Introduction
Diverticular disease is one of the most common conditions, in which patients with age less than 40 years comprise around 10%, and more than 70% in people in the eighth decade of life, with prevalence being similar in both men and women [1]. Over 20% of patients who had diverticulitis, one of the complications of diverticular disease, is expected to have at least one recurrence of the disease [2]. Annually in the United States, nearly 1 million outpatient visits and more than 200,000 inpatient admission are due to diverticulitis, at the cost of more than 9 billion USD annually [3]. Far more concerning is the rate of young-age diverticulitis, in which the incidence of diverticulitis rose by more than 132% in individuals from 40 to 49 years old [4]. Increasingly, diverticular disease has been linked to cardiovascular events [5] and stroke [6]. Cardiovascular events are expected to be responsible for 32% of the global deaths [7], and with over 23 million patients impacted with heart failure [8], understanding a critical cardiovascular condition such as acute myocardial infarction (AMI) remains crucial to reduce mortality for the general public.
Myocardial infarction is one of the most common cardiovascular diseases, with serious consequences in terms of mortality, morbidity, and cost to society [9]. Current incidence projection of this high-mortality disease is expected to double in 25 years, due to various multifactorial factors [10]. Beyond the rising trends of obesity, metabolic syndrome, and smoking, exploring other risk factors can provide additional clues to combating this deadly disease. Both AMI and diverticular disease have common risk factors, such as lifestyle factors, smoking [11], or even obesity [12]. Current literature has linked diverticular disease to AMI [13–17], but information on the association between diverticular disease and AMI is scattered.
Thus, we conducted a systematic review and meta-analysis of studies examining the impact of diverticular disease on the risk of AMI events.
Methods
Search strategy
Two investigators (TSr and NS) independently conducted searches in the PubMed and Embase databases from inception through August 2024 using search terms as specified in Supplementary Data S1. No language restrictions were applied. The same investigators independently assessed the eligibility of the retrieved records, with further discussions involving one investigator (TSu) to resolve any conflicts. Abstracts and unpublished studies were excluded.
Eligibility criteria
The eligibility criteria were as follows: Included studies must be cohort studies published as original research, assessing the association between diverticular disease and risks of AMI. As diverticular disease ranges from asymptomatic diverticulosis to complications of colonic diverticulosis, the International Classification of Disease codes 10th revision provided an accurate general diverticular disease diagnosis [18]. The term diverticulosis is used if there is a presence of diverticular without inflammation, diverticulitis is used if there is inflammation of the diverticular, and diverticular disease is used as a broader term encompassing both, used only when referring to diagnoses in the included studies that did not differentiate between both. Studies must include a group of individuals with diverticular disease and another group without diverticular disease, and must provide effect estimates representing the association between diverticular disease and the risk of AMI in the form of relative risk (RR) or hazard ratios (HR), accompanied by 95% confidence intervals (CIs) or survival curves. Raw data adequate for calculation of the effect sizes is acceptable.
Data extraction
We employed a standardised data collection protocol to extract the following information: last name of the first author, study country, study design, publication year, total number of participants, study participants’ recruitment protocol, diverticular disease diagnosis/definition, myocardial infarction diagnosis, follow-up duration, age, gender, and variables adjusted for in multivariate analysis.
Two investigators (PD and NS) applied the Newcastle-Ottawa Scale for cohort studies to evaluate research quality, focusing on the quality of participant recruitment, comparability between groups, and accuracy of outcome ascertainment [19].
Statistical analysis
Data analysis was conducted using Review Manager 5.4 software from the Cochrane Collaboration. Point estimates with standard errors from each study were combined using DerSimonian and Laird’s generic inverse variance method [20]. Due to heterogeneous background populations and protocols among the studies, a random-effects model was employed. Statistical heterogeneity was assessed using Cochran’s Q test, supplemented by I2 statistics to quantify the proportion of total variation across studies attributable to heterogeneity rather than chance. I2 values categorise heterogeneity as insignificant (0–25%), low (26–50%), moderate (51–75%), or high (> 75%) [21]. A funnel plot will be utilized to examine potential publication bias if sufficient studies are available. The Egger regression test will be conducted using Stata BE 18 (StataCorp LP, College Station, TX, USA) for the regression analysis.
Results
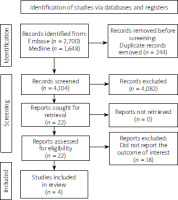
Our search strategy identified 4348 studies (1648 from Embase and 2700 from PubMed). After removing 244 duplicates, we reviewed those studies by title and abstract, excluding 4082 studies that did not meet the eligibility criteria related to study design, participants, or article type. Subsequently, we thoroughly reviewed 22 articles and excluded 18 for not reporting the relevant outcome. Ultimately, 4 studies met the eligibility criteria for our meta-analysis. Figure 1 illustrates our search methodology and selection process, and Table I details the characteristics and quality assessment of the included studies.
Table I
Baseline characteristics of all eligible studies included in the meta-analysis
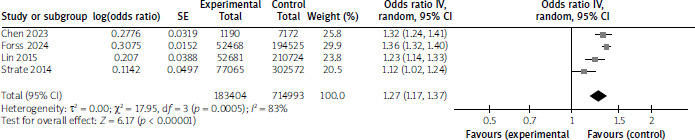
A total of 4 cohort studies with 898,397 participants investigated the association between diverticular disease and AMI. The average participant age was 57 years, and 43% were male, as shown in Table I (Average participant age was utilised from 3 studies, except Strate et al. (2014) due to no mean age reported). The pooled analysis revealed that individuals with diverticular disease had an increased risk of myocardial infarction, with a pooled RR of 1.27 (95% CI: 1.17, 1.37; p < 0.001, Figure 2), when compared to individuals without diverticular disease. High statistical heterogeneity was observed (I2 = 83%). Of the 4 included studies, only one (Lin et al.) provided any stratification by diverticular disease subtype (diverticulosis vs. diverticulitis). The remaining studies combined the whole spectrum of diverticulosis into a pooled cohort. None of the studies provided sufficient data to perform subgroup analyses based on symptom status (symptomatic vs. asymptomatic). Furthermore, reporting of established cardiovascular risk factors (smoking, obesity, etc.) was inconsistent and incomplete across studies, precluding adjusted pooled analyses.
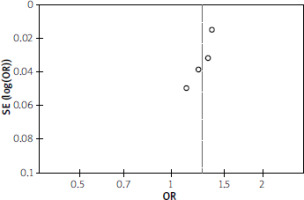
A funnel plot evaluating publication bias for all studies shows no obvious asymmetry, suggesting no publication bias, as shown in Figure 3. Additionally, the Egger regression test (p = 0.800) confirms the absence of publication bias.
Discussion
This study is the first systematic review and meta-analysis to consolidate data on how diverticular disease affects the risk of incident AMI. The pooled analysis indicated that diverticular disease patients have 27% higher risk of an AMI event compared to individuals without diverticular disease.
Several explanations exist for our findings, despite the limited literature of the pathophysiological process behind diverticular disease and acute myocardial infarction. First, a gut-heart connection has been proposed via oxidative stress and ongoing inflammation process. Diverticular disease, in some of its spectrum, has been known to express tumour necrosis factor a (TNF-a) [22], as well as higher levels of C-reactive protein (CRP) and interleukin-6 (IL-6), pre-diagnostically [23]. TNF-a and IL-6 are directly linked to coronary artery disease and AMI events [24, 25], which may explain our results. In addition, persistent low-grade inflammation can occur up to 24 months after post-acute events of diverticular disease [26], and may generate inflammatory cytokines contributing to acute myocardial infarction subsequently. Interestingly, one of our selected studies performed a subgroup analysis [16], in which both diverticulosis and diverticulitis had statistically significant higher risk of AMI events, suggesting that any spectrum of diverticular disease had similar risk of AMI events. Finally, one study [14] indicated that diverticular disease had a higher risk of venous thromboembolism, and the concurrent of venous thromboembolism and diverticular disease could lead to higher risk of cancer [27] in this subpopulation group.
Increasingly, some studies have indicated changes of microbiota in patients with diverticular disease, compared to those without [28–30]. The depletion of Clostridium cluster IV, Clostridium cluster IX, and Lactobacillaceae [28], in which there are anti-inflammatory producing species [31, 32], was reported when comparing between diverticular and non-diverticular disease groups. Clostridium cluster IX is known to produce propionate, which is associated with health-promoting effects including anti-lipogenic, cholesterol-lowering, anti-inflammatory, and anti-carcinogenic action [28]. Moreover, some pathogenic species, such as Fusobacteria, Prevotella, and Paraprevotella, are more abundant in the diverticular disease group [30]. Prevotella has shown to be related to colitis [33], whereas Fusobacterium is correlated with the pathogenesis of colorectal cancer through activation of host inflammatory responses [34]. With decreasing amounts of anti-inflammatory microbiota and increasing amounts of pathogenic microbiota, gut microbiome can be linked to the potential causes in our study [35].
Our study has several major advantageous. As the first study to investigate the current evidence of the relationship between diverticular disease and acute myocardial infarction events, the large population size facilitates precision and improves estimates of the effect size or association [36]. This review strictly adhered to recommendations from the PRISMA guidelines. Efforts were made to search grey literature with no language restrictions, reducing publication bias. However, the current meta-analysis has several limitations. Firstly, the included meta-analysis showed high heterogeneity, possibly influenced by differences in baseline characteristics such as prevalence of diverticular disease, characteristics of diverticular disease as defined by each author, and follow-up duration. Secondly, because all studies were conducted mainly on developed country populations, the findings may not be generalisable to other populations. Thirdly, as the original papers did not present detailed data of the patients, or fine differentiation between diverticular spectrum, subgroup analyses including smoking, obesity, diabetes, and others could not be done. Lastly, although the risk of AMI is increased by almost 30%, the role of acetylsalicylic acid in this patient group is currently unknown. Large studies aiming at benefit and risk, such as incidence of gastrointestinal bleeding, must be evaluated before a recommendation can be rendered.
Conclusions
Our meta-analysis found that diverticular disease was associated with a higher risk of AMI. Because diverticular disease is highly prevalent in the general population, understanding its mechanism and pathophysiology is crucial to reduce the incidence of AMI as well as improve long-term survival outcomes.