Percutaneous left ventricular assist devices (pLVAD) are increasingly used during high-risk percutaneous coronary interventions (PCI) [1]. However, their hemodynamic benefits are accompanied by potential complications, including intracardiac thrombosis, which increases the risk of central and peripheral embolization. Here, we present the case demonstrating the successful use of cerebral embolic protection for managing Impella CP-associated thrombosis risk during device removal, highlighting this technique as a viable alternative to systemic thrombolysis.
A 61-year-old male presented with non-ST-segment elevation myocardial infarction complicated by heart failure with reduced left ventricular ejection fraction of 20%. Following Heart Team evaluation, he was referred for revascularization of multivessel disease involving the left main coronary artery with pLVAD support (Figure 1 A). His medical history included type II diabetes mellitus, arterial hypertension, and persistent atrial fibrillation. Prior to the procedure, the patient underwent cardiac magnetic resonance imaging that confirmed the viability of the heart muscle and absence of contraindications to pLVAD use, including left ventricular thrombus. The vascular access sites were evaluated by computed tomography, which demonstrated favorable anatomy for large bore access (LBA).
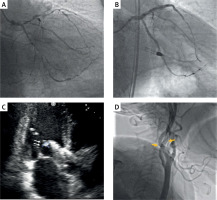
The procedure began with fluoroscopic- and ultrasound-guided LBA obtained in the right femoral artery. Following 14-French sheath insertion, intraarterial heparin was immediately administered. A standard heparin-based dextrose solution was used for pLVAD purge system. Due to unfavorable radial anatomy and the need for firm catheter support, the PCI sheath was inserted into the left common femoral artery. According to the local protocol, activated clotting time (ACT) was monitored every 30 min, with a target above 250 s during the PCI, and less than 200 s prior to the pLVAD removal and LBA closure. Protamine sulfate was not used in the presented case. During the PCI, the Impella Controller displayed recurrent suction alarms. Although hemodynamic parameters did not indicate hypovolemia and the pump position was confirmed by fluoroscopy, echocardiography revealed a floating echo in the inlet area of the pLVAD, despite proper pump positioning and normal right ventricular function (Figure 1 C). PCI of the left main coronary artery (LMCA), left anterior descending artery (LAD), and circumflex artery (Cx) was completed successfully (Figure 1 B). Given the high risk of embolization during device removal, FilterWire EZ Embolic Protection System (Boston Scientific, Mountain View, CA, USA) was deployed in the carotid arteries following completion of the PCI and removal of the guiding catheter (Figure 1 D). The pump was subsequently removed without complications, and arterial access was closed using a combination of percutaneous suture-based and collagen plug-based devices. The patient’s subsequent hospitalization was uneventful, with no neurological deficits or signs of peripheral embolization. At 2 months post-procedure, the patient remains clinically stable.
Figure 1
Multimodal imaging during Impella CP-supported high-risk percutaneous coronary intervention. A – Initial coronary angiogram demonstrating multivessel disease. B – Final angiographic result after complete revascularization. C – Transthoracic echocardiogram showing an echogenic mass (white arrows) at the inlet area of the Impella CP, suggesting either sheath-related thrombus or displaced chordae tendineae. D – Fluoroscopic image of bilateral cerebral protection devices (orange arrows) deployed prior to the Impella pump removal
Management of Impella-associated thrombosis during high-risk PCI is not well established. In cardiogenic shock cases requiring prolonged mechanical support, thrombosis may develop due to non-physiological shear stress and platelet activation caused by the pump’s complex geometrical structure. Previous reports have described successful outcomes using alteplase infusion for 8–38 h and Impella extraction under Sentinel Cerebral Protection System (Boston Scientific, Mountain View, CA, USA) [2–4].
In our case, we observed an echogenic mass in the Impella inlet area during non-emergent high-risk PCI, potentially representing either thrombus from the sheath or displaced chordae tendineae. This finding emphasizes the importance of heparin administration BEFORE large-bore sheath insertion and careful device positioning guided by both echocardiography and fluoroscopy.
We ultimately chose to avoid the excessive bleeding risk associated with alteplase administration, opting instead for off-label use of a Filter Wire EZ Cerebral Embolic Protection system, which enabled safe procedure completion. To our knowledge, this represents the first reported use of this system in the context of high-risk PCI with pLVAD support.







