Our study analyzed the treatment outcomes of patients with acute mesenteric ischemia who received endovascular treatment as first-line therapy. We found that the use of various endovascular techniques and equipment (ante- and retrograde access, reperfusion catheters, balloon-mounted and self-expanding stents) appropriately selected for the type and location of the lesion allows for effective treatment of patients with acute visceral artery occlusion.
Introduction
The treatment of acute mesenteric ischemia (AMI) remains a significant diagnostic and therapeutic challenge. This life-threatening vascular emergency, often caused by ischemia of the superior mesenteric artery (SMA), can lead to intestinal necrosis, multi-organ failure, and high mortality [1]. AMI primarily affects elderly patients, frequently as a consequence of progressive stenosis from atherosclerotic changes in the visceral arteries [2]. Many patients experience prodromal symptoms of chronic mesenteric ischemia, such as postprandial pain, weight loss, and anorexia. Atherosclerotic plaques are typically located in the proximal segments of the visceral arteries and are often heavily calcified. A critical reduction in flow – triggered by events such as circulatory decompensation, dehydration, or hemorrhage – can precipitate thrombosis of a pre-stenotic artery, leading to acute intestinal ischemia. Although traditional AMI management involves urgent open surgical repair, recent studies have proposed endovascular or hybrid techniques to reduce the procedure’s high associated morbidity and mortality. Data from vascular registries indicate significantly lower 30-day mortality for patients undergoing endovascular revascularization compared to open surgery [3–8]. This endovascular approach is primarily applicable to thrombotic occlusions of the SMA.
This paper presents a single-center, retrospective analysis of treatment outcomes for patients with AMI at a vascular surgery department equipped with both classical surgical and modern endovascular capabilities. We report the results of 25 patients with AMI due to SMA occlusion who underwent endovascular recanalization via either antegrade or retrograde techniques.
Aim
We conducted a retrospective study on patient series with acute visceral artery occlusions (SMA and/or the celiac trunk) to assess the effectiveness and safety of their treatment. All patients were treated with modern endovascular or hybrid techniques, which in some cases included the use of reperfusion stroke-dedicated catheters.
Material and methods
Between 2022 and 2025, 25 consecutive patients (15 women, 10 men; median age: 74 years, range: 55–83) were admitted with symptomatic acute mesenteric ischemia. Common comorbidities included arterial hypertension, dyslipidemia, diabetes mellitus, ischemic heart disease, atrial fibrillation, and chronic renal failure. A history of arterial revascularization (carotid, coronary, or lower limb) for atherosclerotic disease was also prevalent. Detailed patient clinical characteristics are summarized in Table I.
Table I
Patient and lesion characteristics (N = 25)
The etiology of occlusion was atherosclerotic thrombosis in 23 patients and peripheral embolism in 2 patients. The embolic occlusions were located at the level of the second or third division of the SMA. Among the 23 patients with atherosclerotic occlusions, the lesion was located in the initial segment of the SMA in 20 patients. In 3 patients, the atherosclerotic lesion was situated more than 10 cm from the SMA origin. Three other patients had occlusions involving both the celiac trunk (CT) and SMA, while an isolated CT occlusion was present in 2 patients. In all cases involving the CT, the atherosclerotic lesion involved the artery’s origin, often leaving only a minimal visible segment on computed tomography angiography (angio-CT) or digital subtraction angiography (DSA). The mean lesion length was 21 ±12.7 mm (range: 16–53 mm). Sixteen (64%) patients had a complete occlusion; the remaining patients presented with 95–99% stenosis and trace distal flow (Table I).
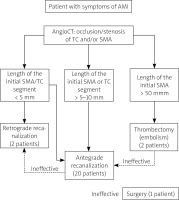
All patients presented with symptoms of bowel ischemia, with a symptom-to-admission time ranging from 3 to over 24 h (median: 5 h). Diagnosis was confirmed by angio-CT of the visceral arteries in all cases. Mean time from admission to revascularization was 2 h; therefore, the mean total ischemia time was 7 h. The revascularization strategy was determined according to the institutional protocol (Figure 1), which prioritized an endovascular approach as the first-line treatment. The specific technique – including access site (femoral, brachial, or radial) and recanalization route (antegrade from the aorta or retrograde via collaterals) – was tailored to the individual lesion. Open surgical or hybrid procedures were considered only when endovascular attempts were unsuccessful or anatomically precluded.
The preferred access site was the brachial artery, used in 23 (92%) patients, followed by radial and femoral access in 1 patient each. A 6F, 90 cm sheath (Terumo or Cook) was used, through which 4F or 5F guiding catheters of various curvatures were introduced. Lesion crossing was achieved using 0.0142 or 0.0182 guidewires, supported by diagnostic or dedicated microcatheters (e.g., Rebar, Medtronic; Progreat, Terumo). Stenting strategies were selected based on lesion location: balloon-mounted stents were deployed for proximal lesions, including drug-eluting stents in 4 (16%) cases, while self-expanding stents (e.g., Pulsar, Biotronik; Precise, Cordis) were used for lesions in the distal SMA. In 2 patients with embolic occlusions located approximately 5–10 cm from the SMA origin, aspiration thrombectomy was performed using specialized neurointerventional catheters (Sofia, MicroVention; Glutton, Plus Medica GmbH).
Standard pre-procedural management involved a multidisciplinary assessment with consultations from vascular surgery, general surgery, and internal medicine. The risks and benefits of the planned life-saving procedure were discussed with each patient, from whom written informed consent was subsequently obtained. Patient evaluation was conducted before the procedure and repeated on post-procedural days 1 and 2. This assessment included biochemical analysis of markers for bowel ischemia, such as troponin and lactate. Post-procedural imaging with computed tomography (CT) angiography or conventional arteriography was performed on day 1 or 2 if the patient’s clinical condition showed no improvement or deteriorated. Follow-up imaging was subsequently scheduled at 3 and 6 months after discharge.
Following the endovascular procedure, all patients were initiated on dual antiplatelet therapy (DAPT) on the same day. In isolated cases where surgical revision was required to assess bowel necrosis, DAPT was started post-operatively. These patients received unfractionated heparin in the peri- and post-procedural period, with a target activated clotting time of approximately 200 s. Patients were discharged 3–8 days after the procedure. The standard discharge recommendation was DAPT (aspirin plus clopidogrel or ticagrelor) for a minimum of 30 days, extended optimally to 3–6 months.
The primary endpoint of this retrospective analysis was a composite of all-cause mortality or the development of irreversible bowel ischemia requiring surgical resection. Secondary endpoints included procedure-related complications, such as visceral artery perforation, access site complications, re-occlusion, or death not attributable to bowel ischemia. Other surgical complications, including peritonitis or pancreatitis, were also recorded. Additionally, the technical feasibility of endovascular repair for the occlusions or stenoses was assessed, based on the successful application of various recanalization techniques.
Results
Recanalization was successful in 24 patients. The mean procedure time for all endovascular recanalizations was 28 min (range: 12–150 min). A retrograde approach was used in 2 cases: 1 patient underwent endovascular recanalization of the SMA via collaterals from the celiac trunk (Figure 2), while the other required a hybrid approach with retrograde open mesenteric stenting, involving direct puncture of the SMA after surgical exposure. One patient, in whom neither antegrade nor retrograde recanalization was feasible due to occlusions of the SMA and celiac trunk, underwent an ilio-mesenteric bypass. In 20 patients, a single stent provided complete lesion coverage; this included 3 patients who received one stent in the SMA and another in the CT. Three patients required the implantation of two stents. This included the retrograde open mesenteric stenting case, which used a covered balloon-mounted stent, and 2 other cases where a self-expanding stent was placed distally and a balloon-mounted stent proximally to ensure precise lesion coverage. Aspiration thrombectomy (Figure 3) was performed in 2 patients with embolic occlusions using modern, large-lumen reperfusion catheters (primarily designed for intracranial thrombectomy). These catheters can be safely navigated into the distal visceral arteries and effectively remove embolic material, typically achieving success within 1–3 passes.
Figure 2
Retrograde endovascular recanalization of the superior mesenteric artery. A – Initial angiogram of the celiac trunk showing the common hepatic artery (asterisk) and the gastroduodenal artery (white arrows). Retrograde collateral flow fills the proximal superior mesenteric artery (black arrow), but there is no antegrade flow to the distal superior mesenteric artery (X marks the site of proximal occlusion). B – A microcatheter (white arrow) is advanced retrograde through the gastroduodenal and pancreaticoduodenal arteries into the true lumen of the occluded superior mesenteric artery (black arrow, A). C – A guidewire (white arrows) is successfully navigated across the occluded segment into the aortic lumen and captured within an introducer sheath (black arrow), establishing a through-and-through access. D – Final angiogram after predilation and stent implantation (stent position indicated in the inset) demonstrates successful recanalization with restored antegrade flow into the proximal SMA (black arrow) and its branches. The guidewire path through the collateral circulation is still in place (thick arrow)
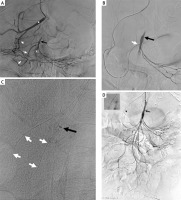
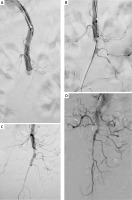
Figure 3
Aspiration thrombectomy for superior mesenteric artery embolism. A – Initial angiogram of the SMA showing an embolic occlusion (arrow) approximately 10 cm from the vessel origin. B – A reperfusion catheter (arrow) advanced to the site of the embolus. C – Completion angiogram after aspiration embolectomy, showing successful removal of the embolic material. D – Final result demonstrating restored antegrade flow into the distal SMA branches without peripheral embolization
The primary recanalization strategy was an antegrade approach from the aortic side. Given the acute angulation of the mesenteric arteries, brachial access was the preferred site. Following placement of a 6F/90 cm sheath, the origin of the occluded artery was catheterized. Recanalization was then achieved using 0.014” or 0.018” guidewires, supported by a shaped diagnostic catheter and a microcatheter. After successful wire passage to the distal vessel, the lesion was pre-dilated with an angioplasty balloon. A stent was subsequently implanted, with 2–3 mm of protrusion into the aortic lumen for lesions involving the origin of the SMA or CT. In 1 patient with a heavily calcified lesion, preparatory dilation was performed using a 6 × 60 mm shockwave lithotripsy balloon, which achieved full lesion expansion prior to stenting. For distal lesions, self-expanding stents were used. Pre-dilation was routinely performed in these cases to achieve full lesion expansion, compensating for the stent’s lower radial force. In 1 patient with celiac trunk occlusion, an initial attempt at retrograde recanalization via collaterals between the inferior and superior mesenteric arteries was unsuccessful. The procedure was therefore converted to a hybrid approach. Following surgical exposure of the SMA, successful wire passage was achieved into the aortic lumen. A covered stent was implanted from the aortic ostium. Due to incomplete lesion coverage distally, a balloon-mounted stent was deployed to ensure full revascularization.
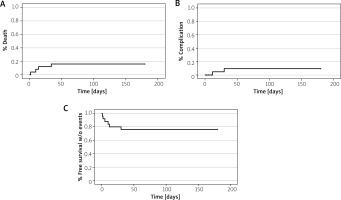
Postprocedural dialysis was not required for any patient, even among those with pre-existing renal impairment. Four in-hospital deaths occurred, representing a mortality rate of 16%. Two deaths were due to diffuse bowel necrosis requiring extensive resection. One patient died from complications of pancreatitis and peritonitis, and one death resulted from a cardiac event (electromechanical dissociation). In two additional patients, symptoms of bowel ischemia recurred on post-procedural days 10 and 15. Both patients underwent successful surgical intervention with partial bowel resection and experienced no further complications. The Kaplan-Meier analysis estimated the in-hospital all-cause mortality rate at 16%. The rate of major complications (excluding death) was 8%, resulting in a cumulative 30-day rate of major adverse events of 24% (Figure 4).
Figure 4
Kaplan-Meier analysis of clinical outcomes: A – all-cause mortality; B – non-fatal complications; C – composite event-free survival
The mean length of stay in the vascular department was 8.8 days (range: 3–58 days). During hospitalization, the degree of bowel ischemia was monitored using a combination of clinical symptoms and biochemical parameters. Serum lactate demonstrated the highest sensitivity and specificity, normalizing in all patients following successful revascularization. In contrast, other parameters – including leukocytosis, granulocyte percentage, CRP, and troponin – had a relatively prolonged normalization time and were suboptimal as early indicators, although troponin levels decreased more rapidly than typically observed in myocardial ischemia. Notably, in patients who developed bowel necrosis despite technically successful recanalization, lactate levels failed to normalize. This contrasted with patients who achieved recanalization without subsequent necrosis, in whom lactate levels consistently returned to normal. None of the patients required postprocedural dialysis, although some of these patients presented with laboratory signs of chronic renal failure.
Follow-up examinations were conducted at 3 and 6 months after discharge. No patient developed recurrent stenosis, regardless of whether the initial lesion was in the proximal or distal segment of the mesenteric artery. Clinically, no patients reported symptoms of chronic mesenteric ischemia, such as abdominal claudication.
Discussion
Based on our own results, it can be concluded that the endovascular approach to acute bowel ischemia is a valid alternative to traditional surgical revascularization. This single-center analysis of 25 patients demonstrates that modern endovascular techniques can achieve successful recanalization in a majority of cases (96%), with an in-hospital mortality rate of 16%. Our outcomes align with growing evidence that endovascular intervention, when feasible, can reduce the high morbidity and mortality historically associated with open repair for AMI [3–6]. A key finding of our study is that endovascular treatment is effective not only for embolic occlusions of the SMA but also for those of thrombotic etiology, where successful revascularization can be particularly challenging. In our cohort, 23 (92%) patients had occlusions due to atherosclerotic thrombosis, often with heavily calcified, proximal lesions. The European Society for Vascular Surgery has noted that endovascular revascularization may be particularly effective in thrombotic occlusions, likely due to pre-existing collateral circulation that develops in these chronically ischemic patients, allowing the bowel to tolerate ischemia longer [3, 4]. This physiological adaptation was evident in our patients, many of whom had prodromal symptoms, and may contribute to the technical success and favorable outcomes we observed in this subgroup [3, 4, 9–24].
However, the endovascular method is not without its technical challenges. We encountered difficulties such as catheterizing the occluded visceral artery origin, navigating guidewires across the lesion, or traversing complex collateral pathways. The high anatomical variability of the visceral arteries should always be taken into account; thus, meticulous pre- and intraprocedural imaging diagnostics aimed at identifying possible anatomical variants is pivotal for planning a safe and effective intervention [10–14]. The specific anatomy of the visceral arteries is a primary determinant of success; the acute angulation of the SMA from the aorta makes brachial or radial access preferable, as it allows for better force transmission and catheter stability. In some patients, these technical hurdles necessitate a change in strategy [10–14]. In our series, 1 patient required conversion to a hybrid retrograde open mesenteric stenting procedure after failed endovascular attempts, and another underwent ilio-mesenteric bypass. This underscores the critical importance of a vascular center equipped with both endovascular and surgical expertise, allowing for a flexible, individualized approach. Furthermore, to address the challenge of a heavily calcified lesion, intravascular lithotripsy was employed in 1 patient. This technique achieved full lesion expansion with a lower risk of dissection compared to high-pressure balloon angioplasty alone. The procedure involved four cycles of sonic energy delivery: the first during balloon inflation to 3 atmospheres (atm), the second at 4 atm, and the final two cycles at 6 atm. This approach represents a significant advancement for the safe management of ostial lesions involving the aortic wall [15–18].
A critical limitation of the endovascular approach, emphasized in the literature, is the inability to directly assess bowel viability [4, 9, 19–22]. Our data confirm that even successful revascularization does not guarantee viable intestine. In patients with extensive necrosis, bowel resection is necessary, and when it concerns a majority of the small intestine, it typically results in a fatal outcome, as observed in two of our cases. We found that serum lactate was the most sensitive biomarker for monitoring; it normalized rapidly after successful revascularization in patients without necrosis but failed to do so in those who developed bowel necrosis despite a patent artery. The exact time window for irreversible necrosis remains debated [4, 9, 19–22]. In our practice, we considered exploratory laparotomy in patients with long symptom duration and significantly elevated biomarkers, while those with shorter ischemia times and rapidly improving lactate levels were managed conservatively with good outcomes [4, 9, 19–22].
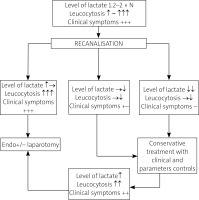
Based on our observations, we suggest a post-procedural algorithm to identify patients who, despite technically successful endovascular revascularization, may require re-laparotomy for suspected irreversible bowel ischemia. Patients with a good prognosis typically present with pre-procedural serum lactate levels not exceeding 1.5–1.7 times the upper limit of normal (ULN), after which their lactate usually drops to 1–1.1 times the ULN within 2–3 h alongside rapid clinical improvement. Conversely, in patients with a poor prognosis – typically presenting with pre-procedural lactate above 1.5–1.7 times the ULN – the decision for re-laparotomy is indicated by the persistence of clinical symptoms, such as unchanging abdominal pain and diminished bowel sounds, together with a lack of post-procedural lactate reduction and often a rising leukocyte count. While serial lactate levels are the primary predictor, the proposed decision algorithm integrates other clinical parameters. Its clinical utility, however, requires prospective validation. A suggested post-procedural algorithm is shown in Figure 5.
Figure 5
Decision algorithm for re-laparotomy, based on serial lactate levels and clinical response after endovascular revascularization
Our mortality and complication rates are consistent with contemporary literature. A recent meta-analysis reported 30-day mortality for endovascular treatment between 15% and 39%, compared to 33–50% for open surgery [3, 4, 23, 24]. Our rate of 16% falls favorably within this range. Similarly, the need for laparotomy in our cohort (16%) aligns with published data [3, 4, 23–25]. Of note, this study has the inherent limitations of a single-center, retrospective analysis with a relatively small sample size. The results reflect the experience of a specialized vascular center with significant expertise in these techniques and may not be fully generalizable. Furthermore, the choice of treatment modality was not randomized, and patient selection undoubtedly influenced the outcomes.
Conclusions
Our single-center experience demonstrates that an endovascular-first strategy for AMI is a feasible, safe, and effective treatment paradigm. This approach achieved a high rate of technical success and was applicable to both embolic and the more common thrombotic occlusions, with acceptable associated mortality. Successful implementation hinges on a multidisciplinary strategy, a versatile armamentarium of devices – including microcatheters, specialized stents, and modern reperfusion catheters – and the readiness to convert to a hybrid or open surgical procedure when technical challenges arise. Critically, while endovascular revascularization is pivotal for restoring blood flow and improving survival, the patient’s ultimate prognosis remains dependent on the extent of bowel necrosis at presentation. This underscores the indispensable role of early diagnosis and rapid intervention in the management of this life-threatening condition.