Introduction
Coronary artery disease remains a major global health burden, with coronary artery bypass grafting (CABG) offering proven long-term benefits for patients with extensive disease [1]. Aspirin reduces vascular complications and mortality, with guidelines recommending 75–100 mg daily for chronic coronary syndrome [2, 3]. Low-dose aspirin is also advised perioperatively to reduce thrombotic risk and support graft patency [1]. While long-term efficacy of low-dose aspirin is well established, evidence regarding the optimal dose in the early postoperative period remains limited and conflicting [1, 4, 5].
The first definitive multicenter study supporting early postoperative aspirin administration, which challenged prior concerns regarding bleeding risk and platelet dysfunction against the obtained survival benefits, utilized a wide range of early postoperative aspirin doses [6]. In fact, the total aspirin dose administered within 48 h after revascularization in this study ranged from 80 to 650 mg. Subsequent studies also employed a variety of postoperative dosing regimens, highlighting the absence of a single standardized approach and reflecting the diversity of accepted clinical practices [1, 7].
This observational, exploratory, and preliminary study was driven by the hypothesis that aspirin-induced platelet inhibition in the early postoperative period after CABG may vary between individuals. To investigate this, we analyzed arachidonic acid (AA)-induced platelet aggregation in patients receiving postoperative aspirin monotherapy.
Aim
This preliminary report of observational, exploratory findings aimed to investigate the interpatient variability in AA-induced platelet aggregation during the early postoperative period following CABG in patients receiving monotherapy with aspirin at various doses based on the physician’s decision.
Material and methods
Blood samples from 40 patients who underwent on-pump CABG for chronic coronary syndrome between January 2020 and December 2023 at the Department of Cardiovascular Surgery and Transplantology, St. John Paul II Hospital in Krakow, Poland, were analyzed in this preliminary, prospective, observational study. Fasting venous blood samples were collected from the antecubital vein from all 40 patients on 5–7 days postoperatively. Blood samples at the 3-month follow-up were available for only 10 patients. For this subset, samples from both the early postoperative period (5–7 days) and the 3-month follow-up were available and analyzed.
Platelet aggregation was assessed using the standardized laboratory method with optical aggregometry with a two-agonist panel consisting of 1 mM arachidonic acid (AA) and 2 µM adenosine diphosphate (ADP), at the Molecular Biology Laboratory, St. John Paul II Hospital, Kraków, Poland. AA-induced platelet aggregation was employed as a surrogate marker for assessing adequate aspirin-induced inhibition. A predefined threshold of 20% aggregation was applied, in accordance with standardized laboratory protocols routinely used in clinical practice by the Molecular Biology Laboratory, St. John Paul II Hospital, Kraków, Poland. This 20% cutoff has also been utilized in previous research studies, reinforcing its relevance and consistency across investigative settings [8–10].
All surgeries were performed based on Heart Team qualification and utilized cardiopulmonary bypass (CPB). All patients were receiving 75 mg of aspirin preoperatively, according to guideline-directed medical therapy, and aspirin was not discontinued before the operation. Patients who received preoperative P2Y12 inhibitors or GP IIb/IIIa antiplatelet therapy, and those who required perioperative platelet transfusions, were excluded from the analysis. Postoperatively, aspirin was resumed within the first 24 h at a dose of either 75 mg in 33 (82.5%) patients or 150 mg in 7 (17.5%) patients, as determined by the physician’s decision. The analytic exploratory observation was performed based on prospective blood sample collection, and the aspirin dose was not an intervention related to the study. Patients were primarily male (85%), with a median age of 60.5 (53.5–67) years (Table I).
Table I
Baseline patient characteristics
Statistical analysis
Categorical variables were summarized as counts and percentages, and continuous data as mean ± SD or median with IQR, depending on distribution. Fisher’s exact test was used for categorical comparisons, and the Shapiro-Wilk test assessed normality. Group comparisons employed Student’s t-test or Mann-Whitney U test, as appropriate. Variability in AA-induced platelet aggregation across aspirin doses was analyzed using robust Levene’s test. All analyses were performed using Stata statistical software: Release 18.5 (StataCorp LLC, College Station, TX, USA), with significance set at p < 0.05.
Ethical statement
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki and received approval from the Bioethics Committee of Jagiellonian University Medical College (1072.6120.280.2019). All participants provided informed consent, and ethical guidelines were strictly followed to ensure the integrity and safety of the research process.
Results
Based on blood samples analyzed from all 40 patients, on postoperative days 5–7, AA-induced platelet aggregation differed significantly between the 75 mg and 150 mg aspirin dose groups (37% [2–72] vs. 3% [0–1]; p = 0.02, respectively) (Figure 1 A). In contrast, ADP-induced aggregation remained similar between the two aspirin doses (57.5% [47.5–64] vs. 60% [48–66]; p = 0.65, respectively), which was within normal and expected levels. A significant difference in the variance of AA-induced aggregation was observed between the aspirin dose groups (p < 0.001, Figure 1 B). The 150 mg dose group exhibited lower variability, although a single “non-responder” case was identified. Notably, on postoperative days 5–7, more than two-thirds of the patients (69%) receiving 75 mg of aspirin did not achieve an aggregation level below 20%, which was predefined as the threshold for sufficient inhibition.
Figure 1
A – Platelet aggregation on postoperative days 5-7 with AA-induced and ADP-induced aggregation by the ASA dose group. Medians, Q1-Q3 are presented, Mann-Whitney U test. B – AA-induced platelet aggregation on postoperative days 5-7 by the ASA dose group. Medians, Q1-Q3 are presented, robust Levene’s test. C – Subgroup analysis of AA-induced platelet aggregation in 10 patients receiving 75 mg aspirin: individual data on postoperative days 5-7 and at 3-month follow-up
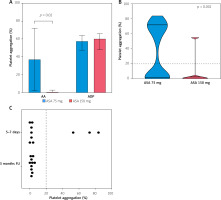
Among the available sample data, a subset of 10 patients had paired measurements on both 5–7 days postoperatively and at the 3-month follow-up, while receiving a consistent daily dose of 75 mg of aspirin. In this non-random subset, defined solely by sample availability, all patients demonstrated AA-induced platelet aggregation levels below the 20% threshold at 3 months. This finding contrasts with their early postoperative results, where 30% of the same group were classified as non-responders despite receiving the identical aspirin regimen (Figure 1 C).
Discussion
Although reduced aspirin responsiveness in the early postoperative phase following CABG has been previously reported in small initial studies, preoperative discontinuation of aspirin in their protocols was an explicit limitation which also no longer reflects current guideline-directed practice [11, 12]. Unlike earlier reports, this study did not involve preoperative discontinuation of aspirin, allowing for an observation which relates to current clinical practice and is theoretically supported by the continued perioperative antiplatelet management. Moreover, what distinguishes this exploratory analysis from previous studies is its focus on interpatient variability rather than group-level effects, offering a more individualized perspective on aspirin responsiveness.
The observed variability – particularly among patients receiving 75 mg of aspirin daily – highlights the potential influence of individual pharmacodynamic differences in the early post-CABG period. These findings raise important questions about the adequacy of low-dose aspirin in certain patients during this critical phase. This observed diminished efficacy of low-dose aspirin in the early postoperative period, with 69% of patients not achieving adequate platelet inhibition, may be attributed to multiple factors, including the inflammatory response related to the CABG surgery, the effect of CPB, and increased postoperative platelet turnover, all of which were reported in other clinical contexts previously [13–15].
This study is notably limited by its small sample size, which constrains the generalizability of the findings and positions the results as hypothesis-generating only. Moreover, this exploratory observational study did not assess clinical outcomes and, therefore, does not propose therapeutic recommendations, as the results are observational and based solely on laboratory data. While previous small-scale studies have reported diminished aspirin responsiveness in the early postoperative phase, this analysis differed from previous reports because it maintained uninterrupted perioperative aspirin therapy and focused on interpatient variability rather than aggregated group effects. Notably, greater variability in platelet inhibition was observed among patients receiving 75 mg of aspirin daily compared to those on 150 mg. Whether higher aspirin doses during the first 3 months post-CABG can improve graft patency without increasing the bleeding risk remains to be determined and will be addressed in the upcoming multicenter OPTIMUS-CABG trial (NCT07195149).
Conclusions
This exploratory, preliminary study raises the hypothesis of significant interindividual variability in platelet inhibition during the early postoperative period following CABG, particularly with low-dose aspirin (75 mg). A bimodal response pattern appeared to drive this variability, with patients showing either sufficient inhibition or minimal responsiveness – less evident at the 150 mg dose. In the subset of 10 patients with repeated measurements, AA-induced aggregation patterns shifted over time – while 30% were classified as non-responders early postoperatively, all achieved adequate inhibition by the 3-month follow-up.







