Summary
This study analyzed radiation exposure, contrast use, and predictors of major adverse cardiac events (MACE) in 281 patients undergoing percutaneous coronary intervention (PCI) for calcified lesions. Results showed that operator, procedural – such as ad-hoc procedures and interventions on chronic total occlusions – and patient factors like male gender and chronic kidney disease significantly influenced radiation dose and contrast volume. Higher radiation exposure was associated with complex procedures and increased number of interventions. The risk of MACE was elevated in patients with chronic kidney disease and severe calcifications. The findings underscore the importance of managing procedural and patient-related factors to minimize radiation, contrast exposure, and MACE during PCI for calcified lesions.
Introduction
Challenges encountered during percutaneous coronary interventions (PCI) of calcified lesions may impede stent delivery and optimal expansion, leading to decreased procedural success and higher rates of periprocedural complications in comparison to treatment of atherosclerosis without calcium [1]. In cases of severe calcification, suboptimal stent deployment in particular was linked to both short- and long-term major adverse cardiac events (MACE) [2]. Compared to PCI of non-calcified lesions, more complex and time-consuming procedures are required to modify calcifications, potentially resulting in increased radiation exposure and contrast administration to patients [3]. Consequently, the risks of radiation-induced skin injury and malignancy, as well as contrast-induced nephropathy (CIN), are elevated [4–6]. Notably, a dose–area product (DAP) value of 500 Gy·cm2 during PCI is regarded as a trigger level for patient follow-up to monitor potential skin injuries [7]. Moreover, for every additional 100 ml of contrast medium administered, the CIN risk increases by one point according to Mehran’s scoring system [8]. However, there is a limited number of studies evaluating the impact of calcified lesion modification techniques on both patient radiation exposure and contrast usage [3, 9].
In this single-center study, data pertaining to radiation exposure and contrast utilization from a patient cohort undergoing PCI for calcified lesions were processed and analyzed. Furthermore, several key determinants of MACE were identified.
Material and methods
Study population and data collection
A total of 281 patients who underwent successful PCI for calcified lesions at our catheterization laboratory between March 30, 2018, and April 1, 2025, were enrolled in the study. Procedural success was defined as the attainment of technical goals without any complications during the intervention, such as coronary artery perforation, stent loss, burr entrapment, etc. Procedures were performed by three experienced interventional cardiologists, hereafter referred to as operators A, B, and C. Assistance was provided by several interventional cardiology fellows, with one assigned to each procedure. During the study period, the three operators attempted an additional eight PCIs for calcified lesions – accounting for 2.8% of the total cases – which were ultimately unsuccessful due to the inability to advance the wires. Patient characteristics such as age, sex, weight, and height, along with procedural details – including vascular territory of the lesion, contrast volume used, DAP, fluoroscopic and procedural durations, access route, and the number of balloons and stents deployed per patient – were collected for each procedure. Additional data encompassed the technique employed for calcification modification, operator’s name, duration of hospitalization, and whether a temporary pacemaker (TPS) was implanted or an adjunctive technology for intravascular imaging was used. Furthermore, information regarding the presence or absence of comorbidities such as hypertension (HT), dyslipidemia (DLP), diabetes mellitus (DM), and chronic kidney disease (CKD), as well as family history of coronary artery disease (CAD), smoking history, and serum creatinine levels before and after PCI, was extracted from the patients’ medical records. Procedural duration was defined as the time from arterial puncture to arterial closure, achieved using a hemostatic bandage after sheath removal. MACE was mainly recorded through scripted phone interviews conducted from a fellow cardiologist with patients or their close relatives at 6 months and 1 year following PCI. In certain cases, additional information was obtained from patients’ medical records. MACE was defined as the composite of cardiac death (CD), non-fatal myocardial infarction, target vessel revascularization (TVR), and pulmonary embolism (PE). The proportion of patients experiencing a greater than 25% relative or 0.5 mg/dl absolute increase in serum creatinine within 72 h of contrast exposure was estimated to assess the incidence of contrast-induced nephropathy (CIN). No other significant serum indices for kidney injury prediction were documented.
Equipment and endpoints
A Siemens single-plane (Siemens, Artis Zee, Erlangen, Germany) cardiac angiography unit facilitating real-time measurements of DAP and irradiation time was used for all procedures. Operators A and B carried out PCI under fluoroscopy at 15 frames per second (FPS) and standard collimation practice, while operator C performed fluoroscopy at 7.5 FPS and an increased collimation. Cineangiography was conducted at 15 FPS by all operators [10]. Apart from the primarily used non-compliant (NC) balloons (BROSMED Medical, Dongguan, China) for calcification modification through angioplasty, super-high pressure OPN NC (SIS MEDICAL, Frauenfeld, Switzerland), drug-coated (DC) (from various manufacturers), and cutting balloons (BOSTON SCIENTIFIC, Marlborough, MA, USA) were also deployed. Intravascular lithotripsy (IVL) was conducted using the Shockwave system (Shockwave Medical, Santa Clara, CA, USA), while rotational atherectomy (RA) was conducted using the Rotablator system (Boston Scientific, Marlborough, MA, USA). PCI procedures were categorized as ad-hoc when performed immediately following coronary angiography (CA), and as elective when carried out at a later stage. Patients’ radiation exposure was quantified using DAP and effective dose (ED). The ED (in mSv) was estimated by multiplying DAP (in Gy·cm2) with a conversion factor, i.e., 0.2 for adult males and 0.28 for adult females, as recommended by the European Heart Rhythm Association practical guide [11]. DAP was chosen as the primary endpoint since it provided a direct measurement, while ED was an estimation. Contrast (Scanlux) usage and MACE were also considered primary endpoints. Secondary endpoints included irradiation and procedural times.
Calcification modification techniques and severity assessment
Selection of the most appropriate modification technique was at the sole discretion of the operators, primarily contingent upon the degree of calcium buildup. In that respect, balloon angioplasty was more frequently employed for the management of mild to moderate calcifications, whereas more aggressive modalities, i.e., IVL and RA, were reserved for severe calcium deposits. Details about the employed technique can be found in a Society for Cardiovascular Angiography and Interventions consensus document, which provides an algorithm to help guide use of the various calcium modification strategies [12].
Intravascular imaging was employed in 25.6% of patients. Consequently, lesion calcification severity was assessed angiographically across the entire cohort, despite its lower sensitivity compared to intravascular imaging [12]. Specifically, two experienced cardiologists independently evaluated the angiograms in a blinded manner, categorizing calcifications into two groups: mild/moderate and severe. Mild calcifications were defined by the absence of radiopacities on the angiogram. Moderate calcifications were characterized by radiopacities that are visible only during the cardiac cycle before contrast injection, typically affecting only one side of the vessel lumen. Severe calcifications were identified by radiopacities visible without cardiac motion before contrast injection, usually involving both sides of the vessel lumen [13].
Statistical analysis
The statistical analysis was performed using the SPSS software package (IBM SPSS Statistics, version 25.0; IBM Corp., Armonk, NY, USA). Categorical variables are presented in terms of counts and percentages, and their associated differences were evaluated with the c2 or Fisher’s exact test, depending on whether the expected cell frequencies were high or low, respectively. Continuous variables are described by either mean ± standard deviation (SD) or median with interquartile range (IQR), depending on the normality of their distribution, which was assessed using Shapiro-Wilk test. For normally distributed variables, means between two groups were compared using Student’s t-test. In contrast, when dealing with variables not following normality, differences between two groups were assessed using the (non-parametric) Mann-Whitney U test. A multiple linear regression analysis was conducted using DAP and contrast as the primary outcome variables and investigating various predictors. The natural logarithm of both variables was modeled due to their positively skewed distributions. Additionally, binary logistic regression models were constructed to identify factors affecting the occurrence of MACE and procedures linked to high DAP (≥ 500 Gy·cm2) or low DAP (< 500 Gy·cm2). Predictor variables considered key descriptors of radiation, patient characteristics, disease, or procedure were included in the models if they demonstrated a significant impact (p ≤ 0.05) on the primary outcome in preliminary univariate regressions and exhibited no or weak correlation (Pearson’s correlation coefficient < 0.2 or Cramer’s V coefficient < 0.3) with one another. Dummy coding was used to handle multi-level categorical predictor variables in analyses. A free intercept was employed in the multivariate models. The assumptions of the multilinear models were evaluated in SPSS through various diagnostic tools, including histograms, normal probability-probability plots, scatter plots of the residuals, variance inflation factor values and the Durbin-Watson test. For the binary logistic multivariate models, goodness of fit was assessed with the Hosmer-Lemeshow test, while linearity in the log-odds was evaluated with the Box-Tidwell test. The obtained exponentiated b coefficients represent the relative influence of each variable on primary outcomes. Multiple imputation was employed to address missing values, which were below ~2% across all variables. A propensity score matching (PSM) adjustment was employed in sub-cohort comparisons to mitigate confounding bias. Specifically, propensity scores were generated using logistic regression analysis incorporating key predictor variables as covariates. Nearest neighbor matching was then used to create two equal-sized balanced groups, and the statistical significance of differences between them was assessed. A two-sided p ≤ 0.05 was considered statistically significant in all analyses.
Results
Baseline and procedural characteristics for the entire patient cohort are summarized in Table I. 281 patients (~81% male) were included in the study, with a median age of 71 (64–78) years and a median BMI of 27.44 (24.84–30.42) kg/m2. Median DAP of all PCIs was 262.65 (169.12–407.11) Gy·cm2. Associated median irradiation time was 26.6 (18.5–37.9) min. During a median procedural length of 83 (60–115) min, a volume of 250 (180–335) ml of contrast agent was administered. Approximately 66% of patients presented with HT and DLP, while the prevalence of DM and CKD was ~40% and 13.5%, respectively. 7.5% of patients underwent prior CABG and ~6% of interventions were performed at CTO vessels. CIN following interventions was estimated to affect ~7% of the entire population, with ~18% observed specifically within the CKD subgroup. Nearly 81% of patients underwent elective PCI, while the remainder underwent ad-hoc PCI. Radial (femoral) access was used in ~52% (~41%) of the procedures. Operator A performed the majority (~63%) of interventions, followed by operator B with ~24%, and operator C with 12.5%. On average, 4.6 ±2.1 balloons and 1.9 ±1.2 stents were deployed per patient. More than ~82% of patients were treated with a single PCI targeting a single vessel. Calcified lesions in the LAD artery, whether exclusively or in conjunction with other vessels, were observed in ~59% of patients. Calcifications were primarily modified using NC balloons and RA. Approximately 30% of these were classified as severe, and the rest as mild or moderate. Adjunctive technologies for intravascular imaging were employed in ~26% of interventions.
Table I
Baseline and procedural characteristics for the entire patient dataset (N = 281)
| Parameter | value |
|---|---|
| Baseline and procedural characteristics | |
| Age [years] | 71 (64–78) |
| Male, sex | 227 (80.8%) |
| BMI [kg/m2] | 27.44 (24.84–30.42) |
| DAP [Gy·cm2] | 262.65 (169.12–407.11) |
| ED [mSv] | 58.02 (36.66–84.71) |
| Irradiation time [min] | 26.6 (18.5–37.9) |
| Procedural time [min] | 83 (60–115) |
| Contrast [ml] | 250 (180–335) |
| CKD | 38 (13.5%) |
| DM | 113 (40.2%) |
| HT | 184 (65.5%) |
| DLP | 185 (65.8%) |
| CAD | 90 (32.0%) |
| Smoking | 93 (33.1%) |
| CAD family history | 43 (15.3%) |
| Prior CABG | 21 (7.5%) |
| TPS implantation | 49 (17.4%) |
| CTO | 16 (5.7%) |
| Hospitalization time [days] | 7 (4–10) |
| CIN | 30 (10.9%) |
| PCI type | |
| Ad-hoc | 54 (19.2%) |
| Elective | 227 (80.8%) |
| Access route | |
| Radial | 147 (52.3%) |
| Femoral | 116 (41.3%) |
| Radial + femoral | 16 (5.7%) |
| Other | 2 (0.7%) |
| Operator | |
| A | 178 (63.3%) |
| B | 68 (24.2%) |
| C | 35 (12.5%) |
| Number of balloons | |
| ≤ 4 | 153 (54.4%) |
| > 4 | 128 (45.6%) |
| Number of stents | |
| 0 | 22 (7.8%) |
| 1 | 100 (35.6%) |
| > 1 | 159 (56.6%) |
| Number of PCI | |
| 1 | 235 (83.6%) |
| 2 | 45 (16.0%) |
| 3 | 1 (0.4%) |
| Number of treated vessels | |
| 1 | 232 (82.6%) |
| 2 | 30 (10.7%) |
| 3 | 18 (6.4%) |
| 4 | 1 (0.3%) |
| Artery | |
| LAD | 132 (47.0%)† |
| LCX | 32 (11.4%)† |
| RCA | 66 (23.5%)† |
| LM | 2 (0.7%)† |
| Other | 49 (17.4%)‡ |
| Modified calcification technique | |
| RA | 81 (28.8%)§ |
| IVL | 19 (6.8%)§ |
| Balloon angioplasty | |
| NC | 93 (33.1%)§ |
| OPN NC | 3 (1.1%)§ |
| Cutting | 0 (0%)§ |
| DC | 0 (0%)§ |
| Other | 65 (17.4%)¶ |
| Intravascular imaging | |
| IVUS | 65 (23.1%) |
| OCT | 7 (2.5%) |
| Severity of calcifications | |
| Mild or moderate | 194 (69.0%) |
| Severe | 87 (31.0%) |
BMI – body mass index, CABG – coronary artery bypass graft, CAD – coronary artery disease, CIN – contrast-induced nephropathy, CKD – chronic kidney disease, CTO – chronic total occlusion, DAP – dose–area product, DC – drug-coated, DLP – dyslipidemia, DM – diabetes mellitus, ED – effective dose, HT – hypertension, IVL – intravascular lithotripsy, IVUS – intravascular ultrasound, LAD – left artery descending, LCX – left circumflex, LM – left main, NC – non-compliant, OCT – optical coherence tomography, PCI – percutaneous coronary intervention, RA – rotational atherectomy, RCA – right coronary artery, TPS – temporary pacemaker. *No data available for 5 and 7 patients for 6-month and 1-year MACE, respectively.
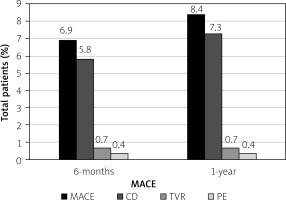
Figure 1 depicts the proportion of MACE within the overall patient population in two distinct time periods following PCI. Specifically, the incidence of MACE was ~7% within 6 months and increased to ~8.5% at 1 year after the procedure. CD was the predominant adverse event, accounting for ~84-87% of all MACE cases. The increase in MACE percentage over time is due to a 1.5% rise in CD, as TVR and PE proportions remained stable.
Figure 1
Proportions of major adverse cardiac events (MACE) at 6 months and 1 year
CD – cardiac death, PE – pulmonary embolism, TVR – target vessel revascularization.
P-values obtained from univariate regressions examining the relationship between the natural logarithm of DAP and contrast with key predictor variables are displayed in Tables II and III, respectively. Also shown are multivariate regression results expressed as fold changes (exponentiated b coefficients) in DAP and contrast, respectively. The corresponding fold changes are presented with 95% confidence intervals (95% CI) and p-values. Multivariate analysis demonstrated that both outcome variables were significantly influenced by operator and irradiation time. Specifically, procedures conducted by operator C (vs. those by A and B) were associated with reductions in DAP by a factor of 0.55 (95% CI: 0.49–0.62; p < 0.001) and in contrast by 0.81 (95% CI: 0.71–0.92; p = 0.001). A 1-minute prolongation of irradiation time resulted in a 1.02-fold increase of both outcomes (95% CI: 1.01–1.03; p < 0.001). Furthermore, male sex was associated with a 1.22-fold rise in DAP (95% CI: 1.10–1.35; p < 0.001). Each unit increase in BMI conferred a 1.03-fold elevation in DAP (95% CI: 1.02–1.04; p < 0.001). Additionally, ad-hoc (vs. elective) interventions were linked to a 1.42-fold rise in DAP (95% CI: 1.19–1.68; p = 0.02). Increasing stent and balloon numbers were associated with ~1.1-fold elevation (95% CI: 1.0–1.2; p ≤ 0.03) in both DAP and contrast use. In univariate analysis, age, CTO procedures and access route were significantly associated with DAP and contrast usage, but in multivariate analysis these relationships were not significant. Similar patterns were observed for the number of balloons, hospitalization, and calcification severity with DAP, and for the number of stents with contrast.
Table II
Results of univariate and multivariate linear regression analysis for dose–area product (1.86 ≤ Durbin-Watson ≤ 1.91; 1 < variance inflation factor ≤ 1.33; adjusted–R2 = 0.73)
| Variable | Analysis | ||||
|---|---|---|---|---|---|
| Univariate | Multivariate* | ||||
| Primary outcome | Predictor | P-value | Fold change | 95% CI | P-value |
| LnDAP | BMI† | < 0.001 | 1.029 | 1.020–1.039 | < 0.001 |
| Sex‡ | < 0.001 | 1.220 | 1.101–1.351 | < 0.001 | |
| Operator§ | < 0.001 | 0.547 | 0.486–0.616 | < 0.001 | |
| Irradiation time† | < 0.001 | 1.022 | 1.020–1.025 | < 0.001 | |
| Number of stents¶ | < 0.001 | 1.110 | 1.023–1.202 | 0.01 | |
| PCI typeǁ | 0.02 | 1.142 | 1.032–1.264 | 0.01 | |
| Number of balloons†† | < 0.001 | 1.075 | 0.988–1.168 | 0.09 | |
| CTO** | < 0.001 | 1.064 | 0.882–1.284 | 0.51 | |
| Age† | 0.01 | 0.998 | 0.994–1.003 | 0.46 | |
| Access route⁑ | 0.02 | 1.052 | 0.968–1.146 | 0.24 | |
| Severity! | 0.02 | 1.047 | 0.963–1.140 | 0.28 | |
| Hospitalization time [days]† | 0.02 | 1.005 | 0.998–1.011 | 0.16 | |
| CAD** | 0.67 | ||||
| Prior CABG** | 0.80 | ||||
| CAD family history** | 0.94 | ||||
| Smoking** | 0.24 | ||||
| DM** | 0.85 | ||||
| CKD** | 0.94 | ||||
| DLP** | 0.77 | ||||
| HT** | 0.66 | ||||
| Intravascular imaging** | 0.32 | ||||
| Technique‼ | 0.14 | ||||
| Artery$ | 0.30 | ||||
| TPS** | 0.96 | ||||
BMI – body mass index, CABG – coronary artery bypass graft, CAD – coronary artery disease, CI – confidence interval, CKD – chronic kidney disease, CTO – chronic total occlusion, DAP – dose–area product, DLP – dyslipidemia, DM – diabetes mellitus, HT – hypertension, IVL – intravascular lithotripsy, LAD – left artery descending, PCI – percutaneous coronary intervention, RA – rotational atherectomy, TPS – temporary pacemaker.
Table III
Results of univariate and multivariate linear regression analysis for contrast administration (Durbin-Watson = 2.21; 1.05 ≤ variance inflation factor ≤ 1.29; adjusted–R2 = 0.47)
| Variable | Analysis | ||||
|---|---|---|---|---|---|
| Univariate | Multivariate* | ||||
| Primary outcome | Predictor | P-value | Fold change | 95% CI | P-value |
| LnContrast | Irradiation time† | < 0.001 | 1.015 | 1.012–1.018 | < 0.001 |
| Operator‡ | < 0.001 | 0.807 | 0.710–0.916 | 0.001 | |
| Number of balloons§ | < 0.001 | 1.102 | 1.008–1.204 | 0.03 | |
| Age† | 0.002 | 0.996 | 0.991–1.000 | 0.07 | |
| CTO¶ | < 0.001 | 1.176 | 0.969–1.428 | 0.10 | |
| Number of stentsǁ | < 0.001 | 1.072 | 0.983–1.170 | 0.11 | |
| Access route†† | 0.03 | 1.055 | 0.968–1.152 | 0.22 | |
| BMI† | 0.57 | ||||
| Sex* | 0.37 | ||||
| PCI type⁑ | 0.07 | ||||
| Hospitalization time [days]† | 0.55 | ||||
| CAD¶ | 0.33 | ||||
| Prior CABG¶ | 0.92 | ||||
| CAD family history¶ | 0.53 | ||||
| Smoking¶ | 0.06 | ||||
| DM¶ | 0.48 | ||||
| CKD¶ | 0.97 | ||||
| DLP¶ | 0.45 | ||||
| HT¶ | 0.38 | ||||
| Intravascular imaging¶ | 0.25 | ||||
| Severity! | 0.10 | ||||
| Technique‼ | 0.76 | ||||
| Artery$ | 0.70 | ||||
| TPS¶ | 0.37 | ||||
BMI – body mass index, CABG – coronary artery bypass graft, CAD – coronary artery disease, CI – confidence interval, CKD – chronic kidney disease, CTO – chronic total occlusion, DLP – dyslipidemia, DM – diabetes mellitus, HT – hypertension, IVL – intravascular lithotripsy, LAD – left artery descending, PCI – percutaneous coronary intervention, RA – rotational atherectomy, TPS – temporary pacemaker.
Table IV details intergroup differences in radiation exposure, procedural time, and contrast, stratified by operator (A and B vs. C), before and after PSM. Pre-adjustment significant differences in baseline and procedural characteristics between operator subgroups were limited to irradiation time and hospitalization days (p = 0.03 for both). After adjustment, these differences were no longer statistically significant (p ≥ 0.83 overall). Compared to operator C, all unadjusted parameters were found to be significantly higher in the operator A and B subgroup (p ≤ 0.03), except for procedural time, where no significant difference (p = 0.87) was observed. Specifically, when using the medians of operators A and B as reference values, DAP, patient ED, irradiation time, and contrast were 53.2%, 55.6%, 18.0%, and 28.0% higher, respectively. After adjustment, DAP and ED percentage differences remained statistically significant (p < 0.001), although they decreased by 11.5% and 13.2%, respectively. However, contrast usage and irradiation and procedural times were no longer statistically different between the two subgroups (p ≥ 0.07).
Table IV
Median values (with IQR) of dose–area product, patient effective dose, irradiation and procedural time, and contrast for two operator groups (A and B vs. C): A – before PSM; B – after PSM
| A | ||||
|---|---|---|---|---|
| Before PSM | Operator | |||
| Metric | A and B (N = 246)* | C (N = 35)* | P-value† | PD‡ |
| DAP [Gy·cm2] | 281.46 (196.37–433.76) | 131.63 (85.63–179.47) | < 0.001 | 53.2 |
| ED [mSv] | 61.10 (42.81–90.10) | 27.12 (17.21–36.01) | < 0.001 | 55.6 |
| Irradiation time [min] | 27.2 (18.6–39.2) | 22.3 (14.9–31.2) | 0.031 | 18.01 |
| Procedural time [min] | 83 (60–115) | 75 (60–120) | 0.865 | 9.6 |
| Contrast [ml] | 250 (180–347.5) | 180 (160–240) | < 0.001 | 28.0 |
| B | ||||
|---|---|---|---|---|
| After PSM | Operator | |||
| Metric | A and B (N = 30)* | C (N = 30)* | P-value† | PD‡ |
| DAP [Gy·cm2] | 222.65 (152.49–310.04) | 129.75 (84.58–161.29) | < 0.001 | 41.7 |
| ED [mSv] | 46.38 (36.34–62.34) | 26.69 (16.92–32.26) | < 0.001 | 42.4 |
| Irradiation time [min] | 21.0 (18.0–33.1) | 22.3 (14.4–36.4) | 0.994 | 6.2 |
| Procedural time [min] | 77.5 (60–106.2) | 75 (60–120.5) | 0.865 | 3.2 |
| Contrast [ml] | 200 (180–257.5) | 175 (157.5–225) | 0.068 | 12.5 |
P-values derived from univariate binary logistic regression analyses evaluating the association between high DAP and MACE, with various key predictor variables, are presented in Tables IV and V, respectively. Also shown are multivariate odds ratios (OR) for predictor variables with p ≤ 0.05 in univariate analysis, along with their 95% confidence intervals and p-values. Univariate analysis demonstrated significant associations between high DAP (3 500 Gy·cm2) and CTO, number of PCI, artery, BMI, and age (Table IV). Apart from the latter, all other variables remained significant predictors of high DAP in the subsequent multivariate analysis. Specifically, the likelihood of procedures with a high DAP was markedly elevated in cases involving CTO interventions, with an OR of 14.61 (95% CI: 4.21–50.60; p < 0.001). Additionally, an increase in the number of PCI was significantly associated with a high DAP (OR = 12.47; 95% CI: 5.35–29.06; p < 0.001). Procedures performed not solely on the LAD showed an elevated risk, with an OR of 2.88 (95% CI: 1.29–6.41; p = 0.01). Furthermore, each one-unit increase in BMI was associated with a 14% increase in the odds of high DAP (OR = 1.14; 95% CI: 1.05–1.25; p = 0.002).
Table V
Results of univariate and multivariate binary logistic regression analysis for dose–area product (DAP < 500 Gy·cm2: 0; DAP ≥ 500 Gy·cm2: 1). (Hosmer-Lemeshow ≥ 0.14; Box-Tidwell: p ≥ 0.109).
| Variable | Analysis | ||||
|---|---|---|---|---|---|
| Univariate | Multivariate* | ||||
| Primary outcome | Predictor | P-value | OD | 95% CI | P-value |
| DAP | |||||
| CTO† | < 0.001 | 14.61 | 4.21–50.60 | < 0.001 | |
| Number of PCIs‡ | < 0.001 | 12.47 | 5.35–29.06 | < 0.001 | |
| Artery§ | 0.01 | 2.88 | 1.29–6.41 | 0.01 | |
| BMI¶ | 0.03 | 1.14 | 1.05–1.25 | 0.002 | |
| Age¶ | 0.05 | 0.99 | 0.95–1.03 | 0.61 | |
| Sexǁ | 0.16 | ||||
| Operator†† | 1.00 | ||||
| Number of stents‡ | 0.18 | ||||
| PCI type** | 0.14 | ||||
| Access route⁑ | 0.07 | ||||
| Hospitalization days¶ | 0.83 | ||||
| CAD† | 0.28 | ||||
| Prior CABG† | 0.86 | ||||
| CAD family history† | 0.56 | ||||
| Smoking† | 0.23 | ||||
| DM† | 0.57 | ||||
| CKD† | 0.65 | ||||
| DLP† | 0.48 | ||||
| HT† | 0.33 | ||||
| Intravascular imaging† | 0.31 | ||||
| Severity! | 0.06 | ||||
| Technique‼ | 0.19 | ||||
| TPS† | 0.77 | ||||
BMI – body mass index, CABG – coronary artery bypass graft, CAD – coronary artery disease, CI – confidence interval, CKD – chronic kidney disease, CTO – chronic total occlusion, DAP – dose–area product, DLP – dyslipidemia, DM – diabetes mellitus, HT – hypertension, IVL – intravascular lithotripsy, LAD – left artery descending, OD – odds ratio, PCI – percutaneous coronary intervention, RA – rotational atherectomy, TPS – temporary pacemaker.
Regarding MACE, univariate analysis identified significant associations with CKD and age (Table VI). 6-month and 1-year MACE were also significantly associated with smoking history and calcification severity, respectively. All other predictor variables were excluded from the subsequent multivariate analysis, which identified CKD as a significant predictor of MACE for both periods, along with severity for the prolonged one. Specifically, the risk of MACE increased in the presence of CKD, with odds ratios of 4.40 (95% CI: 1.47–13.15; p = 0.008) at 6 months and 5.65 (95% CI: 2.14–14.88; p < 0.001) at 1 year. The presence of severe calcifications, as opposed to mild or moderate, was associated with higher odds of 1-year MACE (OR = 2.55; 95% CI: 1.01–6.43; p = 0.046). Note that age was not included in the 1-year MACE model due to a violation of the linearity assumption.
Table VI
Results of univariate and multivariate binary logistic regression analysis for 6-month and 1-year MACE (for 6 months: Hosmer-Lemeshow = 0.57; Box-Tidwell: p = 0.816; for 1 year: Hosmer-Lemeshow = 0.63; Box-Tidwell: p = 0.042)
| Variable | Analysis | ||||
|---|---|---|---|---|---|
| Univariate | Multivariate* | ||||
| Primary outcome | Predictor | P-value | OR | 95% CI | P-value |
| 6-month MACE | CKD† | < 0.001 | 5.887 | 2.085–16.617 | < 0.001 |
| Smoking† | 0.03 | 0.146 | 0.018–1.169 | 0.07 | |
| Age‡ | 0.003 | 1.060 | 0.995–1.129 | 0.07 | |
| DAP‡ | 0.25 | ||||
| Irradiation time‡ | 0.47 | ||||
| Procedural time‡ | 0.30 | ||||
| Contrast‡ | 0.09 | ||||
| BMI‡ | 0.65 | ||||
| Sex§ | 0.39 | ||||
| Operator¶ | 0.59 | ||||
| PCI typeǁ | 0.16 | ||||
| Number of stents†† | 0.95 | ||||
| Access route** | 0.60 | ||||
| Number of balloons⁑ | 0.23 | ||||
| Number of PCI†† | 0.98 | ||||
| CAD† | 0.36 | ||||
| Prior CABG† | 0.69 | ||||
| CAD family history† | 0.24 | ||||
| DM† | 0.51 | ||||
| DLP† | 0.47 | ||||
| HT† | 0.76 | ||||
| Intravascular imaging† | 0.32 | ||||
| Severity! | 0.11 | ||||
| Technique | 0.20/0.86? | ||||
| Artery$ | 0.72 | ||||
| TPS† | 0.40 | ||||
| 1-year MACE | CKD† | < 0.001 | 7.222 | 2.801–18.620 | < 0.001 |
| Severity! | 0.047 | 2.554 | 1.015–6.426 | 0.046 | |
| Age‡,?? | 0.01 | ||||
| Smoking† | 0.051 | ||||
| DAP‡ | 0.39 | ||||
| Irradiation time‡ | 0.93 | ||||
| Procedural time‡ | 0.11 | ||||
| Contrast‡ | 0.12 | ||||
| BMI‡ | 0.20 | ||||
| Sex§ | 0.09 | ||||
| Operator¶ | 0.71 | ||||
| PCI typeǁ | 0.33 | ||||
| Number of stents†† | 0.86 | ||||
| Access route** | 0.19 | ||||
| Number of balloons⁑ | 0.16 | ||||
| Number of PCI†† | 0.35 | ||||
| CAD† | 0.40 | ||||
| Prior CABG† | 0.57 | ||||
| CAD family history† | 0.86 | ||||
| DM† | 0.16 | ||||
| DLP† | 0.77 | ||||
| HT† | 0.89 | ||||
| Technique | 0.37/0.83? | ||||
| Artery$ | 0.62 | ||||
| TPS† | 0.97 | ||||
BMI – body mass index, CABG – coronary artery bypass graft, CAD – coronary artery disease, CI – confidence interval, CKD – chronic kidney disease, CTO – chronic total occlusion, DAP – dose–area product, DLP – dyslipidemia, DM – diabetes mellitus, HT – hypertension, IVL – intravascular lithotripsy, LAD – left artery descending, MACE – major adverse cardiovascular effects, OD – odds ratio, PCI – percutaneous coronary intervention, RA – rotational atherectomy, TPS – temporary pacemaker.
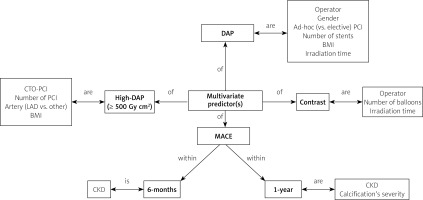
For enhanced visual clarity, the multivariate predictors of DAP, contrast, and MACE are schematically summarized in Figure 2.
Discussion
The present study demonstrated that the patients’ radiation exposure (in terms of DAP) during PCI of calcified lesions was predominantly influenced by operator’s practice. By employing a reduction in fluoroscopic frame rate from 15 to 7.5 FPS coupled with increased collimation [10], operator C carried out procedures associated with a lower median DAP of ~42% compared to that of procedures performed by the other two operators (129.75 Gy·cm2 vs. 222.65 Gy·cm2; p < 0.001) – an achievement accomplished without significantly affecting median irradiation or procedural time (p ≥ 0.87). This level of reduction, which may not significantly alter the overall radiation risk for an individual patient during a single procedure, becomes pronounced when considering the cumulative effect over multiple procedures, potentially playing a key role in a comprehensive risk mitigation strategy. Although switching to lower FPS may lead to safety issues due to an inevitable decline in image quality, this was not observed in our study. Evidently, all PCIs performed using this dose-saving feature were successful, underscoring operator C’s ability to visualize all critical aspects of the coronary anatomy required for proper execution of stages that do not demand the highest image quality, such as balloon dilatation and stent expansion. In terms of long-term post-procedural effects, multivariate analysis showed that MACE was not associated with operator (Table VI). Although procedures performed by operator C were associated with an almost twofold proportion of 1 year MACE compared to those conducted by operators A and B, this difference did not reach statistical significance (14.3% vs. 7.3%; p = 0.159). Notably, the median DAP of all PCIs included in the present study (262.65 Gy·cm2) far exceeds the European diagnostic reference level (85 Gy·cm2) [14]. A key step in reducing this discrepancy is for all operators to consistently adopt operator C’s dose-saving practice. Moreover, radiation exposure is also affected by operators’ radiation protection awareness, as well as their willingness to minimize radiation. Hence, focus should be paid to spreading awareness and encouraging the adoption of effective yet simple dose-saving techniques among all operators. Formal training programs can play a pivotal role in achieving this goal [15].
Male sex and increasing BMI were also significant predictors of elevated DAP, consistent with findings from previous studies [16]. Notably, male sex was associated with a higher DAP than female sex (males: 285.08 (182.65–431.34) Gy·cm2; females: 217.16 (133.64–288.20) Gy·cm2; p = 0.001), even though the two subgroups did not have a significantly different median BMI (males: 27.49 (24.91–30.48) kg/m2; females: 27.34 (24.39–29.40) kg/m2; p = 0.57). A potential reason for this sex-based difference could be weight, as males were heavier than females (males: 81 (75–90) kg; females: 70 (60–79) kg; p < 0.001). Indeed, larger patients require higher doses due to the automatic exposure control feature, which amplifies tube voltage and current to uphold optimal radiation levels at the image receptor, compensating for the increased amount of tissue traversed by X-rays. Unlike weight, which cannot be easily modified, reducing the amount of patients’ BMI exposed through the use of non-lead shielding materials has the potential to reduce DAP [16]. Unfortunately, the development of such materials remains a challenging task, necessitating further research efforts. Multivariate analysis also confirmed a significant association of DAP with PCI type, number of stents, and irradiation time. Specifically, ad-hoc (vs. elective) PCIs were associated with increased DAP, consistent with previous studies [9]. Compared to elective procedures, which include only the intervention component, ad-hoc interventions also encompass the diagnostic component, during which cardiologists primarily utilize cineangiography-dominated projections to assess the lesion, resulting in a significant increase in radiation exposure.
Multivariate binary logistic regression analysis identified CTO procedures, number of PCI, targeted artery, and BMI as significant predictors of high DAP (≥ 500 Gy·cm2). Note that there was no association between the operator (A and B vs. C) and DAP levels, as all procedures performed by C resulted in a DAP ≤ 485.51 Gy·cm2. Additionally, the observed association between PCI not exclusively targeting the LAD and increased high-DAP risk was primarily driven by the inclusion of the 49 multi-artery procedures within this subgroup. Nonetheless, the number of treated arteries per patient was excluded from the analysis due to its correlation with the targeted artery variable (Cramer’s V = 0.44). Analyzing data from a multicenter US registry, Christakopoulos et al. [17] found that excess patient radiation dose was related to high DAP and the presence of moderate or severe calcifications. One third of their patient cohort received a high air kerma dose (> 5 Gy), suggesting that the risk of skin injury persists despite the use of modern procedural techniques at experienced centers. Currently, our catheterization laboratory lacks a follow-up protocol for post-PCI radiation skin damage. Although no cases have been reported to date, the aforementioned findings suggest the need to reconsider our approach, especially for high BMI patients with calcified CTO lesions. When treating such patients, radiation management strategies can be incorporated into the planning process to reduce radiation exposure while ensuring the effectiveness of the intervention. For example, ad-hoc CTO PCI should be avoided to prevent dose elevation from the diagnostic component, except in rare cases [17]. Unlike DAP findings, the PSM analysis showed no significant difference in contrast use between operators (A and B vs. C), indicating consistent practice among them. However, more contrast was administered to patients as irradiation time and number of balloons increased according to our multivariate analysis. Although not a direct cause–effect relationship, operators should be aware that the decreased use of the radiation activation pedal may potentially lead to lower contrast volumes. Additionally, using fewer balloons reduces the need for contrast imaging to verify their proper placement and full deployment. A strategy that focuses on limiting balloon usage will often involve a more streamlined and efficient approach, which can reduce the number of steps requiring contrast injections. For example, provisional stenting for bifurcation lesions [18], which is often the default approach, limits the need for a second stent and associated balloon inflations.
Previous studies demonstrated increased CIN risk in elderly patients and those with CKD, HT, and/or DM [6], emphasizing the need to administer a reduced amount of contrast to them. However, our multivariate analysis did not identify CKD, HT, DM, or age as significant predictors of contrast volume, likely due to the urgency of certain procedures and the operator’s overall effort to minimize contrast use across all patients, regardless of age or comorbidities. Notably, in patients with advanced CKD on hemodialysis, no cases of CIN were observed, as their routine dialysis schedules were adjusted in accordance with the planned PCI. Given their cardioprotective role [19], sodium-glucose cotransporter-2 inhibitors (SGLT2I) were administered during the pre- and post-PCI period in ~7.5% of the total cohort, all of whom had high cardiovascular risk, DM, and serum creatinine levels below ~3 mg/dl. Interestingly, a recent meta-analysis demonstrated that SGLT2I may also confer renoprotective effects in diabetic patients undergoing PCI, reducing the risk of developing CIN by up to 63% compared to patients not using SGLT2I [20]. However, a randomized trial yielded conflicting results, indicating no nephroprotective benefits [21]. Clearly, further research is necessary to clarify the debate surrounding the effectiveness of SGLT2I in preventing CIN within this patient population. The present study corroborates prior research indicating that CKD significantly elevates the risk of post-PCI MACE [22]. Notably, in our cohort, ~41-42% of MACE patients had CKD; all of them experienced cardiac death. Most experts agree that CKD affects the cardiovascular system via multiple concurrent pathophysiological mechanisms. In particular, CKD increases oxidative stress, systemic inflammation, and/or endothelial dysfunction, which accelerate atherosclerosis and potentially raise the MACE rate [23]. Given the elevated risk linked to CKD in the context of calcified lesions, thorough evaluation and personalized treatment plans are key to reducing MACE. Existing evidence indicates that further research is needed to assess whether more aggressive management of modifiable CAD risk factors – such as blood pressure, lipids, and glycemic control – beyond current guideline targets can lower MACE incidence in the CKD population [23]. Improved clinical outcomes in this population may be achieved by using IVUS or OCT instead of solely angiographic guidance during interventions [24]. Furthermore, PCI with paclitaxel-eluting stents was associated with a higher 36-month MACE rate in non-hemodialysis CKD patients compared to everolimus-eluting stents, while no significant difference was observed in hemodialysis patients [25].
The current study also demonstrated that the presence of severe, as opposed to mild or moderate, calcifications further amplifies 1-year MACE risk, consistent with previous studies [26]. Selecting the optimal modification technique for calcification is a critical consideration, contingent upon the extent and location of calcification, patient risk factors, and operator expertise. However, in our multivariate model, the modification technique, particularly RA versus others, was not a significant predictor of increased MACE risk (p ≥ 0.20), consistent with a recent meta-analysis showing comparable efficacy between IVL and RA [27]. Furthermore, another recent meta-analysis revealed that combining RA with a cutting or scoring balloon for moderate to severe calcified lesions results in significantly lower MACE compared to RA followed by a plain balloon [28]. Interestingly, patients with non-dilatable lesions after RA exhibited a lower risk of periprocedural myocardial infarction compared to those with balloon-uncrossable lesions, likely due to reduced calcium debris production [29].
Besides limitations stemming from its retrospective and single-center design, the present study has additional constraints. Potential confounders that may have influenced DAP were not captured or adjusted for, such as patient-to-detector distance and projection angles. Also, the severity of the calcified lesions was not assessed through the most sensitive method, i.e. intravascular imaging, which was employed in less than 26% of the study population. Nonetheless, it was included in the analysis following angiographic assessment, despite the method’s limited sensitivity. Moreover, collecting MACE data through telephone interviews with patients or their close relatives may potentially introduce information bias. Lastly, ED was not directly measured or estimated through accurate Monte Carlo simulations, but rather estimated from the DAP recorded by the equipment.
Conclusions
In this study involving patients who underwent PCI on calcified lesions, multivariate analysis identified operator and irradiation time as significant predictors of increased DAP and contrast use. Following PSM adjustment for confounding variables, contrast use was no longer independently associated with operator. Male sex, ad-hoc (vs. elective) procedures, as well as increasing BMI and number of stents were associated with elevated DAP. Additionally, higher contrast use was observed by raising the number of deployed balloons. Notably, CTO interventions, increasing BMI, and number of performed PCIs emerged as significant predictors of high DAP (≥ 500 Gy·cm2). MACE risk increased with the presence of CKD; severe (vs. mild or moderate) calcifications further amplified it within 1 year.