Left atrial appendage closure (LAAC) lowers embolic risk in selected atrial fibrillation (AF) patients and reduces bleeding related to long-term oral anticoagulation (OAC) [1]. Catheter ablation and sinus rhythm maintenance may limit atrial remodelling, improve contractile function, and reduce cardiovascular events. Performing AF ablation and LAAC in a single session can reduce hospitalisations, anaesthesia exposures, transseptal punctures, and imaging studies, accelerating achievement of rhythm control plus stroke prevention without chronic OAC. Pulsed-field ablation (PFA), an electroporation-based modality, minimises thermal injury compared with radiofrequency or cryoablation and likely causes less pulmonary ridge oedema [2–4]. This provides a rationale for a PFA first, LAAC-second strategy, as limited ridge swelling should not significantly distort device sizing.
We report one of the first Polish one-stop PFA-LAAC cases.
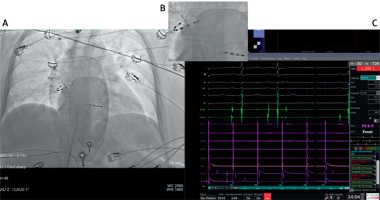
A 79-year-old woman with paroxysmal AF, CHA2DS2-VA = 3, HAS-BLED = 3, and a contraindication to long-term OAC after prior intracranial haemorrhage presented with symptomatic, drug-refractory AF (EHRA III). Comorbidities included hypertension, asthma, peptic ulcer, and euthyroid hyperthyroidism. Transthoracic echocardiography showed an enlarged left atrium (49 mm) with no other abnormalities. Dabigatran 150 mg was last taken 24 h before the procedure. Under deep propofol sedation, transesophageal echocardiography (TEE) excluded LAA thrombus and provided baseline measurements. Two ultrasound-guided right femoral venous access sites were obtained (PFA/LAAC systems), and a diagnostic RV-electrode was placed via the second sheath. Transseptal puncture was performed using FlexCath Cross™ (infero-posterior site). Electrical cardioversion restored the sinus rhythm. The PulseSelect™ catheter (Medtronic) was advanced sequentially to each pulmonary vein for isolation, with applications at ostial and antral positions according to the manufacturer’s guidance. Efficacy was confirmed by entrance/exit block in all veins (Figure 1 C).
Figure 1
Fluoroscopic and electrophysiological monitoring during pulsed-field ablation (PFA) for atrial fibrillation. A – Anteroposterior fluoroscopic projection showing the positioning of the PFA catheter within the left atrium. B – Magnified view of the catheter in the right superior pulmonary vein, confirming stable contact and complete circumferential expansion of the array. C – Corresponding electrogram recordings from the ablation system confirming exit block at the pulmonary vein ostium
In the second stage, a Nitinol AnchorMan® 23-mm occluder (MicroPort CardioAdvent) with 12 “3D-folding” modules, 12 anchors, and a PET membrane, was implanted per the instructions for use. Before release, standard criteria were verified: stable anchoring, acceptable peri-device leak (target < 3 mm), device compression (target ~10–25%), favourable device plane, and absence of conflict with the mitral valve, the circumflex artery, or the LUPV. The device was then detached, and femoral haemostasis was achieved with a “Z” stitch. Total procedure time was 63 min (ablation 43 min), fluoroscopy 16 min; DAP 6,935 cGy·cm2; contrast 30 ml. No periprocedural complications occurred.
Dual antiplatelet therapy was prescribed for 45 days, followed by single antiplatelet therapy for 12 months. Although anticoagulation after AF ablation is currently recommended based on individual thromboembolic risk, irrespective of the rhythm status, emerging randomised evidence, such as the ALONE-AF trial, suggests that OAC discontinuation may be feasible in carefully selected patients who remain arrhythmia-free for ≥ 1 year after ablation [5]. However, this approach is not yet the standard of care for patients at high thromboembolic risk. Also, in the OCEAN trial, rivaroxaban did not significantly reduce the composite of stroke, systemic embolism, or new covert embolic stroke compared with aspirin in patients who had undergone successful ablation ≥ 1 year earlier, while bleeding outcomes favoured aspirin [6].
In the present high-risk case, the goal of avoiding long-term NOAC was primarily addressed by concomitant LAAO following the results of the OPTION trial, showing that left atrial appendage closure after catheter ablation was non-inferior to continued OAC for stroke and systemic embolism and was associated with a significantly lower risk of non–procedure-related bleeding compared with long-term anticoagulation [7]. Thus, post procedural antithrombotic therapy followed a device-healing strategy (DAPT for 45 days, then SAPT for 12 months) aimed at minimising device-related thrombosis, with planned follow up imaging. Follow-up includes TTE at 45–90 days and Holter ECG at ~6 months.
The same-session PFA-LAAC – pulmonary vein isolation by PFA followed by immediate LAAC – was feasible, efficient, and safe in this high-bleeding-risk patient with contraindication to long-term OAC. The strategy leverages PFA’s non-thermal nature to maintain reliable LAA sizing. It compresses the care pathway into a single encounter, potentially reducing resource use and time-to-benefit.
Nevertheless, careful assessment for acute post-ablation tissue oedema is advisable when performing same-session PFA-LAAO. In a multicentre comparative study, Tam et al. reported a mean 69.4% increase in pulmonary ridge thickness immediately after PFA and a higher prevalence of significant peri-device leak (PDL > 3 mm) at 3 months after combined PFA + LAAO compared with standalone LAAO (20% vs. 2.2%), despite similar overall PDL rates [8]. These findings suggest that acute ridge thickening after ablation may adversely affect final sealing (and potentially sizing) and underscore the need for meticulous intraprocedural imaging, repeat measurements after PVI, and scheduled TEE and/or CT follow-up.
As one of the earliest Polish experiences, this case supports a broader evaluation of PFA-LAAC in carefully selected patients through registries and prospective studies. In the United States, reimbursement for a combined one-stop ablation plus LAAC procedure has been available since 2024.







