker | 19 (6.42%) | 4 (3.01%) | |
| Ex-smoker | 45 (15.20%) | 24 (18.05%) | |
| Active smoker | 204 (68.92%) | 94 (70.68%) | |
| Previous comorbidities | |||
| Hypertension | 101 (34.12%) | 51 (37.78%) | 0.461§ |
| Diabetes mellitus | 92 (31.08%) | 59 (43.70%) | 0.011§ |
| Hyperlipidemia | 169 (57.09%) | 51 (37.78%) | < 0.001§ |
| COPD | 8 (2.70%) | 9 (6.67%) | 0.090§ |
| Coronary artery disease | 43 (14.53%) | 35 (25.93%) | 0.004§ |
| CABG | 4 (1.35%) | 5 (3.70%) | 0.146# |
| Peripheral artery disease | 9 (3.04%) | 9 (6.67%) | 0.137§ |
| Cerebrovascular disease | 5 (1.69%) | 7 (5.19%) | 0.056# |
| Pulmonary embolism | 1 (0.34%) | 1 (0.74%) | 0.529# |
| Deep vein thrombosis | 2 (0.68%) | 2 (1.48%) | 0.593# |
| Renal diseases | 1 (0.34%) | 11 (8.15%) | < 0.001# |
| Malignancy | 4 (1.35%) | 3 (2.22%) | 0.683# |
| STEMI territorya | |||
| Anterior | 89 (30.07%) | 55 (40.74%) | 0.029§ |
| Lateral | 23 (7.77%) | 4 (2.96%) | 0.090§ |
| Inferior | 175 (59.12%) | 71 (52.59%) | 0.204§ |
| Posterior | 22 (7.43%) | 9 (6.67%) | 0.933§ |
| Culprit vessel | |||
| LAD | 101 (34.12%) | 56 (41.48%) | 0.096§ |
| Cx | 61 (20.61%) | 17 (12.59%) | |
| RCA | 134 (45.27%) | 62 (45.93%) | |
| Number of affected vessels | |||
| Two | 249 (84.12%) | 92 (68.15%) | < 0.001§ |
| Three | 47 (15.88%) | 43 (31.85%) | |
| Symptom to revascularization time [h] | 3 (2–4) | 2 (2–5) | 0.493‡ |
| Emergency to revascularization time [min] | 44.5 (27.5–96.5) | 37 (27–73) | 0.079‡ |
| Type of revascularization | |||
| Culprit-only | 133 (44.93%) | 86 (63.70%) | < 0.001§ |
| Complete | 163 (55.07%) | 49 (36.30%) | |
| Number of stents | |||
| One | 158 (53.38%) | 69 (51.11%) | 0.030§ |
| Two | 111 (37.50%) | 42 (31.11%) | |
| Three or more | 27 (9.12%) | 24 (17.78%)* | |
| Total length of stents | 33 (24–53.5) | 35 (24–55) | 0.418‡ |
| Creatinine, preoperative [µmol/l] | 0.85 (0.74–0.98) | 0.92 (0.79–1.15) | < 0.001‡ |
| Creatinine, postoperative [µmol/l] | 0.84 (0.75–0.95) | 0.90 (0.79–1.16) | < 0.001‡ |
| LVEF, postoperative, % | 51.08 ±7.25 | 40.60 ±10.03 | < 0.001† |
| Medications | |||
| Antiaggregants | 296 (100.00%) | 128 (94.81%) | < 0.001# |
| Anticoagulants | 1 (0.34%) | 2 (1.48%) | 0.232# |
| B-blockers | 215 (72.64%) | 98 (72.59%) | 1.000§ |
| ACE inhibitors | 84 (28.38%) | 51 (37.78%) | 0.051§ |
| ARBs | 12 (4.05%) | 4 (2.96%) | 0.779§ |
| Antilipemics | 286 (96.62%) | 116 (85.93%) | < 0.001§ |
| Calcium channel blockers | 32 (10.81%) | 14 (10.37%) | 1.000§ |
| Vasodilators | 78 (26.35%) | 26 (19.26%) | 0.110§ |
| Diuretics | 25 (8.45%) | 62 (45.93%) | < 0.001§ |
| Follow-up time [months] | 29.43 ±7.50 | 30.19 ±7.50 | 0.335† |
Multivariable logistic regression revealed that culprit-only revascularization (OR = 2.645, 95% CI: 1.533–4.563, p < 0.001), three or more stents (OR = 2.691, 95% CI: 1.265–5.726, p = 0.010), high preoperative creatinine (OR = 7.873, 95% CI: 2.384–25.992, p < 0.001), low postoperative LVEF (OR = 0.898, 95% CI: 0.867–0.930, p < 0.001) and diuretic use (OR = 3.915, 95% CI: 1.933–7.931, p < 0.001) were independently associated with increased MACE risk. On the other hand, previous hyperlipidemia (OR = 0.345, 95% CI: 0.200–0.597, p< 0.001) was independently associated with decreased MACE risk. Other variables included in the analysis – diabetes mellitus (p = 0.78), CAD (p = 0.14), renal disease (p = 0.51), anterior STEMI (p = 0.07), number of affected vessels (p = 0.09), postoperative creatinine (p = 0.93), antiaggregant use (p = 0.21), and antilipemic use (p = 0.10) – were found to be non-significant (Table III).
Table III
Significant factors independently associated with major adverse cardiac events, multivariable logistic regression analysis
Next, we compared mortality-related factors. The study population comprised 387 survivors (89.8% of the cohort) and 44 non-survivors (10.2% of the cohort). The groups showed no significant differences in age (p = 0.948) or sex distribution (p = 1.000). However, non-survivors had significantly higher rates of diabetes mellitus (p < 0.001), renal diseases (p < 0.001), and three-vessel involvement (p = 0.013), along with lower antilipemic use (p < 0.001) and b-blocker adherence (p = 0.008). Notably, non-survivors exhibited worse LVEF (p < 0.001), higher postoperative creatinine levels (p < 0.001), and longer symptom-to-revascularization times (p = 0.009) (Table IV).
Table IV
Characterization of patients with respect to mortality occurrence
| Parameter | Mortality | P-value | |
|---|---|---|---|
| No (n = 387) | Yes (n = 44) | ||
| Age [years] | 49 (46–50) | 48.5 (45.5–50) | 0.948‡ |
| Sex | |||
| Female | 43 (11.11%) | 4 (9.09%) | 1.000# |
| Male | 344 (88.89%) | 40 (90.91%) | |
| Immigrant | 29 (7.49%) | 5 (11.36%) | 0.373# |
| Smoking | |||
| Non-smoker | 36 (9.30%) | 3 (7.14%) | 0.832¶ |
| Passive smoker | 22 (5.68%) | 1 (2.38%) | |
| Ex-smoker | 61 (15.76%) | 8 (19.05%) | |
| Active smoker | 268 (69.25%) | 30 (71.43%) | |
| Previous comorbidities | |||
| Hypertension | 136 (35.14%) | 16 (36.36%) | 1.000§ |
| Diabetes mellitus | 124 (32.04%) | 27 (61.36%) | < 0.001§ |
| Hyperlipidemia | 208 (53.75%) | 12 (27.27%) | 0.002§ |
| COPD | 13 (3.36%) | 4 (9.09%) | 0.084# |
| Coronary artery disease | 66 (17.05%) | 12 (27.27%) | 0.144§ |
| CABG | 7 (1.81%) | 2 (4.55%) | 0.232# |
| Peripheral artery disease | 16 (4.13%) | 2 (4.55%) | 0.704# |
| Cerebrovascular disease | 10 (2.58%) | 2 (4.55%) | 0.351# |
| Pulmonary embolism | 2 (0.52%) | 0 (0.00%) | 1.000# |
| Deep vein thrombosis | 3 (0.78%) | 1 (2.27%) | 0.351# |
| Renal diseases | 5 (1.29%) | 7 (15.91%) | < 0.001# |
| Malignancy | 6 (1.55%) | 1 (2.27%) | 0.532# |
| STEMI territorya | |||
| Anterior | 123 (31.78%) | 21 (47.73%) | 0.051§ |
| Lateral | 25 (6.46%) | 2 (4.55%) | 1.000# |
| Inferior | 228 (58.91%) | 18 (40.91%) | 0.034§ |
| Posterior | 26 (6.72%) | 5 (11.36%) | 0.229# |
| Culprit vessel | |||
| LAD | 135 (34.88%) | 22 (50.00%) | 0.122§ |
| Cx | 73 (18.86%) | 5 (11.36%) | |
| RCA | 179 (46.25%) | 17 (38.64%) | |
| Number of affected vessels | |||
| Two | 313 (80.88%) | 28 (63.64%) | 0.013§ |
| Three | 74 (19.12%) | 16 (36.36%) | |
| Symptom to revascularization time [h] | 2 (2–4) | 4 (2–8) | 0.009‡ |
| Emergency to revascularization time [min] | 42 (27–87) | 36.5 (27.5–82) | 0.317‡ |
| Type of revascularization | |||
| Culprit-only | 191 (49.35%) | 28 (63.64%) | 0.102§ |
| Complete | 196 (50.65%) | 16 (36.36%) | |
| Number of stents | |||
| One | 204 (52.71%) | 23 (52.27%) | 0.922§ |
| Two | 138 (35.66%) | 15 (34.09%) | |
| Three or more | 45 (11.63%) | 6 (13.64%) | |
| Total length of stents | 35 (24–54) | 31.5 (21.5–49) | 0.332‡ |
| Preoperative creatinine [µmol/l] | 0.86 (0.75–1.00) | 0.98 (0.79–1.17) | 0.002‡ |
| Postoperative creatinine [µmol/l] | 0.85 (0.75–0.97) | 1.05 (0.81–1.34) | < 0.001‡ |
| Postoperative LVEF, % | 49.14 ±8.70 | 35.67 ±8.26 | < 0.001† |
| Medications | |||
| Antiaggregants | 387 (100.00%) | 37 (84.09%) | < 0.001# |
| Anticoagulants | 3 (0.78%) | 0 (0.00%) | 1.000# |
| b-blockers | 289 (74.68%) | 24 (54.55%) | 0.008§ |
| ACE inhibitors | 124 (32.04%) | 11 (25.00%) | 0.434§ |
| ARBs | 15 (3.88%) | 1 (2.27%) | 1.000# |
| Antilipemics | 374 (96.64%) | 28 (63.64%) | < 0.001# |
| Calcium channel blockers | 42 (10.85%) | 4 (9.09%) | 1.000# |
| Vasodilators | 98 (25.32%) | 6 (13.64%) | 0.126§ |
| Diuretics | 67 (17.31%) | 20 (45.45%) | < 0.001§ |
| Follow-up time [months] | 29.29 ±7.53 | 33.02 ±6.42 | < 0.001† |
Analysis of the multivariable model showed that high postoperative creatinine (OR = 4.501, 95% CI: 1.729–11.718, p = 0.002) and low postoperative LVEF (OR = 0.862, 95% CI: 0.820–0.906, p < 0.001) were independently associated with increased mortality risk. Conversely, history of hyperlipidemia (OR = 0.338, 95% CI: 0.134–0.854, p = 0.022), b-blocker use (OR = 0.326, 95% CI: 0.120–0.888, p = 0.028) and antilipemic use (OR = 0.118, 95% CI: 0.034–0.405, p < 0.001) were independently associated with lower likelihood of mortality. Other variables included in the analysis – history of diabetes mellitus (p = 0.23), renal disease (p = 0.81), inferior STEMI (p = 0.79), number of affected vessels (p = 0.60), symptom-to-revascularization time (p = 0.07), preoperative creatinine (p = 0.34), antiaggregant use (p = 0.53), and diuretic use (p = 0.48) – were found to be non-significant (Table V).
Table V
Significant factors independently associated with mortality, multivariable logistic regression analysis
Discussion
The principal finding of this study is that complete revascularization offers improved outcomes among young STEMI patients with multivessel disease, significantly reducing MACE compared to recipients of culprit-only PCI. Critically, postoperative renal dysfunction and impaired LVEF were independent predictors of adverse outcomes. These findings extend the established benefits of complete revascularization to a younger population, which addresses knowledge gaps, since this subgroup of the STEMI population has been underrepresented in randomized trials.
We found a 61% reduction in MACE risk with complete revascularization. The COMPLETE trial, which enrolled older patients (mean age 62 years), showed a 26% reduction in cardiovascular death or myocardial infarction but no significant mortality difference [9]. Similarly, in our younger cohort, unadjusted mortality rates did not differ significantly between revascularization strategies (p = 0.10), though multivariable analysis identified renal dysfunction and impaired LVEF as independent predictors of mortality. This observation gains further significance when considering that young STEMI patients typically present with different risk profiles. These demographic variations may influence both therapeutic response and underlying plaque biology, with intravascular ultrasound studies suggesting that younger patients exhibit greater inflammatory activity in the context of cardiac involvement [14, 15]. The 3.8-fold reduction in urgent CABG requirements in our complete revascularization group indicates that untreated non-culprit lesions can lead to recurrent ischemic events, consistent with prior research [16, 17].
The identification of postoperative renal dysfunction as a powerful predictor is a crucial output. Patients with elevated postoperative creatinine exhibited increased MACE and mortality risk, potentially reflecting contrast-induced nephropathy, hemodynamic instability, cardiorenal interactions, or underlying endothelial dysfunction [18]. Although contrast volume data were unavailable, the association between renal dysfunction and adverse outcomes remained significant after multivariable adjustment. This agrees with pathophysiological evidence showing that acute kidney injury triggers elevated systemic inflammation and oxidative stress, which could worsen myocardial injury, endothelial damage, and recovery through direct or indirect impacts [19, 20]. Similarly, reduction in post-revascularization LVEF was an independent predictor of both MACE and mortality, demonstrating the anticipated limitations in reversing damage despite urgent revascularization.
It is crucial to examine the seemingly paradoxical association of pre-existing hyperlipidemia with improved prognosis. This relationship is probably associated with adherence to (and the subsequent benefits of) statin treatment. Supporting this interpretation, statin adherence was significantly higher in the complete revascularization group (87% vs. 71%) and independently predicted reduced mortality in multivariable models. The protective association of b-blocker therapy further substantiates this interpretation and demonstrates that younger patients benefit greatly from adherence to medications prescribed for comorbidities.
Early guidelines addressing therapeutic approaches to patients with multivessel disease cautioned against non-culprit intervention without demonstration of ischemia [21, 22]. Such recommendations reflected concerns about procedural complexity and plaque destabilization. However, recent randomized controlled trials have uprooted this approach. The PRAMI trial first demonstrated that preventive PCI reduced the composite endpoint (cardiac death, nonfatal MI, refractory angina) from 23% to 9% [23], while the CvLPRIT trial showed 55% MACE reduction with complete revascularization [24]. Subsequent DANAMI-3-PRIMULTI [7] and COMPARE-ACUTE [8] trials reinforced these benefits. Finally, the COMPLETE trial [9] provided definitive evidence that complete revascularization reduced cardiovascular death or myocardial infarction by 26% and ischemia-driven revascularization by 49%, irrespective of the approach to complete revascularization (simultaneous intervention during the index procedure or staged intervention). Our data corroborate these findings while extending them to younger patients, which is crucial given that available trials largely included older cohorts [6–9]. An important consideration is the potential for reverse causation. Patients with more severe baseline illness – including those with worse renal function, lower LVEF, or more complex anatomy – may have been preferentially managed with culprit-only PCI due to hemodynamic concerns or unfavorable lesion characteristics. Although multivariable analysis adjusted for measurable confounders, including SYNTAX score, we cannot rule out that unmeasured factors influenced both treatment selection and outcomes. Future prospective studies with pre-specified treatment algorithms are needed to definitively establish causality.
The optimal timing of non-culprit revascularization warrants careful consideration. Our institutional protocol favoring staged revascularization (median 3 days after primary PCI) aligns with prior recommendations and leads to achievement of early complete revascularization while shortening the initial procedure and preventing concerns about contrast overload during the acute event. It is however notable that Di Mario et al. [25] reported comparable outcomes between immediate and staged strategies, while a randomized trial of 214 patients found both staged and simultaneous multivessel PCI outperformed culprit-only intervention [26]. Other studies also indicate comparable safety profiles between strategies regarding contrast nephropathy, stroke, and bleeding [9, 27].
Complete revascularization can reduce future risks by stabilizing vulnerable non-culprit plaques, which frequently are often thin-cap fibroatheromas despite being defined as angiographically mild [14, 17]. These plaques exhibit increased susceptibility to rupture as a result of the pro-inflammatory burden following the initial STEMI event [28]. Furthermore, restoring perfusion to territories beyond the infarct zone may improve ventricular remodeling and reduce arrhythmic risk. The identification of anterior STEMI and three-vessel disease as predictors of adverse outcomes emphasizes the need for comprehensive anatomical assessment in young patients where extensive disease may indicate aggressive atherosclerosis.
Despite including a respectably large cohort and addressing the knowledge gap concerning relatively young patients with STEMI, the single-center design limits the generalizability of our findings to other settings. The retrospective analysis carries inherent risk of selection bias and confounding due to factors that were not included in patient records. Propensity score matching was not performed due to incomplete documentation of factors influencing the revascularization strategy. Although multivariable regression adjusted for measurable confounders, residual confounding by indication cannot be excluded, as operators may have preferentially selected culprit-only approaches for higher-risk patients with unfavorable anatomy. Detailed angiographic characterization of non-culprit lesions (plaque morphology, lesion size, and quantitative stenosis) was unavailable, and these are factors that could have influenced treatment choice. Contrast volume data were not systematically recorded, precluding direct assessment of the contribution of contrast-induced nephropathy to postoperative renal dysfunction. The absence of functional assessment – fractional flow reserve (FFR) and instantaneous wave-free ratio (iFR) – is another notable limitation, particularly since FFR data in the DANAMI-3-PRIMULTI [7] and COMPARE-ACUTE [8] trials were successful in demonstrating improvements in certain outcomes. One particular concern may be the fact that we examined all cause mortality instead of cardiovascular specific mortality; however, this is unlikely to alter mortality-related outcomes, as multivariable analyses adjusted for potential confounders. Importantly, anatomical complexity as assessed by SYNTAX score did not differ between groups, reducing the likelihood that treatment allocation was driven by lesion severity. Although STEMI is more common among males, the dominance of males in our cohort could indicate that these findings may not be directly generalizable to females.
Despite these limitations, our findings carry immediate clinical implications. First, complete revascularization should be strongly considered for young STEMI patients with multivessel disease, ideally during index hospitalization. Second, renal function and LVEF assessment provide critical prognostic information that should guide post-procedural management. Third, antilipemic treatment appears to be a strong prognostic indicator benefiting patients who adhere to treatments. Future prospective studies should establish optimal patient selection criteria using advanced imaging (IVUS/OCT) to identify vulnerable plaques [13], evaluate long-term outcomes, and assess cost-effectiveness in healthcare systems. The emerging role of coronary flow reserve (CFR) assessment, as demonstrated by Kozomara et al. [29], should also be examined in young populations.
Conclusions
This study demonstrated that complete revascularization significantly reduces MACE and urgent CABG requirements in young STEMI patients with multivessel disease. We also identified renal dysfunction and impaired LVEF as independent predictors, providing evidence that expands previously established relationships into younger STEMI cohorts. The protective impacts of antilipemic and antihypertensive use support the benefits of comorbidity treatments in CAD. Therefore, we believe our findings support the expansion of European Society of Cardiology (Class IIa) recommendations for routine non-culprit revascularization [30] to younger cohorts. As cardiovascular disease increasingly affects younger populations, these findings demonstrate the needs for providing optimized care in this group.