Traditionally, female sex is considered a potential factor of increased perioperative mortality in surgical revascularization. We analyzed the potential impact of sex differences in long-term survival among patients treated with the off-pump coronary artery bypass procedure, which involved arterial revascularization (anastomosis of both mammary arteries). A potentially better long-term survival rate was observed in the female group; however, large-scale studies are required to confirm the suggested association.
Introduction
Surgical revascularization, alongside percutaneous intervention, is a viable therapeutic option with satisfactory long-term results [1, 2]. Off-pump coronary artery bypass grafting is considered a valuable strategy, particularly for avoiding systemic inflammatory responses and diminished coagulation disturbances compared with the on-pump technique, as well as for its protective effects on neurocognitive function [3–5].
Accumulating evidence indicates that biological sex modulates the predisposition to coronary artery disease [6]. In general, women generally develop coronary artery disease (CAD) later in life and more frequently present with smaller-caliber coronary vessels and diffuse microvascular dysfunction, accompanied by atypical symptomatology [7].
The vast majority of patients referred for surgical revascularization are male [8]. In contrast, female sex is considered a possible perioperative mortality risk factor and included in the EUROSCORE [9]. Sex-related differences in vascular biology, including endothelial responsiveness, nitric oxide signaling, and inflammatory gene expression, have been reported [10] and may impact intraoperative hemodynamic stability. In previous reports, females’ higher rates of perioperative complications, such as a higher myocardial injury rate, have been explained by smaller body surface area, underdiagnosis or delayed treatment of CAD, and differing pharmacokinetics of antiplatelet and vasoactive medications. Sex-specific pathophysiology and technical nuances of off-pump revascularization are increasingly recognized as crucial for improving precision-based perioperative care in patients undergoing off-pump coronary artery bypass (OPCAB) surgery.
Aim
The study aimed to identify potential long-term prognostic factors after surgical revascularization in patients undergoing OPCAB using bilateral mammary arteries.
Material and methods
Patients
In total, 276 participants, including 32 females, with a median age of 62 years (range: 55–68), were included in the retrospective analysis. Only patients who underwent planned off-pump coronary artery bypass grafting between 2008 and 2015 were enrolled in the study. The inclusion criteria included the use of bilateral internal mammary arteries. The primary endpoint included the sex-related impact on long-term outcome.
All patients were operated on through median sternotomy. Both mammary arteries were harvested, and either the saphenous vein for venous grafts or the left radial artery was additionally taken if required. After heparinization, as measured by activated clotting time (ACT), anastomoses were performed. During the procedure, the ACT was maintained within 360 s. A deep pericardial suture was applied for heart weaning for circumflex artery revascularization. The anastomoses were performed after the insertion of an intraluminal shunt and regional stabilization with an Octopus III device (Medtronic Ltd, USA). The protamine was administered in two doses after completion of the anastomoses. The graft blood flow was estimated using ultrasonography equipped with a linear 6.5 MHz transducer (Verify Q, USA) as described in a previous report [2].
Statistical analysis
If the data did not follow a normal distribution, the continuous variables were reported as medians and interquartile ranges (Q1–Q3). Categorical data were presented as numbers and percentages. The Mann-Whitney test was used to compare interval parameters between the analyzed groups. Categorical data were compared using a c2 test of independence. Statistical analysis was performed using JASP version 0.14.1 (University of Amsterdam, Netherlands) (https://jasp-stats.org). Survival analyses, including Kaplan-Meier curve estimation, were performed using Python with the lifelines library (version 0.30.0). A p-value < 0.05 was considered statistically significant for all performed tests.
Results
There were 64 (23%) deceased patients among the 276 analyzed participants, with a median follow-up time of 3,307 days (range: 1,703–5,414 days). The analyzed population was divided into two groups: males (244) and females (n = 32). There were no differences in follow-up time between the groups (p = 0.179). Detailed information is presented in Table I.
Table I
Comparison of demographic, clinical, echocardiographic, and laboratory results between the analyzed groups
[i] Ao – ascending aorta diameter, AST – alanine aminotransferase, BMI – body mass index, CRP – C-reactive protein, Hb – hemoglobin, Hct – hematocrit, HDL – high-density lipoprotein, LAD – left atrium diameter, LDL – low-density lipoprotein, LVED – left ventricular end-diastolic diameter, LVEF – left ventricular ejection fraction, n – number, Plt – platelets, PVD – peripheral artery disease, RVED – right ventricular end-diastolic diameter, TG – triglycerides, WBC – white blood count, Q – quartile.
There were no significant differences between the sexes regarding either the number of anastomosed grafts (p = 0.492) or graft composition: only bilateral internal mammary arteries (BIMAs) (p = 0.545) vs BIMA + saphenous venous bypass graft (SVBG) (p = 0.061) vs. BIMA + left radial artery (LRA) (p = 0.564). Significant differences in blood flow were observed between the groups regarding left internal mammary artery (LIMA) grafting (p = 0.025) and venous grafting (p = 0.045). Analysis of the diameter of the coronary arteries, measured by applied intraluminal shunts, showed that the female group had a lower left descending artery diameter (1.50 (1.50–1.81) mm vs. 1.75 (1.50–2.00) mm, p = 0.030). When the coronary arteries’ diameter is relativized to BMI, no differences between the sexes are observed, as shown in Table II.
Table II
Comparison of demographic, clinical, echocardiographic, and laboratory results between the analyzed groups
[i] BIMA – bilateral internal mammary arteries, BMI – body mass index, Cx – circumflex artery, IABP – intra-aortic balloon pump, LAD – left anterior descending artery, LIMA – left internal mammary artery, LRA – left radial artery, mCx – marginal circumflex artery, n – number, RCA – right coronary artery, RIMA – right internal mammary artery, SVBG – saphenous venous bypass graft, Q – quartile.
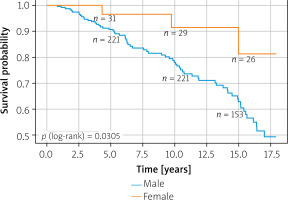
Significant differences in long-term survival between the analyzed groups were noted (p = 0.031) in Kaplan-Meier curve analysis as presented in Figure 1.
Discussion
Our analysis of long-term outcomes after OPCAB surgery in patients operated on using bilateral mammary arteries revealed a significant influence of biological sex. Our study confirms the natural trend observed in the general population, which favors better survival among females [11]. This study highlights female sex as an independent favorable prognostic factor. This advantage in the female population reflects a complex interaction of sex-specific aspects of vascular physiology and cardiovascular biology that are characteristic for women. In the female population, dyslipidemia was diagnosed less frequently, which may have a statistically significant impact on overall survival, despite optimal lipid-lowering therapy, indicating no differences in lipid profile results.
More resilient endothelial function across the lifespan, related to nitric oxide-mediated vasomodulation, is observed in females [12]. In the context of the off-pump technique, which minimizes global myocardial ischemia injury, women may particularly benefit from the reduced oxidative and inflammatory burden. This may translate into fewer microvascular complications and improved long-term myocardial perfusion.
More regulated innate and adaptive immune responses have been reported in women [13, 14]. The main advantage of the OPCAB technique is its ability to minimize inflammatory amplification associated with surgical intervention, particularly when cardiopulmonary bypass (CPB) is applied. The present single-center study results highlight the safety of long-term outcomes, with satisfactory completeness of revascularization achieved through the off-pump technique, as completeness of revascularization is considered a key factor for cardiovascular mortality [15].
The combination of off-pump surgery and intrinsically modulated female immune responses may synergistically reduce long-term vascular inflammation, mediastinal infection risk, and chronic endothelial dysfunction.
Another possible explanation for the observed phenomenon is that females may have more robust myocardial microvascular networks and an enhanced ischemic preconditioning capacity [16, 17]. OPCAB, which preserves native coronary perfusion during grafting, leverages this biology.
Smaller diameters of coronary arteries in female hearts have been reported [18], but, conversely, atherosclerotic progression after revascularization is frequently slower. The possible mechanisms may be based on differences in plaque composition and a lower systemic burden of calcific disease [19]. We also compared the diameter of the coronary arteries with the body mass index and found no significant differences between the sexes. In previous analyses, the diameter of the coronary arteries in women was relative to heart mass and showed no significant differences [20]. In our analysis, we observed significant differences in graft blood flow between the two sexes, although within an acceptable range in the analyzed groups, which may indicate a possible role in patients’ outcomes.
The present results favor female sex in long-term OPCAB surgery performed with arterial grafting, including bilateral mammary arteries. Our results are not supported by the KROK registry, as most female patients were grafted with veno-arterial grafting (using only one artery, the left mammary artery) [21]. Other possible female-specific factors that are highly influential across long-term follow-up periods are related to lower lifetime exposure to smoking in many populations and better adherence to long-term medical therapy [22, 23].
One potential factor contributing to the superior long-term survival rate in the female population may be hormonal differences, which were not examined in this study. Even after menopause, lifelong estrogen exposure continues to exert lasting structural and metabolic effects, including improved collagen organization in the vascular wall, more favorable lipid-handling pathways, and enhanced mitochondrial resilience against oxidative stress [24–26].
One may suggest that the interaction between the OPCAB technique and female physiology may result in a disproportionate benefit for women, such as maintenance of native pulsatile flow that preserves autoregulatory mechanisms potentially more effectively in female microcirculation and widely postulated reduced perioperative renal injury, because women often have a more favorable renal trajectory in the absence of CPB-related insults [27].
We present the results of a long-term survival analysis from a single center. All patients underwent surgery using the OPCAB technique, with bilateral mammary arteries used. The limited number of females enrolled in the study results from the male predominance in the group and a more conservative approach among females regarding the use of the internal mammary artery. Due to the anatomical increased risk of sternal wound dehiscence and sternum instability, arterial revascularization, including the use of bilateral internal mammary arteries in women, is not commonly performed.
Conclusions
Female sex appears to be associated with improved long-term prognosis in the analyzed population treated with OPCAB revascularization. It may be related to synergistic contributions from vascular biology, immune regulation, hormonal legacy effects, healthier postoperative behavior patterns, and a unique interaction between the off-pump technique and female physiological characteristics. While early perioperative risks may be higher in women due to anatomical and referral delay factors, the long-term survival in women may surpass that of men once the immediate postoperative period is successfully navigated. Large-scale studies are required to confirm the suggested association.