Summary
Gerbode defects, once primarily congenital, are now more frequently acquired – typically as a complication of cardiac surgery or infective endocarditis. However, due to the rarity of the condition, the available literature is limited to individual case reports and small case series, highlighting a significant gap in high-quality evidence regarding optimal treatment strategies. Our study, based on data from a single-center prospective registry, demonstrates that catheter-based, percutaneous closure of right atrium–left ventricle (RA–LV) shunts is a viable and less invasive alternative to traditional surgical correction. These findings suggest a potential shift in clinical practice toward minimally invasive intervention for selected patients with acquired Gerbode-type defects. Further multicenter studies and long-term outcome data are needed to validate this approach and form standardized treatment guidelines.
Introduction
The term Gerbode defect (GD) describes a very rare condition: a defect in the membranous portion of the ventricular septum with left ventricular-to-right atrial communication [1]. Depending on the shunt anatomy or jet direction, we can distinguish direct GD, with the shunt entering the right atrium (RA) directly from the left ventricle (LV), and an indirect connection of the LV with the RA via the right ventricle [2].
Gerbode defects can be categorized as congenital (rare, < 0.1%) or acquired. The acquired GD form, mostly iatrogenic, is currently more common. Iatrogenic GD may occur following cardiac surgery (mitral and aortic valve replacement, tricuspid annuloplasty, surgery for congenital defects such as tetralogy of Fallot or double outlet right ventricle [DORV]), electrophysiological procedures (e.g., arrhythmia ablation in the region of the atrioventricular node), or transcatheter therapies (e.g., transcatheter aortic valve implantation [TAVI]) [3–6]. Non-iatrogenic GD may result from infective endocarditis, penetrating or blunt chest trauma, or myocardial infarction [7, 8].
In the past, GD was a routine indication for surgery in both acquired and congenital cases. The rapidly growing number of patients with high surgical risk, due to significant comorbidities which may influence the peri- and postoperative course (prosthetic valves with increased thrombotic risk, conduction disturbances, fibrosis, or deterioration of tissue quality), has led to a search for less invasive techniques.
As the GD is a rare condition, there are no formal guidelines or recommendations on treatment. There are several approaches that can be applied for this kind of defect, such as conservative treatment, cardiac surgery, transcatheter intervention, or intraoperative device closure [9]. With the development of imaging (MR, 3D-TEE) and percutaneous techniques, an accessible and feasible method of alternative treatment is now available for patients with LV-RA leak, especially in high-risk groups [3, 10]. Data from the multicenter, international study MIOS-MFO concerning treatment of patients with percutaneous closure of perimembranous VSD with a KONAR-MF device indicate that this procedure is a safe and technically feasible option [11]. The rate of complications was low, and such management may be considered an alternative for surgery, especially in high-risk patients.
We evaluated the application of a telescopic system using a steerable sheath with mother-in-child, venous access with no rail and 3D navigation to assess the efficacy of such an approach in GD patients.
Aim
The aim of this study was to assess the clinical characteristic, procedural efficiency, and results of transvenous treatment of GD using a telescopic system in 7 consecutive patients.
Material and methods
Study design and population
This single-center, prospective registry study analyzed all consecutive patients treated using a percutaneous telescopic system between 2003 and 2025. Inclusion criteria were age over 18 years, symptomatic GD confirmed by an echocardiography specialist, and formally approved by the local Heart Team for transcatheter closure. Patients with symptoms of active infective endocarditis (IE) were excluded from the registry.
Pre-procedural evaluation
Pre-procedural assessment consisted of clinical evaluation (symptoms, NYHA class, liver function), documentation review with regard to previous heart surgery, and exclusion of active infection. Transesophageal echocardiography with 3D (RT-3D-TEE) and multiplanar reconstruction was used to assess the defect’s location, size, margins, and relations to adjacent tissues and valves (tricuspid and aortic) to evaluate potential procedural risks. Additionally, right heart catheterization was performed to measure pulmonary pressures.
The Heart Team – consisting of a cardiac surgeon, invasive cardiologist, and imaging specialist – analyzed potential risks and benefits in the context of the patient’s preferences, comorbidities, and expected technical difficulties.
Procedure
Successful closure was defined as successful device delivery and implantation with complete closure of the GD without any persistent leak and without periprocedural complication (e.g., device migration, interference with adjacent prosthetic or native valves).
Periprocedural safety was assessed by analyzing both periprocedural and in-hospital complications, with the focus on major complications (pericardial effusion, thromboembolism, occluder instability, stroke, new permanent conduction disturbances, new-onset hemolysis).
All procedures were performed in conscious sedation under RT-3D TEE guidance, without the use of a contrast agent. A steerable 8.5 F sheath (Agilis, Abbott Vascular, USA) with a telescopic system coronary catheter (125 cm JR4 5F diagnostic catheter inside of the 100 cm 6F EBU guiding catheter) was introduced into the RA through the right femoral vein. A 0.035” straight hydrophilic wire was used to cross the defect, after which the system was replaced with a delivery sheath. The sheath’s tip was positioned in the left ventricular outflow tract (LVOT). The type and size of closure devices were selected based on CT and intraprocedural TEE. Median procedural time was 50 min (range: 45–60 min). Device success was assessed using TEE. A comprehensive description of the procedure can be found in Table I.
Table I
Procedural data
Results
Patients’ characteristics
We included 7 patients (3/7, 43% male), median age 54.7 years (30–85 years). Six patients had a history of previous heart surgery, including aortic valve replacement (n = 3), mitral valve replacement (n = 1), tetralogy of Fallot repair followed by aortic valve replacement (AVR) (n = 1), and mitral/tricuspid annuloplasty, with left atrial appendage closure (n = 1). One patient was referred after being successfully treated for infective endocarditis in the past. All patients presented with symptomatic right-sided heart failure (NYHA class III, n = 6; NYHA class IV, n = 1), with echocardiographic evidence of right ventricular dilatation. Four patients had hepatomegaly, four leg edema, and five hemolysis. Median preprocedural pulmonary artery pressure (mPAP) was 41 mm Hg (34–70 mm Hg). Most of the patients were in sinus rhythm, one had atrial fibrillation, and two had a DDD pacemaker (Table II).
Table II
Patients’ characteristics
[i] F – female, M – male, MAVR – mechanical aortic valve replacement, mechanical, MMVR – mechanical mitral valve replacement, ToF – tetralogy of Fallot, IE – infective endocarditis, NYHA – New York Heart Association class, RV – right ventricle, BAV – bicuspid aortic valve, MVRS – mitral valve repair surgery, TVRS – tricuspid valve repair surgery, LAAC – left atrial appendage closure, AF – atrial fibrillation, DDD – DDD pacemaker.
Procedural outcomes
Percutaneous closure was achieved in 6/7 patients (86%). One patient had a small residual shunt despite implantation of the largest available membranous VSD occluder, resulting in persistent hemolysis requiring surgery. Two of 7 patients (33%) developed new atrioventricular conduction disturbances requiring pacemaker implantation; one of them also developed cardiac tamponade, from a temporary pacing electrode.
Echocardiography at discharge confirmed complete closure in all successfully treated patients, with no new aortic or tricuspid regurgitation. In 6/7 patients (86%), assessment of tricuspid regurgitation showed trace to mild tricuspid regurgitation, in 1 patient mild to moderate jet.
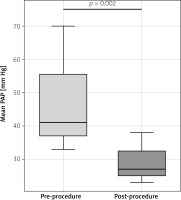
Postprocedural right heart catheterization showed a significant reduction in mPAP (median: 27 mm Hg; 23–39 mm Hg; p = 0.002) (Figure 1).
Six patients were NYHA I at discharge. One patient with coexisting pulmonary hypertension was NYHA III. No hepatomegaly or edema was observed at discharge.
Discussion
Surgical risk
Acquired GD most commonly follows previous heart surgery or infective endocarditis [4–6, 8]. No standardized treatment guidelines exist due to the rarity of this condition. Management must be individualized, based on the shunt’s hemodynamics, comorbidities, and endocarditis risk. Small, asymptomatic defects may be treated conservatively; however, in patients with prior surgery, repeated operations, valve explantation, and tissue fragility increase perioperative morbidity and mortality [1, 12, 13]. In our series, mean EuroSCORE was 4.8% though perceived surgical risk was significantly higher due to prior procedures and tissue fragility. Six of 7 patients (86%) had undergone at least one prior cardiac operation. In this cohort, percutaneous GD closure offered an alternative to high-risk surgery, achieving complete closure in 6/7 patients (86%). However, data remain insufficient for direct comparison with surgery, where reported success is only moderate (~75%) [13].
Percutaneous telescopic GD closure
Percutaneous telescopic GD closure offers a minimally invasive alternative to surgery. The approach requires careful imaging, appropriate device selection, and access planning (antegrade vs. retrograde). As percutaneous repair of GD is considered as an off-label method, there is no consensus on the optimal approach. The choice of antegrade or retrograde approach depends on shunt anatomy, operator preference, and vascular access, with the retrograde approach reported both in congenital and acquired defects [14]. We proposed antegrade access with use of a steerable sheath and telescopic system, the combination of which with 3D RT TEE allowed precise device positioning. Visualization of the tricuspid valve is challenging, but the septal leaflet is usually distinct and may be used to navigate during the procedure. The GD anatomy (tortuosity and length) may differ from congenital membranous VSD, allowing the use of various occluders: membranous VSD, ADO, or ASO, or newer devices such as the KONAR-MFO [4–6, 15, 16]. The latter allows either an antegrade or a retrograde approach and is widely used for VSD closure, with high procedural success and a low rate of atrioventricular conduction disturbances [15]. In our series, 6/7 shunts (86%) were closed using a membranous VSD and one with ADO II (Figure 2). While our series is small, these results are consistent with emerging evidence supporting off-label transcatheter closure of membranous defects using double-disk devices [15, 16]. Such devices allow antegrade or retrograde approaches and have demonstrated acceptable procedural success and low rates of complications in other studies.
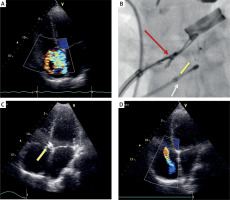
Figure 2
A – TEE showing right atrium with significant color Doppler jet indicating GD/tricuspid regurgitation. B – Occluder implantation: yellow arrow – asymmetrical Amplatzer pmVSD occluder attached to delivery system; red arrow – dedicated delivery system; white arrow – pacing electrode. Mechanical valve situated just above occluder. C – TEE confirming occluder implanted at site. D – TEE of the right atrium showing mild tricuspid regurgitation jet and complete reduction of GD jet
All patients in this series had severe right heart failure and were considered high surgical risk. The procedures were performed under conscious sedation, without the need for extracorporeal circulation. RT 3D-TEE navigation allowed the entire procedure to be conducted without contrast agents. The median procedural time was 50 min, facilitated by the telescopic approach, which improves maneuverability. Data in the literature concerning procedure time in percutaneous closure of acquired GD are limited in congenital cases; procedures usually take 30–50 min, with the fluoroscopy time under 15 min. These factors suggest that the percutaneous telescopic approach may be a feasible alternative in the high-risk population.
Complications
Reported complications of GD closure include residual shunt and hemolysis, usually transient. In this series, 1 patient developed persistent hemolysis due to a trivial shunt and required surgical revision. Conduction disturbances, particularly AV block, are another potential complication due to proximity to the conduction bundle. Data on AV block in Gerbode patients are limited; in surgical cases, AV block of various degrees was reported in 36% of patients and up to 43% in the endocarditis subgroup [8, 13, 17]. Yuan reported the incidence up to 75% in surgical patients compared with 23% in the interventional group [17]. In the percutaneous closure of perimembranous VSD, AV block occurs in about 0.5–1.5% of cases, while the incidence of atrioventricular block after surgery is 1.1% [14, 18, 19]. In our series, 2/7 patients (28%) required pacemaker implantation. This emphasizes the need for softer occluders to minimize tissue compression and reduce conduction disturbances.
Conclusions
In this small series, percutaneous closure of an acquired Gerbode defect using a steerable sheath and telescopic system under the 3D-RT TEE was feasible in 5/7 patients (71%).
While percutaneous closure in this high-risk, heterogenous population appears to be feasible and reasonably safe, we would like to highlight the need for appropriate patient selection and operator experience. Further studies with longer follow-up are needed to confirm the safety and efficacy of this approach.