Summary
Risk stratification after transcatheter aortic valve implantation (TAVI) remains challenging because conventional surgical risk scores do not fully capture frailty, nutritional status, and systemic inflammation. In this single-center cohort of 785 patients, a lower preprocedural hemoglobin-albumin-lymphocyte-platelet (HALP) score was independently associated with increased long-term all-cause mortality after TAVI. A HALP cut-off value of 36.82 effectively discriminated long-term survival. These findings suggest that the HALP score may provide complementary prognostic information and help refine preprocedural risk assessment in patients undergoing TAVI.
Introduction
Aortic stenosis (AS) is the most common valvular heart disease in the elderly and remains a major cause of morbidity and mortality if left untreated [1, 2]. Transcatheter aortic valve implantation (TAVI) has revolutionized the management of severe AS, providing a minimally invasive and effective alternative to surgical valve replacement, particularly in the elderly or high surgical risk patients [3, 4]. More recently, its indication for use has been expanded to include selected moderate- and low-risk populations following multidisciplinary Heart Team evaluation [5, 6]. Despite procedural refinements, post-TAVI mortality rates still range between 5% and 24%, underscoring the need for improved risk stratification [7–10].
Conventional surgical risk scores, for example the EuroSCORE II and the Society of Thoracic Surgeons (STS) score were primarily designed to estimate short-term mortality after surgical valve replacement [11, 12]. However, these tools fail to fully reflect the multifactorial nature of outcomes in elderly TAVI patients who often present with multiple comorbidities, frailty, and nutritional deficiencies [13, 14]. Hence, novel biomarkers integrating systemic inflammation and nutritional status may offer complementary prognostic value beyond traditional surgical scores.
AS shares key pathophysiological mechanisms with atherosclerosis, including endothelial dysfunction, lipid deposition, calcification, and chronic inflammation [15–17]. Various inflammatory indices, such as C-reactive protein (CRP) and systemic immune-inflammation index (SII), have been associated with adverse outcomes after TAVI [18, 19]. Likewise, malnutrition and low serum albumin levels – reflecting both poor nutritional and inflammatory status – are established predictors of cardiovascular morbidity and mortality [20, 21].
As a composite biomarker, the HALP score reflects the interaction between nutritional status, inflammatory activity, and underlying hematologic parameters. This easily obtainable index has demonstrated prognostic utility in various cardiovascular and oncologic settings [22–24]; however, its value in predicting long-term mortality following TAVI remains unexplored.
Aim
Therefore, we aimed to evaluate the prognostic significance of preprocedural hemoglobin–albumin–lymphocyte–platelet (HALP) levels for long-term mortality in a large single-center TAVI cohort, and to investigate whether HALP-based risk stratification may offer complementary prognostic information beyond that provided by conventional risk models.
Material and methods
Study design and population
This investigation was designed as a retrospective cohort study conducted at a high-volume tertiary center with extensive experience in structural heart interventions. All patients who underwent TAVI for symptomatic severe aortic stenosis between January 2018 and January 2024 were screened. Procedural candidacy for TAVI was confirmed after multidisciplinary Heart Team review, consistent with contemporary international recommendations [2]. Severe aortic stenosis was diagnosed using standard echocardiographic thresholds, including an aortic valve area < 1.0 cm2, a mean gradient ≥ 40 mm Hg, or a peak transvalvular velocity ≥ 4.0 m/s.
Patients were excluded if they had missing preprocedural laboratory data necessary for HALP score calculation (hemoglobin, albumin, lymphocyte, or platelet count), died during the index hospitalization, had hematologic disorders or malignancies affecting blood counts, acute infection, chronic inflammatory disease, or had incomplete follow-up data. Acute infection was defined as the presence of a documented clinical diagnosis requiring antibiotic therapy at the time of admission, while chronic inflammatory disease was defined as a previously established inflammatory or autoimmune condition under regular medical treatment.
After applying all exclusion criteria, 785 individuals formed the cohort used for the statistical analyses.
The study received approval from the institutional ethics committee and was carried out in accordance with the Declaration of Helsinki. Because the investigation relied exclusively on retrospectively collected data, the need for written informed consent was waived by the committee.
HALP score calculation
Hemoglobin, albumin, lymphocyte, and platelet values and counts were retrieved from venous blood samples collected within 48–72 h preceding the TAVI procedure. A single preprocedural blood sample was used for HALP calculation, and no averaging of multiple measurements was performed. All analyses were performed in the institution’s central laboratory using standardized automated systems.
The HALP score was derived according to the established formula [22, 23]: HALP = (Hemoglobin [g/l] × Albumin [g/l] × Lymphocyte count [109/l])/Platelet count [109/l].
Clinical, echocardiographic, and laboratory data
Demographic characteristics, cardiovascular comorbidities, symptoms, and prior medical history were extracted from the institutional electronic medical records. Surgical risk was quantified using the STS-PROM and EuroSCORE II calculators [12].
Comprehensive transthoracic echocardiography was performed before TAVI according to current guidelines [25, 26]. Measurements included left ventricular ejection fraction (via the biplane Simpson method), chamber dimensions, right ventricular systolic function assesed by tricuspid annular plane systolic excursion (TAPSE), and comprehensive valvular assessment.
Laboratory parameters included complete blood count, renal and liver function tests, C-reactive protein (CRP), and cardiac biomarkers (BNP or NT-proBNP, high-sensitivity troponin). An estimated glomerular filtration rate (eGFR) value < 60 ml/min/1.73 m2, calculated via the CKD-EPI method, was used as the criterion for diagnosing chronic kidney disease.
TAVI procedure
TAVI procedures were carried out in a dedicated catheterization suite by operators with substantial procedural experience. Access strategy and prosthesis selection were individualized based on anatomical findings and Heart Team recommendations. Transfemoral access was preferred as the first-line approach, while subclavian or axillary access or other alternative routes were used in patients for whom transfemoral access was not feasible. According to institutional practice, procedures were performed under either general anesthesia or conscious sedation. Balloon predilatation, postdilatation, and rapid ventricular pacing were applied at the operator’s discretion when clinically indicated.
Clinical endpoints
The primary endpoint of the study was long-term all-cause mortality, defined as death occurring more than 30 days after the index TAVI procedure. The inability to determine specific causes of death limited our ability to evaluate the secondary endpoints. Follow-up data were obtained from hospital electronic medical records and the national death notification system, with a median follow-up duration of 21.8 months.
Statistical analysis
All statistical procedures were conducted using Jamovi software (version 2.6.2; The Jamovi Project, Sydney, Australia). The distribution of continuous variables was examined with the Shapiro–Wilk test, and these variables were summarized either as mean with standard deviation or as median with interquartile range, depending on normality. Categorical variables were reported as frequencies and percentages. Comparisons between patients who survived and those who did not were performed using the independent samples t-test or the Mann–Whitney U test for continuous measures, while Pearson’s c2 or Fisher’s exact test was applied for categorical data.
Variables associated with long-term mortality were initially evaluated using univariable Cox proportional hazards models. Clinically meaningful variables were subsequently included in multivariable Cox regression to identify independent predictors. Hazard ratios (HRs) with 95% confidence intervals (CIs) were reported.
The optimal HALP cut-off value for mortality prediction was established using the maximally selected rank statistics method. Kaplan–Meier survival curves were generated for groups stratified by this cut-off, and differences were assessed with the log-rank test.
To address potential survivor bias related to the exclusion of in-hospital deaths, a sensitivity analysis was performed by including patients who died during the index hospitalization. In this analysis, in-hospital mortality was classified as an event, and the association between the preprocedural HALP score and overall all-cause mortality was evaluated using a multivariable Cox proportional hazards model.
Additionally, risk scores were calculated using the coefficients derived from the Cox proportional hazards model. These scores represent the predicted probability of the event occurring based on the combined effect of all variables included in the model, with higher scores indicating a greater predicted risk. Patients were stratified into four equal-sized groups according to these risk scores: low risk (< 25th percentile), Intermediate-low risk (25th–50th percentile), intermediate-high risk (50th–75th percentile), and high risk (>75th percentile). A p-value of < 0.05 was considered statistically significant for all analyses.
Results
A total of 873 patients undergoing TAVI were screened. Seventy-two patients who experienced in-hospital mortality and 16 patients with missing hematologic parameters were excluded. The remaining 785 patients (median age: 78 [74–82] years, 46% male) were included in the long-term outcome analysis. During a median follow-up of 21.8 (14.2–33.1) months, 62 (8%) patients died and were classified as the non-survivor group. When in-hospital and post-discharge deaths were considered together, the overall mortality of the initially screened cohort was 15.3%.
Compared with survivors, non-survivors were significantly older (80.5 [76.2–84.7] vs. 78 [747–82] years, p = 0.005) and had a higher prevalence of congestive heart failure (32% vs. 19%, p = 0.013), atrial fibrillation (AF) (45% vs. 28%, p = 0.004), and chronic kidney disease (CKD) (37% vs. 17%, p < 0.001). Other clinical and echocardiographic characteristics are presented in Table I.
Table I
Baseline clinical and echocardiographic characteristics according to mortality status
[i] Data are presented as mean ± SD or median (IQR) for continuous variables and as n (%) for categorical variables. Values with a p-value < 0.05 indicate statistical significance and are shown in bold. AF – atrial fibrillation, AKI – acute kidney injury, AR – aortic regurgitation, AVA – aortic valve area, CAD – coronary artery disease, CHF – congestive heart failure, CKD – chronic kidney disease, COPD – chronic obstructive pulmonary disease, CVA – cerebrovascular accident, DM – diabetes mellitus, HT – hypertension, LFLG – low-flow low-gradient, LVEDD – left ventricular end-diastolic diameter, LVEF – left ventricular ejection fraction, LVESD – left ventricular end-systolic diameter, MR – mitral regurgitation, PASP – pulmonary artery systolic pressure, PVL – paravalvular leak, TAPSE – tricuspid annular plane systolic excursion, TR – tricuspid regurgitation, AS – aortic stenosis, LVOT – left ventricular outflow tract, LVOT-VTI – left ventricular outflow tract - velocity time integral.
In terms of laboratory findings, the non-survivor group exhibited a significantly lower HALP score (p < 0.001). In addition, CRP and BNP levels were markedly higher in this group (p < 0.001). Laboratory profiles are summarized in Table II.
Table II
Baseline laboratory characteristics according to mortality status
[i] Data are presented as mean ± SD or median (IQR) for continuous variables and as n (%) for categorical variables. Values with a p-value < 0.05 indicate statistical significance and are shown in bold. Alb – albumin, BNP – B-type natriuretic peptide, CRP – C-reactive protein, HALP – hemoglobin, albumin, lymphocyte, platelet, Hgb – hemoglobin, PLT – platelet count, T Bil – total bilirubin, WBC – white blood cell count. Lymph – lymphocyte count, Neut – neutrophil count, AST – aspartate aminotransfarase, ALT – alanine aminotransfarase. Note: For HALP calculation, hemoglobin and albumin values were converted from g/dl to g/l (×10).
Procedural parameters, including implanted valve size and post-procedural transvalvular gradients, were comparable between the groups. Postoperative acute kidney injury (AKI) was significantly more frequent among non-survivors (18% vs. 5%, p < 0.001), while the paravalvular leak (PVL) showed a borderline association with mortality (p = 0.036) (Table III). On univariable Cox regression analysis, variables associated with long-term mortality included CKD, lower left ventricular ejection fraction (LVEF), reduced LVOT-VTI, and lower HALP score.
Table III
Periprocedural findings of the study population according to mortality status
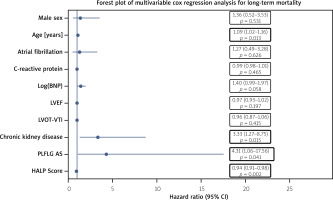
In the multivariable Cox regression model adjusted for relevant clinical and echocardiographic covariates, independent predictors of long-term all-cause mortality were identified. Chronic kidney disease (CKD) was associated with a significantly increased risk of death (HR = 3.33, 95% CI: 1.27–8.75; p = 0.015), and the presence of paradoxical low-flow, low-gradient aortic stenosis (PLFLG AS) was also independently associated with long-term mortality (HR = 4.31, 95% CI: 1.06–17.56; p = 0.041).
Notably, a lower preprocedural HALP score emerged as a strong and independent adverse prognostic factor for long-term mortality (HR = 0.94, 95% CI: 0.91–0.98; p = 0.002). These findings are summarized in Table IV and visually represented in Figure 1, which shows the forest plot of the multivariable Cox regression model for long-term mortality.
Table IV
Multivariable Cox regression analysis for predictors of long-term mortality (survivors) and overall mortality (including in-hospital deaths)
[i] AF – atrial fibrillation; BNP – B-type natriuretic peptide; CKD – chronic kidney disease; LVEF – left ventricular ejection fraction; LVOT-VTI – left ventricular outflow tract velocity time integral; PLFLG AS – paradoxical low-flow, low-gradient aortic stenosis. Note: BNP was entered into the models after natural logarithmic transformation [ln(BNP)].
Figure 1
Multivariable Cox proportional showing independent predictors of long-term mortality after transcatheter aortic valve implantation (TAVI). Hazard ratios (HRs) are presented with 95% confidence intervals (CIs)
To address potential survivor bias, a sensitivity analysis including patients who experienced in-hospital mortality as events was performed. In this overall mortality analysis, the HALP score remained independently associated with all-cause mortality (HR = 0.98, 95% CI: 0.96–1.00; p = 0.015) (Table IV).
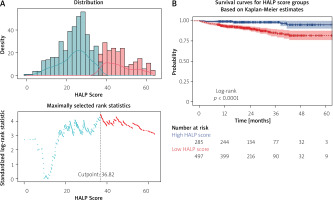
By applying the maximally selected rank statistics method, the HALP score value that most effectively stratified all-cause mortality risk was identified as 36.82. When patients were stratified according to this threshold, those with a low HALP score showed a significantly reduced 60-month survival compared with patients with a high HALP score (log-rank test, p < 0.0001) (Figure 2).
Figure 2
A – Distribution of hemoglobin-albumin-lymphocyte-platelet (HALP) scores and identification of the optimal cut-off value using maximally selected rank statistics. The dashed vertical line indicates the optimal HALP cut-off value of 36.82. B – Kaplan-Meier survival curves according to HALP score groups stratified by the cut-off value of 36.82. Patients with a low HALP score showed significantly reduced long-term survival compared with those with a high HALP score (log-rank p < 0.0001). Numbers at risk are presented below the plot
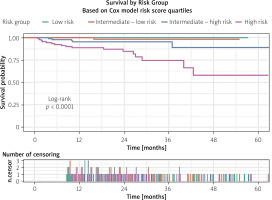
Furthermore, risk scores were calculated based on the coefficients obtained from the Cox proportional hazards model, allowing patients to be categorized into four distinct risk groups: low risk, intermediate-low risk, intermediate-high risk, and high risk. Survival analysis demonstrated clear and statistically significant differences among these groups (log-rank < 0.0001). The discriminatory performance of the model was strong, with a Harrell’s C-index of 0.868, where 0.5 indicates no discrimination and 1.0 represents perfect separation between risk strata (Figure 3).
Figure 3
Survival curves according to quartiles of the Cox risk score. Patients were categorized into low, intermediate-low, intermediate-high, and high-risk groups. Increasing risk score quartiles were associated with progressively reduced long-term survival (log-rank p < 0.0001). The lower panel depicts the number of censoring events across follow-up
Discussion
In this large, single-center TAVI cohort, we demonstrated that lower preprocedural HALP levels –reflecting nutritional and inflammatory status – were closely linked to long-term all-cause mortality. After adjustment for conventional clinical and echocardiographic parameters, the HALP score remained a robust predictor of long-term mortality. To the best of our knowledge, this is the first investigation to assess the prognostic role of HALP in patients undergoing TAVI, and our findings indicate that this simple, low-cost index may offer complementary prognostic information beyond conventional surgical risk scores. It should be acknowledged that the reported long-term mortality rate reflects a survivor cohort, since patients who died during the index hospitalization were excluded from the long-term analysis. When in-hospital and post-discharge deaths were considered together, the overall mortality of the initially screened cohort was 15.3%. Accordingly, the relatively low long-term mortality rate should be interpreted in this context.
The relationship observed in our study between HALP and long-term outcomes aligns with previous evidence highlighting the prognostic relevance of systemic inflammation and nutritional status in cardiovascular disease [23, 24, 27–30]. TAVI candidates are generally elderly, often with multiple comorbidities and varying degrees of frailty. However, traditional risk models – such as EuroSCORE II and STS-PROM – fail to adequately capture the clinical and biological complexity of this population [11, 12]. These scores were originally developed from operative cohorts and are largely designed to estimate perioperative mortality; therefore, they do not account for critical biological factors such as frailty, malnutrition, and systemic inflammation [14, 31]. Moreover, they provide limited insight into long-term risk profiles. Our findings suggest that the HALP score may help fill this gap by integrating markers of low-grade chronic inflammation, impaired immune competence, and protein–energy depletion, thereby offering meaningful prognostic information regarding long-term survival.
Preprocedural hematologic parameters reflect systemic inflammation and malnutrition in elderly TAVI candidates. Reduced hemoglobin levels indicate impaired oxygen-carrying capacity and increased physiological frailty, whereas hypoalbuminemia reflects malnutrition, chronic inflammation, and endothelial dysfunction [32, 33]. Lymphopenia denotes immune suppression and heightened inflammatory stress, while an elevated platelet count is considered a marker of systemic inflammatory activity [34]. Within this framework, the HALP score serves as a composite marker encompassing inflammatory, nutritional, and hematologic domains and holds particular relevance in elderly individuals with severe aortic stenosis.
Previous studies have evaluated post-TAVI outcomes using different biological and functional approaches. Although biomarker-based positron emission tomography (PET)/computed tomography (CT) studies demonstrated temporal changes in inflammatory markers, no consistent associations with early valve degeneration were observed [35]. Similarly, investigations focusing on iron deficiency did not show a significant impact on functional capacity after TAVI [36]. These observations suggest that isolated biomarkers may be insufficient to reflect the multifactorial biological mechanisms underlying long-term outcomes in elderly TAVI patients. In this context, the HALP score, integrating hematologic, inflammatory, and nutritional components, may provide a more comprehensive assessment of long-term risk after TAVI.
Our findings are consistent with the existing literature demonstrating that inflammatory and nutritional markers – such as CRP, NLR, SII, and albumin – predict mortality after TAVI [18, 19, 37, 38]. Moreover, previous studies have shown that the NAPLES score, another composite indicator similar to HALP, is also associated with post-TAVI mortality [39]. Prior research has reported that hypoalbuminemia is linked to prolonged hospitalization, higher rates of readmission, and poorer long-term survival, whereas inflammatory indices such as SII and NLR were associated with increased procedural complications and early mortality [18, 37, 38]. By combining these components into a single measure, the HALP score may provide prognostic discrimination. In our analysis, HALP remained statistically significant even when strong baseline predictors such as chronic kidney disease were included in the model.
Given that the HALP score is inexpensive, readily available, and routinely obtained in the preprocedural setting, it could be incorporated into existing risk models. Moreover, several studies have demonstrated the superiority of machine-learning–based risk algorithms over traditional clinical scores [40, 41]. Integrating HALP into such models may further enhance predictive performance and refine individualized risk assessment. In addition, the strong association between HALP and mortality suggests that anemia, malnutrition, and inflammation may represent modifiable therapeutic targets. Whether interventions addressing these pathways can improve the HALP score and ultimately translate into better long-term outcomes warrants investigation in future prospective studies.
A few limitations merit consideration. First, the retrospective nature of the study restricts causal inference and may be subject to selection bias; in particular, in-hospital mortality cases were excluded to focus on long-term outcomes, which may inadvertently remove the sickest patients and introduce bias. Second, HALP is a biologically dynamic marker, yet only baseline values were analyzed, and serial measurements were not available. Third, although HALP demonstrated prognostic significance, a formal comparative analysis with established surgical risk scores such as EuroSCORE II or STS-PROM could not be performed due to incomplete availability of these scores in the study cohort. Therefore, statements regarding the complementary or incremental prognostic value of HALP should be interpreted as hypothesis-generating. Finally, the HALP cut-off value (36.82) was derived using a data-driven method and should be interpreted with caution, as its clinical applicability requires external validation. Future prospective studies with larger event numbers and longitudinal HALP assessments are warranted to confirm these findings and to determine whether targeted interventions can modify HALP and improve outcomes.
Conclusions
Lower preprocedural HALP levels were associated with poorer long-term survival in patients undergoing TAVI. By integrating nutritional, inflammatory, and hematologic parameters, the HALP score may help identify patients at increased risk who are not fully captured by conventional risk models. Given the retrospective design of the study, these findings should be considered hypothesis-generating and warrant confirmation in prospective studies with serial measurements and comparable patient profiles.







