Introduction
The risk of myelodysplastic syndromes or acute myeloid leukaemia (AML) is a rare but potentially fatal side effect following treatment with chemotherapy, particularly with alkylating agents, topoisomerase II inhibitors, and platinum compounds. In breast cancer patients, this risk is well known, although most of the evidence relates to chemotherapy that is less commonly used today [1–3]. We know that the therapies used increase the risk of cumulative damage to haematopoietic DNA, favouring chromosomal alterations characteristic of myelodysplastic syndromes and AML. The results of Kantarjian et al. confirmed that monosomy 7 is a characteristic cytogenetic abnormality in this context and is usually associated with a worse prognosis [4–6].
Metronomic chemotherapy, which consists of the continuous administration of lower doses of cytotoxic agents, is easy to administer orally and has a pleasant dosage, making the treatment a choice to offer previously poly-treated patients. And it is believed that its mechanism of action may not be related to its cytotoxic activity, but rather, for example, to its angiogenic action [7, 8].
In the case of triple-negative breast cancer (TNBC), strategies and data on long-term survivors who have undergone multiple treatments are scarce. A subgroup analysis of the VICTOR-6 study suggests that metronomic chemotherapy may play a role as a maintenance strategy or in situations where a rapid response is not necessary, reflecting its less aggressive profile [9, 10].
Below we report the case of a patient with advanced TNBC, who underwent subsequent treatments for low-volume disease, and after an almost complete response, began maintenance treatment with metronomic cyclophosphamide. Seven years later she developed a myelodysplastic syndrome associated with monosomy 7. This case was described according to the CARE guidelines [11].
Case report
In 2011 (baseline – month 0), a 35-year-old woman was diagnosed with breast carcinoma after mammography and breast ultrasound. She then underwent partial mastectomy and sentinel node biopsy, confirming a triple- negative invasive ductal carcinoma of the left breast (TNBC), grade 3, with a final stage of IA (pT1cN0M0). There was no elevation of the CA 15.3 tumour marker at diagnosis. Between months 4 and 8, the patient received adjuvant treatment with FEC-D (5-fluorouracil + Epirubicin + Cyclophosphamide + Docetaxel).
She subsequently underwent radiation treatment on the left breast for a total dose of 50 Gy/25 fractions followed by a boost of 10 Gy/5 fractions on the tumour bed (months 9 and 10). In 2012, she began surveillance. A germline DNA testing was completed to check for pathogenic variants of BRCA1 and BRCA2 with negative results.
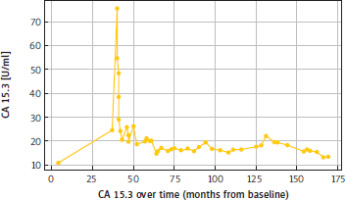
In 2014 (month 37), she presented with a palpable cervical left lymph node recurrence on physical examination, associated with an elevation of the CA 15.3 tumour marker – 75.76 U/ml (normal < 25 U/ml). The re-staging with chest/abdomen/pelvic computed tomography and bone scintigraphy revealed lymph nodes metastases (left cervical, mediastinal and hilar), bone (sternum) and lung (bilateral) metastases (Figure 1, 2). A lymph node biopsy was performed, confirming the recurrence of previously treated TNBC, programmed death-ligand 1 negative (combined positive score 3) and HER2 low (immunohistochemistry 2+ and fluorescence in situ hybridization (FISH) negative) (Figure 3 A–E).
Figure 1
Bone scintigraphy – peripheral radiotracer uptake in a small focal lesion located in the sternal body (metastasis) (blue arrow)
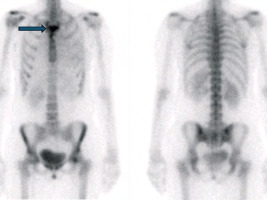
Figure 2
Computed tomography scan – multiple scattered micronodules distributed throughout both pulmonary lobes (blue arrows)
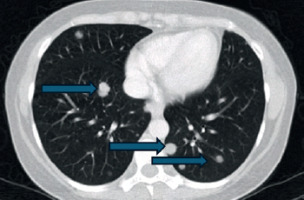
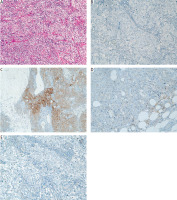
Figure 3
Histopathological and immunohistochemical findings of the primary breast tumour. A) Hematoxylin and eosin staining showing invasive ductal carcinoma with high-grade features (original magnification 100×). B) Negative expression of oestrogen receptors in immunohistochemistry (200×). C) HER2 immunohistochemistry showing equivocal membranous staining (score 2+) (200×). D) Programmed death-ligand 1 expression assessed by combined positive score of 3 (200×). E) Immunohistochemistry showing absence of androgen receptor expression (200×)
She began first-line treatment with gemcitabine/ carboplatin (month 40) (Table 1), presenting with a complete response according to RECIST 1.1 in 2015 (month 46) with normalisation of CA 15.3 (Figure 4). Two dose reductions were required due to haematological toxicity, namely persistent grade 3 neutropenia, according to the Common Terminology Criteria for Adverse Events (CTCAE), version 4.03. Treatment continued until 2016 (12 cycles – month 52) when disease progression was observed in a single hilar lymph node lesion and in the sternum lesion.
Table 1
Chemotherapy regimen
She began a second line of palliative treatment with weekly paclitaxel 80 mg/m2 (D1, D8, D15, every 28 days) (Table 1), which was performed for 7 months, with the disease remaining stable according to RECIST 1.1. However the patient developed persistent grade 2 peripheral neuropathy (CTCAE v4.03).
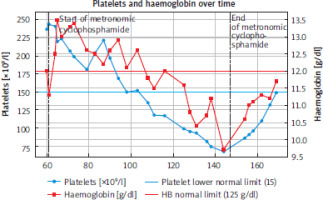
At that time, given that the only measurable disease was a lesion in the sternum, and after discussion in a multidisciplinary group, it was proposed to perform cryoablation of the bone lesion, followed by maintenance treatment with metronomic cyclophosphamide (50 mg oral daily). The patient began this metronomic treatment in 2016 (month 60) (Table 1), and in the multiple subsequent radiological reassessments, she always maintained complete response criteria and good clinical and analytical tolerance, until 2020 (Month 109). At that time the patient developed sustained thrombocytopenia, initially Grade 1 (CTCAE v5), which progressed to Grade 2 (Figure 5) . In 2023 (month 144), a Grade 2 anaemia was also documented.
Figure 5
Graphical representation of haematological toxicity, as reflected by anaemia and thrombocytopenia, from the start of oral cyclophosphamide treatment
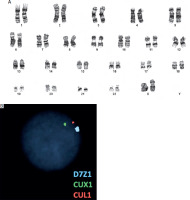
She was referred to the haematology department for further investigation, which included extensive laboratory tests that identified Grade 1 vitamin deficiency, Helicobacter pylori infection, and previous hepatitis C virus infection (without active viral load). A bone marrow aspiration was performed, which showed trilineal dysplasia without excess blasts, with normal immunophenotyping. The cytogenetic study identified the presence of monosomy of chromosome 7 (Figure 6).
Figure 6
Cytogenetic and fluorescence in situ hybridization analysis of bone marrow cells (FISH). A) Conventional karyotype analysis showing no additional numerical or structural chromosomal abnormalities. B) Interphase FISH analysis using probes targeting chromosome 7 (D7Z1/CUX1/CUL1), demonstrating loss of one signal in a subset of nuclei, consistent with monosomy 7
Detected in 15% of the analysed cells, exceeding the technical detection threshold (≥ 10%).
Following these results, it was possible to establish a diagnosis of myelodysplastic syndrome associated with monosomy of chromosome 7, low risk, IPSS-R 2.8 (Revised International Prognostic Scoring System), of multifactorial cause but with a primary contribution from prolonged exposure to chemotherapy. The patient also underwent eradication of Helicobacter pylori infection and began correction of identified vitamin deficiencies.
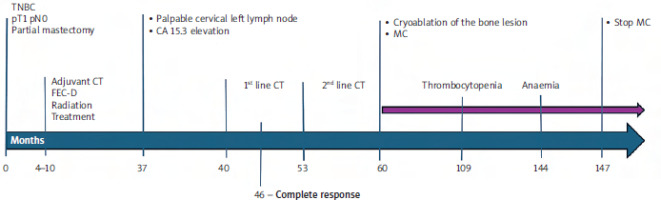
It was decided to suspend cyclophosphamide (month 147) to prevent worsening bicytopenia in a patient who had been disease-free for several years. She remained under haematological surveillance, having recovered from anaemia in September 2024 and from thrombocytopenia in April 2025. She currently remains without evidence of disease progression. The course of the disease is presented in Figure 7, and the cumulative doses of all treatments are described in Table 1.
Figure 7
Timeline of the patient’s clinical course, showing treatments, disease response and key events over time
CT – computed tomography, FEC-D – 5-fluorouracil + Epirubicin + Cyclophosphamide + Docetaxel, MC – metronomic cyclophosphamide, TNBC – triple-negative breast cancer
It is expressed in months from baseline.
Discussion
The risk of developing therapy-related myelodysplastic syndrome (t-MDS) increases with cumulative exposure to DNA-damaging agents, particularly alkylators and platinum compounds. Monosomy 7, as identified in this patient, is a typical cytogenetic hallmark of treatment- related disease and is associated with adverse prognostic implications [12, 13]. In metastatic TNBC, where long-term survival is uncommon, the evidence supporting prolonged maintenance strategies is scarce, and there are no established recommendations regarding the duration of metronomic treatments [14, 15].
This case highlights three clinically relevant aspects. First, the patient experienced an unusually durable disease control, maintaining a complete response for several years in a biologically aggressive subtype. Such long-lasting stability is rarely observed in advanced TNBC and raises the question of whether continuous exposure to maintenance chemotherapy is necessary once sustained remission is achieved [15], but also if biological differences between triple-negative tumours could perhaps justify this behaviour.
Second, the emergence of late-onset bicytopenia and subsequent diagnosis of t-MDS is consistent with cumulative, chemotherapy-related toxicity. Although the impact of prolonged daily cyclophosphamide administered over several years cannot be ruled out, its relative significance compared with prior cytotoxic exposures cannot be ascertained.
While blood counts improved following the discontinuation of metronomic cyclophosphamide, this should not be taken as proof of clonal regression or the reversibility of myelodysplastic syndrome. Haematological improvement is a distinct response domain from cytogenetic response, and no follow-up cytogenetic or FISH analyses were performed given the partial recovery of cytopenias. This represents a limitation of the present report.
The challenge lies in balancing the perceived benefit of extended maintenance therapy against the risk of late toxicity, particularly in patients who exceed expected survival benchmarks. Although metronomic regimens are generally considered less intensive, their long-term safety is insufficiently characterized, as few patients receive these agents for many consecutive years [14, 16, 17]. While a direct causal relationship cannot be established, prolonged maintenance with daily cyclophosphamide was temporally associated with the haematological abnormalities observed in this patient. This case provides supportive evidence that metronomic chemotherapy, even when administered at low doses, may be clinically relevant for patients with a history of cumulative exposure to cytotoxic agents.
In this context, treatment decisions should be individualized, incorporating disease burden, treatment tolerance, expected benefit, and patient preferences. Importantly, the absence of measurable disease for an extended period underscores the need for regular reassessment of the indication for ongoing therapy. In long-term responders, particularly when no measurable disease persists, the role of continuous metronomic chemotherapy becomes uncertain.
Treatment holidays may be considered to mitigate cumulative toxicity without compromising disease control, however, we tend to avoid it for fear of the rapid progression typical of triple-negative aggressiveness. Shared decision-making remains essential, especially when the therapeutic intent is palliative and the equilibrium between quality of life and late toxicities becomes increasingly delicate [15, 16].
Third, although prolonged exposure to alkylating agents is a well-established risk factor for therapy-related myeloid neoplasms, persistent cytopenias are non-specific findings. A comprehensive evaluation is required to rule out or address other possible causes, such as nutritional deficiencies, chronic infections, autoimmune cytopenias, liver disease or drug-induced bone marrow suppression [18]. This diagnostic challenge is particularly relevant in long-term survivors of metastatic triple-negative breast cancer, as prolonged survival allows late treatment-related toxicities to emerge and alternative, potentially reversible causes of cytopenia to coexist. In this patient, identifying vitamin deficiencies, a Helicobacter pylori infection and past hepatitis C exposure highlights the importance of considering a wide range of possible causes of cytopenia before attributing it solely to chemotherapy-related toxicity.
To our knowledge, there is very little published data on t-MDS in long-term survivors of metastatic TNBC who have undergone prolonged metronomic chemotherapy. This reflects the uncommon nature of prolonged survival in this biologically aggressive subtype.
Conclusions
This case highlights the exceptional clinical course of a patient with metastatic TNBC who achieved long-term disease control. This scenario remains rare and is not well characterised in the literature.
This report illustrates that while metronomic cyclophosphamide may offer prolonged disease control in selected patients with metastatic TNBC, sustained exposure to alkylating agents may be associated with clinically relevant late haematological toxicity. The emergence of therapy- related MDS after several years of treatment underscores the need for regular haematological monitoring and periodic reassessment of the ongoing indication for maintenance therapy. In the absence of evidence-based recommendations regarding treatment duration in long-term responders, decisions should be individualized and grounded in shared decision-making, weighing the potential benefits of continued therapy against the cumulative risks and the overall impact on quality of life. Furthermore, persistent cytopenias should prompt an evaluation of alternative, potentially reversible causes, rather than being solely attributed to chemotherapy-induced toxicity.