Arterial wall calcification affects 30–50% of patients with peripheral artery disease. These calcifications complicate endovascular therapy by increasing the risk of dissection, perforation, or embolization. They can also contribute to both early and late recoil and hinder optimal stent deployment. Furthermore, they act as a mechanical barrier, reducing the effect of drug-coated balloons. Intravascular lithotripsy (IVL) is a safe and feasible method of endovascular therapy for calcified femoropopliteal lesions, offering excellent immediate results. Concerning long-term results, no definitive evidence supports the benefit of drug-coated balloon angioplasty over plain balloon angioplasty following IVL in our pilot study. Further studies with substantially larger patient populations will be necessary.
Introduction
Arterial wall calcification affects approximately 30–50% of patients with peripheral artery disease (PAD), especially those with metabolic disorders (e.g. diabetes mellitus and chronic renal insufficiency) and older individuals. These calcifications complicate endovascular therapy by increasing the risk of arterial dissection, perforation, or embolization. They can also contribute to both early and late recoil and hinder optimal stent deployment. Furthermore, they act as a mechanical barrier, reducing the optimal effect of drug-coated balloons (DCB) [1–4].
Atherectomy is one of the treatment options for calcified peripheral arteries, designed to remove plaques. However, it carries a higher risk of peripheral embolization and arterial injury, including perforation. Higher rates of embolization may require the use of an embolic protection device, which increases both procedural costs and duration [5–8].
Intravascular lithotripsy (IVL), inspired by urologic lithotripsy used to treat renal calculi, is a different method of endovascular revascularization of calcified lesions. It uses pressure waves (sonic energy) emitted from a special balloon catheter to fracture calcifications in the intima and media. It is hypothesized that IVL may improve long-term outcomes of angioplasty by increasing arterial wall compliance prior to balloon dilatation. Additionally, IVL may enhance the efficacy of DCB angioplasty by disrupting calcified barriers, thereby enabling improved drug penetration [9–12].
Aim
The aim of this pilot study was to clarify the feasibility, efficacy, and safety of IVL in calcified femoropopliteal stenoses. Another objective was to determine whether IVL can potentiate the long-term effect of DCB angioplasty in calcified femoropopliteal lesions.
Material and methods
Study design
This prospective, single-blind, pilot study was conducted at two centers: the Department of Cardiology and the Department of Radiology (please see affiliations for details).
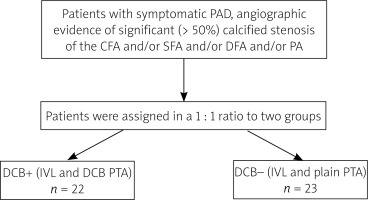
Patients were randomly assigned in a 1 : 1 ratio to two groups: the DCB plus group (DCB+) received IVL followed by dilatation with a DCB catheter, while the DCB minus group (DCB–) underwent IVL alone or, if necessary, subsequent plain balloon angioplasty at low pressure. The following method of patient allocation was employed: At both centers, patients undergoing the procedure in an odd-numbered order were assigned to the DCB– group, while those with an even-numbered order were assigned to the DCB+ group (Figure 1). All enrolled patients were included in the intent-to-treat analysis and remained blinded until the end of the study.
Figure 1
Study flowchart
PAD – peripheral artery disease, CFA – common femoral artery, SFA – superficial femoral artery, DFA – deep femoral artery, PA – popliteal artery, DCB – drug-coated balloon, IVL – intravascular lithotripsy.
At the outset of the study, the following hypothesis was established: IVL applied to calcified lesions may enhance the long-term efficacy of DCB angioplasty by disrupting the calcified barrier. If this holds true, the DCB+ group should demonstrate superior long-term outcomes compared to the DCB– group.
Patient enrollment
The study included patients with symptomatic PAD and angiographic evidence of significant (> 50%) calcified stenosis of the common femoral artery (CFA) and/or superficial femoral artery (SFA) and/or deep femoral artery (DFA) and/or popliteal artery (PA).
IVL system
The IVL system (Shockwave Medical Inc., Santa Clara, USA) uses emitters within a balloon catheter to generate pulsed mechanical discharges. In the inflated balloon catheter, these discharges create rapidly expanding and collapsing bubbles in the catheter fluid, producing shock waves that disrupt calcium deposits in the intima and media. The calcium remains in the arterial wall, forming cracks (analogous to an eggshell). Leaving the calcium in situ reduces the risk of distal embolization [9–12].
The IVL system consists of a generator, connected by a cable to a lithotripsy balloon catheter. For femoropopliteal arteries, the catheter is compatible with a 6–7 F sheath and a 0.014′ guidewire. In this study, the balloon used measured 60 mm in length, with a width ranging from 4.0 mm to 7.0 mm in 0.5-mm increments. The lithotripsy balloon is filled with a mixture of contrast medium and saline. Each catheter allows a maximum of 300 pulses, delivered in 10 cycles [9–12]. As recommended by the manufacturer, we used a slightly oversized IVL catheter to ensure optimal apposition to the arterial wall; for example, if the nominal artery diameter was 6 mm, a 6.5 mm lithotripsy catheter was used. In cases where subsequent DCB dilatation was performed, the balloon diameter matched the nominal diameter of the artery.
Study procedure
After angiographic confirmation of a calcified femoral and/or popliteal stenosis and the patency of at least one crural artery and following successful guidewire passage through the lesion, patients were randomized to either the DCB+ or DCB– group. At the start of each procedure, patients received 5,000 IU of heparin intraarterially.
A lithotripsy balloon catheter was advanced into the lesion and sequentially inflated to 2 atm, 4 atm, and finally 6 atm, using diluted contrast solution. At each balloon pressure, lithotripsy was delivered in 30-pulse increments along the entire lesion length. In the DCB+ group, final postdilatation using DCB was consistently performed. In the DCB– group, low-pressure (6 atm) postdilatation using a plain balloon catheter was performed only if residual stenosis exceeded 30%. Patients were discharged on dual antiplatelet therapy (ASA + clopidogrel), with clopidogrel discontinued 4 weeks after the procedure.
Patient follow-up
Patients underwent clinical follow-up and duplex ultrasonography at 6-month intervals for up to 24 months after the procedure. Significant restenosis (> 50%) was defined as an intrastenotic-to- prestenotic peak systolic velocity ratio (PSVR) exceeding 2.0 [13].
Endpoints
The primary efficacy endpoint was the long-term patency comparison between study groups. The secondary efficacy endpoint was acute procedural success, comparing outcomes between groups, and improvements in the Rutherford classification during follow-up [14, 15].
The primary safety endpoint was surgical revascularization and/or major amputation within 30 days after the procedure. The secondary safety endpoint was surgical revascularization and/or major amputation within 24 months after the procedure.
Assessment of calcification
The degree of calcification was assessed according to the Consensus of Peripheral Academic Research Consortium (PARC) as follows: Degree I (focal) on one side of the artery and on less than half the total length of the lesion; Degree II (mild) on one side of the artery and on more than half of the total length of the lesion; Degree III (moderate) on both sides of the artery in the same location and on less than half of the total length of the lesion; and Degree IV (severe) on both sides of the artery in the same location and on more than half of the total length of the lesion [16]. In our study, the highest degree of calcification within an individual patient was considered.
Ethical aspects
The IVL system is approved for use in our country. Before the procedure, patients received comprehensive information, had their questions answered, and signed an informed consent form for study inclusion and catheterization. The associated risks or inconveniences were limited to those of a standard catheterization procedure. In the event of method failure, IVL did not preclude the use of other endovascular or surgical treatment options. The study was approved by the ethics committees of both medical facilities.
Statistical analysis
For statistical analysis, continuous baseline variables were expressed as mean ± standard deviation (SD), with group comparisons performed using ANOVA or the two-sample t-test. Categorical baseline variables were reported as absolute frequencies and relative percentages, with comparisons between groups conducted using the c2 test. Group comparisons of long-term outcome indicators employed life table analysis and a log-rank test, with graphical representation in the form of patency curves. The Rutherford classification category shift was compared with a c2 test, and the results were represented graphically using stacked bar graphs. All tests were performed at the 5% significance level.
Definitions
Immediate (angiographic, primary, technical) success (result): revascularization with residual stenosis ≤ 30% [15, 17]. Primary patency: an uninterrupted freedom from > 50% restenosis from the time of the procedure [15, 17]. Significant restenosis (> 50%) defined by duplex ultrasonography: an intrastenotic-to-prestenotic peak systolic velocity ratio (PSVR) exceeding 2.0 [13].
Results
Patient population
A total of 45 procedures were performed on 43 enrolled patients, with 2 patients receiving treatment on both lower extremities. Our IVL procedures were conducted between November 1, 2021, and July 15, 2024. Baseline characteristics were similar between the two groups (Table I). Procedural characteristics are presented in Table II.
Table I
Baseline characteristics of patient population
Table II
Procedural characteristics
Immediate results
Angiographic success was achieved in 88.9% of procedures (40/45). Three (6.7%) patients had residual stenosis exceeding 30%. Two (4.4%) patients developed flow-limiting dissection following IVL, which required stent implantation.
In the DCB+ group, immediate success was 90.9% (20/22), while final residual stenosis > 30% occurred in 1 patient and dissection also in 1 patient. In the DCB– group, the primary success rate was 87.0% (20/23). Two patients had residual stenosis > 30% after the procedure, and dissection requiring stent deployment occurred in 1 patient.
The difference between the two groups was not statistically significant (p = 0.58). Figure 2 depicts an example of our IVL procedure.
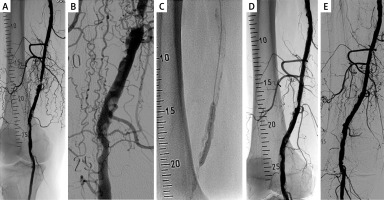
Figure 2
Example of an IVL procedure. A 78-year-old male patient, a smoker with a history of diabetes mellitus, arterial hypertension, and dyslipidemia, presented with Rutherford category 3 peripheral artery disease. Figure A shows a pre-treatment angiogram revealing a heavily calcified femoropopliteal segment. There is a severe (> 90%) proximal stenosis in the distal superficial femoral artery (indicated at position 18), and an additional significant distal stenosis in the P1 segment of the popliteal artery (indicated at position 24). Figure B provides a detailed view of the femoropopliteal segment. Following successful guidewire crossing of the stenoses, the patient was randomized to the DCB– group. Figure C displays a 6.5 ȕ 60 mm lithotripsy balloon catheter positioned at the proximal stenosis. A total of 300 pulses were delivered in both stenoses. Figure D shows a residual proximal stenosis of 40% and residual distal stenosis of 30% after the IVL treatment. Stenoses were further dilated using the same lithotripsy catheter. Figure E demonstrates the final angiographic result with no significant stenosis
Complications
The overall procedural complication rate was 11.1% (5/45). In the DCB+ group, the complication rate was 9.1% (2/22), with 2 patients developing femoral pseudoaneurysms successfully treated with thrombin. In the DCB– group, the complication rate was 13.0% (3/23); 2 patients developed larger groin hematomas, which were managed conservatively, while 1 patient experienced peripheral embolization during guidewire passage through the stenosis. The embolization was successfully resolved via percutaneous aspiration thromboembolectomy. The difference in complication rates between the two groups was not statistically significant (p = 0.67).
No embolization or arterial perforation occurred as a result of IVL or the subsequent balloon dilatation.
Long-term results
No cases required surgical revascularization within 30 days after the procedure. One patient from the DCB+ group underwent surgical endarterectomy of the femoral artery bifurcation after more than 30 days. No amputations were needed during the follow-up period.
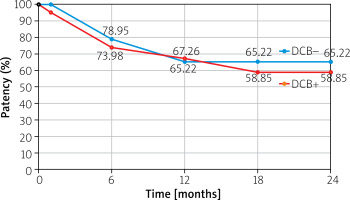
Figure 3 illustrates the primary cumulative patency for both groups during the follow-up period, as assessed by life-table analysis. Differences between the groups were not statistically significant (p = 0.56).
Figure 3
Primary patency curves for both groups by life-table analysis. At 12 months, patency rates were 67.26% (DCB+ group) and 65.22% (DCB– group). At 24 months, patency rates were 65.22% (DCB– group) and 58.85% (DCB+ group) Differences between the groups were not statistically significant (p = 0.56)
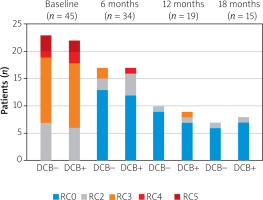
Long-term clinical outcomes were also evaluated using the Rutherford category shift (Figure 4). A significant improvement in Rutherford category was observed in all groups at each post-procedural interval compared with the pre-procedural status (p < 0.001).
Discussion
Early findings on IVL in PAD were published in 2016 and 2017 [18, 19]. However, the effectiveness of this approach in the endovascular treatment of arterial plaque calcification has not yet been thoroughly evaluated. This prospective, two-center, single-blind, pilot study attempted to assess, at least partly, the relevance of this method, especially with respect to long-term results.
It has been well established that dilatation of severely calcified lesions of peripheral arteries with DCB alone is associated with suboptimal immediate results and poorer long-term outcomes [2–4]. Therefore, our study aimed to investigate a key comparison between DCB+ and DCB– groups, assessing whether IVL-mediated disruption of calcifications may improve long-term DCB angioplasty outcomes over plain balloon angioplasty by enhancing the delivery of antiproliferative agents into tissues.
To date, our findings do not indicate a clear benefit of DCB angioplasty compared with plain balloon angioplasty following IVL. However, this finding may be influenced by the small sample size in each group (see Limitations section). To the best of our knowledge, a similar study comparing the effect of DCB and plain balloon angioplasty following IVL has not yet been published.
There are not many studies in the literature that deal with lithotripsy. To allow at least an approximate comparison of our results with others, we present several of them. Nevertheless, we are aware that such comparisons may not be fully accurate. Every study has a distinctive design and endpoints; some studies use stenting, etc. The non-randomized Disrupt PAD I Trial included 35 patients with calcified femoropopliteal stenoses. Rutherford categories 5 and 6 were excluded. IVL and in 14.3% of the cases postdilatation with an uncoated balloon were performed. The long-term patency was monitored for only 6 months and was 82.1% [19]. The Disrupt PAD II Trial was a non-randomized multicenter study involving 60 patients with calcified femoropopliteal lesions (primarily stenoses and 16.7% occlusions), including again only Rutherford categories 2–4. Postdilatation with an uncoated balloon was performed in 3.3% and stent implantation in 1.7%. Primary patency after 12 months was 54.5% [10]. The Disrupt PAD III Trial was a randomized multicenter study that included 306 patients, Rutherford category 2–4, with calcified femoropopliteal lesions (predominantly stenoses and 29% occlusions) treated with either IVL (n = 153) or plain PTA (n = 153) prior to definite treatment using either DCB or stenting. Primary patency – defined as freedom from provisional stenting, freedom from clinically driven target lesion revascularization (CD-TLR), and freedom from restenosis determined by duplex ultrasound at 1 and 2 years – was significantly higher in the IVL group (1 year: 80.5% vs. 68.0; 2 years: 70.3% vs. 51.3%). The need for stenting was significantly lower in the IVL group (4.6 vs. 18.3%). However, the two groups did not differ significantly in freedom from restenosis determined by duplex ultrasound alone at either 1 or 2 years (1 year: IVL 90.0% vs. PTA 88.8%; 2 years: IVL 83.0% vs. PTA 76.3%) [12, 20]. Stavroulakis et al. published a non-randomized trial of 55 patients, in which 71 calcified femoropopliteal lesions were treated with IVL plus DCB. Primary patency after 1 year was 81%, and freedom from CD-TLR was 92%. An important finding was that eccentric calcification of the arteries did not demonstrate inferior long-term results compared to concentric calcification [21].
Primary (angiographic) success in our study was high. This is consistent with the findings of other authors [10, 19, 21–23]. Only the Disrupt PAD Trial III reported a relatively low primary success rate, defined similarly as in our study, with 65.8% in the IVL group and 50.4% in the PTA group [12, 20].
In our population, complications occurred mainly at the puncture site and were not associated with IVL itself in any patient. The frequency of complications in our study did not exceed that reported by others. The literature indicates that the most common complication is flow-limiting dissection necessitating stent placement, with reported incidence rates ranging from 1.1% to 3% [10, 21–23]. It is interesting to note that in the Disrupt PAD III trial mentioned above, the prevalence of flow-limiting dissection reported in the IVL group was statistically significantly lower than in the PTA group (1.4% vs. 6.8%), and provisional stent placement was also lower in the IVL group (4.6% vs. 18.3%) [12, 20].
IVL is a safe treatment option. In our patients, neither surgical revascularization nor amputation within 30 days of the procedure was documented. Only 1 patient from the DCB+ group underwent surgical endarterectomy of the femoral artery bifurcation more than 30 days after the procedure. Colacchio reported in his cohort of 10 patients (12 limbs) with calcified stenosis of the CFA only one greater amputation and subsequent death [24]. Nugteren observed one amputation within 30 days of the procedure. However, this involved a patient with critical limb ischemia [25].
IVL and atherectomy, two crucial methods that focus on calcified arteries, differ in their mechanisms of action. Atherectomy debulks and removes plaques, including calcified plaques, from arteries, potentially causing more dissections and peripheral embolization than IVL [5, 6]. The mechanisms of IVL action are detailed in the Materials and methods section. However, there are no studies in the literature that compare IVL and atherectomy. A notable exception is the work of Baig et al., a retrospective analysis of 68 calcified stenoses of the CFA. Of these, 35 were successfully treated with atherectomy and DCB, and 33 were successfully treated with IVL and DCB. There was no statistically significant difference in the 18-month freedom from CD-TLR between the two groups (91.2% in the atherectomy group and 79.4 % in the IVL group) [8].
The limitations of our study – and thus the potential for bias in the interpretation of the results – are related both to (1) the inherent nature of the study and (2) the small number of patients in each group. Regarding point 1, catheterization studies are mostly single-blind; unlike drug trials, these studies cannot be organized as double-blind because the operator must know the patient’s assignment in order to apply the appropriate procedure, which typically differs between study arms. Regarding point 2, given the small number of patients in our study, we consider it a pilot study preceding a larger trial.
Conclusions
In calcified femoropopliteal lesions, IVL represents a safe and feasible method with favorable immediate outcomes. It enables endovascular management of ‘non-stent zones’ – the femoral bifurcation and the femoropopliteal artery. Concerning long-term results, no definitive evidence supports the benefit of DCB angioplasty over plain balloon angioplasty following IVL in our pilot study. Further studies with substantially larger patient populations will be necessary.