Introduction
Glioblastoma (GBM) is the most common and most aggressive primary brain tumour, classified as grade 4 according to the current World Health Organization (WHO) Classification of Tumours of the Central Nervous System (CNS) [1]. The hallmark of GBM is its invasive growth and rapid spread within the nervous tissue, however, the tumour rarely metastasizes beyond the cranium [2]. Nevertheless, extra-CNS metastases (mostly lungs, pleura, lymph nodes and bones) can occur with an incidence of about 0.4–2% [3]. A review of the literature identifies approximately 12 reported cases of pleural metastases. Furthermore, GBM may transform into gliosarcoma, and radiation-induced secondary gliosarcoma has been reported in only 54 cases to date [4]. Here, we present a rare case of GBM with pleural metastasis and an uncommon TP53 mutation, complicated by secondary gliosarcoma, supported by imaging, histopathology, and next-generation sequencing (NGS).
Case report
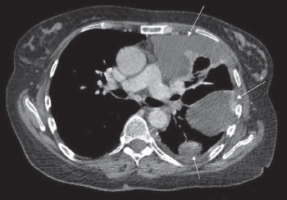
A 55-year-old woman was admitted to the hospital in September 2023 as an emergency because of increasing shortness of breath, reduced exercise tolerance, and a feeling of pressure at the bases of the lungs. She was a non-smoker and did not consume alcohol. Ultrasonography and chest X-ray demonstrated fluid accumulation in the left pleural cavity; moreover, a thoracic computed tomography (CT) scan revealed a hyperdense lesion involving the anterolateral thoracic wall, measuring 90 × 44 × 51 mm, along with several smaller lesions on the posterior wall (Figure 1).
Figure 1
Pleural metastases. Computed tomography scan showing soft-tissue thickening/tumorous lesions involving the anterior, lateral, and posterior chest wall of the left lung (arrows)
Initially, the doctors suspected a pleural mesothelioma. Cytological analysis of the material from the pleural effusion obtained in the drainage showed the presence of numerous reactive mesothelial cells and inflammatory cells. Based on these findings, left-sided video-assisted thoracic surgery with chemical pleurodesis was administered.
Biopsy specimens revealed pleomorphic malignant neoplasm with vascular proliferation and focal necrosis, but the image was nonspecific. Immunohistochemistry demonstrated the following profile: GFAP+, Calretinin–, CD30–, CD31+ (subset), CD34–, CD45 (LCA)–, CK5/6–, CKAE1/AE3–, Desmin–, ERG–, INI1+ (retained), Ki-67 ≈ 30–40% in hot spots, Napsin A–, NFP–, p40–, PAX8–, SMA focal+, SOX10–, Synaptophysin–, TTF1–, and WT1 was negative for nuclear expression. Considering the strong GFAP expression with no evidence of mesothelial marker expression, suspicion of GBM metastasis was raised (Figure 2: 2.A.B).
Figure 2
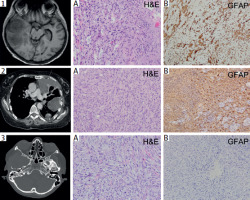
Radiological and histopathological analysis of the tumours in chronological order. 1) Initial glioblastoma. 2) Pleural metastases. 3) Gliosarcoma. The first images in each row show the corresponding computed tomography scans. 1. A) Histopathological sample from the initial brain tumour shows features of glioblastoma, including nuclear atypia and vascular proliferation (200× magnification). 1. B) Tumour cells are GFAP+, indicating glial origin typical of glioblastoma (GBM) (100× magnification). 2. A) Pleural specimen obtained during video-assisted thoracic surgery shows areas of malignancy. 2. B) Immunohistochemically, the tumour cells are GFAP+, supporting the diagnosis of pleural metastasis of GBM (100× magnification). 3. A) Biopsy specimen of the recurrent tumour shows a poorly differentiated neoplasm composed predominantly of spindle cells, lacking typical GBM morphology (200× magnification). 3. B) Negative GFAP immunostaining excludes conventional GBM and, together with other findings, supports gliosarcoma transformation (100× magnification)
Following these findings, and in view of the patient’s medical records, which indicated that she had been diagnosed with GBM in March 2021 and had undergone surgical resection, radiochemotherapy, and subsequent chemotherapy, the radiological and pathological documentation of the primary diagnosis was reviewed (Figure 2: 1.A.B). Together with the patient’s GBM history, these findings supported the diagnosis of pleural metastasis of GBM, consistent with rare cases reported in the literature.
Additionally, further immunohistochemical staining was performed to refine the diagnosis. Microscopic analysis revealed neoplastic cells positive for OLIG2 and negative for IDH1 R132H.
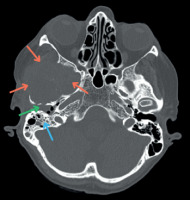
Within three weeks, the patient was surprisingly presented with symptoms of facial nerve paralysis, paroxysmal pain of the right ear, an acute headache on the right side, and bleeding from an external auditory canal. Brain CT revealed an extensive heterogeneous lesion in the place of the resected GBM (Figure 3). Biopsy specimens obtained during otoscopy showed poorly differentiated neoplastic cells positive for SMA and p53, and negative for GFAP, S100, and CK. Considering the overall clinical picture, together with the histopathological and immunohistochemical findings, these results supported the diagnosis of gliosarcoma (WHO grade 4) (Figure 2: 3.A.B). To further characterize the molecular background, NGS was performed, which revealed a TP53 mutation (chr17:7577142, ID: COSM10794, p.G266R, c.796G>A; allele frequency: 30.8%). As the tumour was inoperable, palliative treatment was initiated.
Discussion
This case illustrates a highly uncommon clinical scenario of GBM with pleural metastasis, secondary gliosarcoma transformation, and a rare TP53 p.G266R mutation. Extracranial dissemination of GBM is exceptionally rare, with fewer than 140 reported cases in the literature [5]. Its rarity is largely explained by the aggressive course of GBM, as most patients succumb to the disease before distant metastases can develop or be detected [6]. Additional protective mechanisms include the dense dural structure, lack of true lymphatic connections, and collapse of peritumoral venous channels/vessels [7, 8]. Furthermore, the extracellular matrix of peripheral organs presents a natural barrier against circulating tumour cells [9]. According to the “seed and soil” theory, metastases develop only when tumour cells find a permissive microenvironment. In the case of pleural involvement, activation of VEGFR-1 and MMP-9 expression may facilitate stromal remodelling and metastatic implantation [10]. From the cases that were reported between 1928 and 2008, GBM distant metastases affected diverse sites such as the pleura, lungs, lymph nodes, liver, skin, bones, and others [3, 5]. Multiple dissemination pathways have been proposed, including hematogenous spread, cerebrospinal fluid seeding, and glymphatic drainage [11, 12]. In our patient, the location of the primary tumour within a region of rich venous outflow may suggest a hematogenous or mixed route of dissemination.
Importantly, most reported cases of extracranial GBM metastases occur in the setting of local tumour recurrence [13], as observed here. Iatrogenic factors are also thought to play a critical role: incomplete resection, intraoperative rupture, or inadvertent cell implantation via surgical instruments may all facilitate dissemination [14]. Furthermore, up to 96% of extracranial metastases occur after neurosurgical interventions, particularly craniotomy, which disrupts the blood-brain barrier and allows hematogenous escape of tumour cells [15, 16]. Given our patient’s treatment history, such mechanisms are plausible contributors.
Gliosarcoma is a rare histologic subtype of GBM, accounting for approximately 2% of cases [17]. Most arise de novo (primary gliosarcomas), while others develop after prior GBM treatment (secondary gliosarcomas, SGSs). SGSs are uncommon and frequently exhibit loss of GFAP expression and a sarcomatoid morphology [4], as in our case. One hypothesis is that ionizing radiation promotes DNA damage and secondary mutations, facilitating sarcomatoid transformation [18], which is relevant given our patient’s radiotherapy exposure.
Studies of epithelial tumours have shown that TP53 mutations correlate with epithelial-to-mesenchymal transition (EMT), characterized by the development of spindle- shaped cell appearance, vimentin production, and nuclear expression of TWIST1 and SNAI2 transcription factors. Since similar features are present in the sarcomatous regions of gliosarcomas, this indicates that EMT processes in carcinomas and the sarcomatoid transformation seen in GBM may operate through analogous biological pathways [19]. Next-generation sequencing of the gliosarcoma specimen revealed a rare TP53 p.G266R mutation (c.796G>A; allele frequency 30.8%). Although TP53 inactivation is frequent in GBM [20], this specific substitution has been reported only once, in a 16-year-old patient with gliosarcoma and Li-Fraumeni syndrome (LFS) [21]. Our patient and her relatives lacked clinical features of LFS; however, up to 25% of LFS cases are associated with de novo TP53 mutations and no family history [22].
Conclusions
This case demonstrates three distinctive and rare phenomena: 1) pleural metastasis of GBM, reported only in a small fraction of cases; 2) secondary gliosarcoma transformation, confirmed histologically; 3) detection of a rare TP53 p.G266R mutation, previously described only once. These findings emphasize the importance of integrating radiological, histopathological, and molecular approaches when evaluating atypical GBM progression. Moreover, they suggest that surgical factors, tumour biology, and unique genetic alterations may collectively contribute to both extracranial dissemination and sarcomatoid transformation.