Summary
Developments in fusion imaging (FI) software have facilitated easy use of three-dimensional (3D) roadmaps based on preregistered computed tomography (CT) or magnetic resonance Imaging (MRI) datasets for guidance of cardiac catheterizations. The aim of this study was to report the initial results from the first international prospective registry of cardiac catheterizations guided with fusion of CT and MRI datasets. Direct 2D–3D registration of pre-catheterization CT or MRI is a safe and effective method of guidance of cardiac catheterization in selected congenital heart disease. In selected patients, FI facilitates percutaneous interventions without contrast angiography.
Introduction
Over the last decade, tremendous progress has been made in the field of guidance of cardiac catheterizations. New angiographic systems have enabled improved quality of imaging with significant reductions in radiation exposure [1]. Introduction of three-dimensional rotational angiography (3DRA) brought the third dimension and the ability to color code anatomy to the previously gray and two-dimensional (2D) catheterization guidance [2–6]. Modern imaging platforms allow real-time creation of a 3D reconstruction, which after processing can be automatically overlaid as a static roadmap on the fluoroscopy screen [7–13]. Several centers have incorporated this modality into routine practice, achieving superior diagnostic value of 3DRA and improved guidance together with reduced contrast and radiation exposure compared to the traditional 2D guidance [14–20].
Fixed 3D roadmaps based on preregistered magnetic resonance imaging (MRI) or computed tomography (CT) datasets for guidance of cardiac catheterizations have been available for a few years [11, 21–24]. Several groups have described examples of successful clinical combination of X-ray and MRI techniques including 3D image overlay of previously acquired MRI datasets for guidance of cardiac catheterization [25–27]. More recently, improvements in fusion software enabled direct registration of 2D fluoroscopy with previously acquired 3D datasets (2D–3D registration) [28–34]. Early single-center experiences with the latest fusion imaging (FI) platforms showed benefits in reduced contrast usage, radiation exposure and shorter procedural times [35–39].
Aim
In this paper we present results from the first international prospective registry of cardiac catheterizations guided with fusion of CT and MRI datasets.
Material and methods
We performed a physician-indicated multi-center prospective study including five reference centers (Poland, Germany, United States, South Korea, and Mexico) experienced with application of 3D guidance for treatment of congenital and structural heart disease. Consecutive patients with clinically indicated pre-existing cross-sectional imaging deemed clinically suitable by the operator for FI assisted cardiac catheterization were included in the study. The study protocol was approved by appropriate institutional review boards.
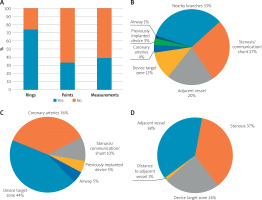
The application of fusion software (VesselNavigator, Philips Healthcare) is based on four steps described in detail elsewhere [40]. Briefly, in the first step, “segmentation,” the software generates a volume-rendered 3D reconstruction from the CT or MRI dataset (Figure 1 A). Segmentation is performed by highlighting and selecting the desired vessel(s) or region. The second stage, “planning,” involves labelling key anatomy with marking rings/points, taking measurements and saving optimal angulations, which can be recalled during the intervention. The third stage, “registration”, involves fusion of fluoroscopy with the labelled 3D reconstruction from the segmentation step and can be done through 2D–3D or 3D–3D registration.
Figure 1
Key information on utilization of fusion of pre-catheter imaging for guidance of cardiac cauterization: four steps of application of fusion imaging (A) Distribution of cardiac procedures (B), accurateness of the initial and intra-procedural registration (C), distribution of type of guidance (D) and overall usefulness of fusion imaging (E)
*2 patients underwent two procedures during one catheterization.

Once mapped, movements of the c-arm and catheterization laboratory table will be tracked in real time and followed in 3D by the fused reconstruction. Finally, “live guidance” of the procedure is performed with the 3D roadmap overlaid in the AP plane (monoplane) and presented in several rendering modes with or without marker rings and other labels. Movement of soft wires/catheters within the border of the overlaid 3D roadmap is used to sense check the accuracy of the fusion alignment. In the event of 3D roadmap misalignment, manual correction can be made either with internal markers or with a small volume contrast injection.
Previously published protocols were modified to assess the quality of non-invasive imaging, accuracy of registration, and subjective usefulness of FI [11, 18, 27, 35, 36]. A three-point scale was used to evaluative the quality of available pre-catheterization imaging (Table I). Accuracy of alignment between fluoroscopy and 3D reconstruction was judged qualitatively then converted to a categorical scale. A modified Likert scale was used to score the overall usefulness of FI for each case.
Table I
Scales used to score quality of pre-catheterization imaging, accuracy of alignment of the three-dimensional (3D) roadmap with 2D fluoroscopy, and overall usefulness of fusion imaging (FI)
Anonymized data, including details of pre-catheterization imaging, registration methods, accurateness of overlay, procedural details, and categorially validated utility of 3D guidance with FI, were collected through a purpose-built internet database application. The operator or technician performing segmentation and/or registration, and the operator utilizing 3D guidance provided answers to corresponding parts of the questionnaire.
Results
Over 24 months, FI was used in 205 patients for guidance (n = 182; 88.8%) or planning (n = 23; 11.2%) of cardiac catheterization (Figure 1 B). A median of 34 records were submitted by each center (range: 26–86 patients). Table II summarizes patient characteristics, pre-catheter imaging and catheterization data.
Table II
Patient characteristics, pre-catheter imaging, and catheterization data
Type of catheterization
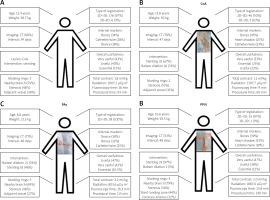
Among 182 patients in whom FI was used for guidance of cardiac catheterization, interventional procedures were performed in 143 (78.6%) patients and diagnostic in 39 (21.4%) patients (Figure 1 B). In the former group, a total of 145 interventions were conducted, including 2 patients who underwent two procedures during one catheterization. Treatment of coarctation of the aorta (CoA; stenting: n = 35, balloon dilatation: n = 16) was the most common intervention, followed by treatment of pulmonary artery stenosis (PA; balloon dilation: n = 21, stenting n = 18) and right ventricular outflow tract dysfunction (RVOT; valve implantation: n = 33, balloon dilation: n = 1) (Figure 2). Less common interventions included device occlusion of collaterals (n = 6), dilation of veins (systemic: n = 3, pulmonary: n = 3), stent implantation in the arterial duct (n = 4), and creation of an interatrial communication (n = 2).
Figure 2
Representation of an ‘average’ patient (A) who underwent fusion of pre-catheter imaging for guidance of cardiac cauterization reported to the registry and the three most common clinical situations guided with fusion imaging including coarctation of aorta (CoA, B), pulmonary artery stenosis (PAs, C), and right ventricular outflow tract obstruction (RVOTO, D). Data are presented as median and percentage
CT – computed tomography, 2D – two-dimensional, 3D – three-dimensional.
Among 39 patients who underwent a diagnostic procedure, right or left heart catheterization was performed in 26 and 6 patients, respectively. In a further 7 patients, a 3D roadmap was used to guide right and left heart catheterization.
Pre-catheterization imaging and segmentation
Computed tomography imaging was used in 137 (66.8%) patients, and MRI in the remaining 68 (33.2%) patients (Table II). Imaging studies were performed at a median interval of 35 days (0–54 months) prior to the date of fusion imaging. Most studies (186, 90.7%) were classified as of good quality, while 9 (4.4%) studies were of suboptimal quality. In the remaining 10 (4.9%) scans, the target structures were poorly visualized, hindering optimal segmentation, including poor quality, blurred MRI scan (n = 3), timing of CT acquisition favoring contrast enhancement on the other heart side (n = 3), poor opacification of target vessels on CT due to contrast washout or significant stenosis (n = 2), significant stent artifacts on CT (n = 1), and poor quality CT scan (n = 1). There were no significant differences between quality grading of CT and MRI scans.
Segmentation and planning
Segmentation was most commonly performed on automatically generated 3D reconstruction (146 cases, 71.2%) or on a combination of the 3D reconstruction and 2D multiplanar reformats (MPRs; 36 cases, 17.6%). The latter alone was used in 23 (11.2%) cases. Difficulty in segmentation of imaging datasets was reported in 13 cases (11 CT and 2 MRI scans), including 9 studies initially judged to be of sufficient quality. The most common difficulties encountered were distinguishing the border between adjacent vessels with similar signal strength (n = 4), segmentation of large vessels with inhomogeneous contrast opacification (n = 2), and selection of small coronary vessels (n = 2).
For planning tools, ring markers were most frequently used (151 patients; 73.7%), followed by measurement lines (79; 38.5%) and marking points (67; 32.7%) (Figure 3).
Registration
2D–3D registration was performed in 176 (96.7%) patients. 3D–3D registration was utilized in the remaining 6 (3.3%) patients (Figure 4). In these, a non-contrast rotation was used in 5 patients and rotational angiography in the remaining one. A median of 4 (range: 3–6) reference points were used to register both the rotational and the previously acquired 3D dataset and were placed on vertebral bodies (5 patients) and the carina (4 patients).
Figure 4
Types and techniques of registration of two-dimensional (2D) fluoroscopy and three-dimensional (3D) reconstruction, including distribution of internal markers: A – types of registration, B – techniques of registration, C – type of internal markers
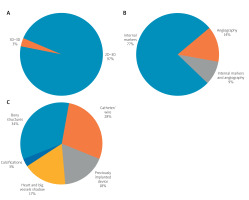
2D–3D registration was most often performed with internal markers (140 patients; 77%), angiography (25; 14%), or a combination of both (17; 9%). Bony structures (34%) and catheters/wires (28%) were most often used for alignment, followed by previously implanted devices (18%), the cardiac silhouette (17%), or calcifications (3%).
Accurate initial 3D roadmap alignment was achieved in 142 (78%) patients (Figure 1 C). In 36 (19.8%) patients, an unimportant minor mismatch was observed. Four patients had an important mismatch (2.2%) requiring realignment of the imaging at the beginning of the catheterization. In all patients in this group, CT data were fused with internal markers for initial 2D–3D registration. In 3 patients, somatic growth after acquisition of the imaging scan was blamed for the inaccurate alignment. In 1 case, poorly selected internal markers for registration resulted in a major mismatch.
3D guidance
Seventeen (9.3%) patients required intra-procedural readjustment of the 3D roadmap due to anatomic distortion caused by the introduction of a stiff wire or sheath (n = 13), somatic growth since acquisition of the pre-catheter imaging (n = 2), or patient movement on the table after initial registration (n = 2) (Figure 1 C). The prevalence of a major intra-procedural mismatch was greatest when stenting the ductus arteriosus (DA) (75%) followed by venous stenting (16%), CoA (8%), PA (6.9%), or RVOT dilation (5.9%). Significant distortion of the anatomy resulting in malalignment of the 3D roadmap occurred most commonly during stent implantation (12 out of 14 interventional catheterizations; 85.7%). Acceptable alignment was reestablished with angiography (1–3 injections) in all but 2 patients.
Catheterizations with 3D guidance only
In 37 (20.3%) patients, catheterization was performed using only 3D guidance; the remaining 145 (79.7%) patients required additional angiography (Figure 1 D). Three-dimensional guidance alone was used in nearly a quarter of RVOT (23.5%) or PA interventions (23.1%) and in 20% of CoA interventions. For the treatment of the three most common lesions reported in the registry, 3D guidance alone was more commonly used for balloon dilation than for stent implantation (29.7% vs. 19.8%), but the difference was not statistically significant.
Usefulness of fusion imaging
Overall, FI for guidance of cardiac catheterization was deemed beneficial in 179 (98.3%) patients, specifically described as essential in 27 (14.8%) patients, very useful in 79 (43.4%), and useful 73 (40.1%) patients (Figure 1 E). In 2 (1.1%) patients FI was not useful, and in one it was misleading (0.5%). An 81-day-old patient (6 kg) with a stenosis of the Sano shunt had significant distortion of the outflow anatomy with introduction of the balloon/stent assembly, rendering the 3D roadmap not useful. This was also the case for a 28-year-old patient undergoing native RVOT stenting, where 3D guidance was not useful due to significant anatomic distortion after introduction of stiff equipment. Fusion imaging was felt to be misleading for guidance around complex right heart anatomy in a 65-day-old (5 kg) patient in whom segmentation of a CT scan performed 34 days earlier produced an inaccurate roadmap due to somatic growth.
Discussion
Three-dimensional imaging for guidance of cardiac catheterization in patients with congenital heart disease has been reported to provide benefit over traditional 2D angiography [2, 3, 5]. However, the growing worldwide experience with 3D imaging in the catheterization laboratory comes exclusively from single-center and retrospective studies. According to our knowledge, this is the first multicenter, prospective registry focused on utilization of 3D guidance for cardiac catheterizations.
The study was designed to collect practical information regarding preparation of 3D reconstruction, techniques of fusion of the 3D roadmap with fluoroscopy, intra-procedural effectiveness of 3D guidance and potential impact on factors such as contrast and radiation exposure or duration of catheterization.
In most patients, 3D reconstruction was derived from CT datasets, which most likely reflects its worldwide higher availability over MRI. Nine out of ten non-invasive studies were subjectively scored as of good quality, with no significant differences between CT and MRI scans. In the remaining 10% of cases, planning tools such as marking rings or points were used to help overcome limitations of the 3D datasets. This shows that suboptimal imaging quality should not discourage reutilization for 3D guidance, as in most cases it can be compensated during the planning stage. Also, we should note that none of the prior imaging was requested or prescribed with fusion in mind; specific image protocol planning could make a difference to the practicality and accuracy of the fused map.
Labelling with planning tools was used in 80% of the registry patients, with marker rings being the most popular. The rings were used to mark the nearest branch or adjacent structure – either a vessel (coronary artery during percutaneous pulmonary valve implantation (PPVI)) or an airway (left PA stenting in a patient after the Norwood operation) – or indicate the landing zone for a stent or an occluder. Additionally, marking rings facilitate the addition of measurement lines, the second most common planning tool used among the registry patients. The rings displayed on MPRs show a perpendicular cut through the highlighted structure allowing simple and reliable measurements. Taking specific measurements of a particular lesion is another advantage of reutilization of CT or MRI scans with fusion software.
Reutilization of previous 3D datasets puts less emphasis on creation of the 3D reconstruction, as it is with 3DRA or echo-fluoro fusion, but instead requires accurate registration technique to achieve a reliable roadmap. Several groups have reported various techniques for accurate registration, based either on use of external fiducial markers or internal markers and acquisition of rotational angiography or a blind spin (3D–3D registration) [11, 22, 25–27]. In most of the reported patients, direct 2D–3D registration was used, which negated the need for preforming additional steps other than storing fluoroscopy required for setting up the iso-center. Internal markers alone were sufficient for registration in 3 out of 4 patients. The combination of 2D–3D registration and internal markers, which added little to the workflow, allowed accurate initial alignment in almost 80% of cases. Still, in every fifth patient an unimportant mismatch was observed partially related to differences in respiratory and cardiac phases or patients’ positioning during scanning and catheterization.
The longer interval between the scan and cardiac catheterization was not an issue during registration in most patients. In fact, MRI performed 47 months earlier and CT performed 54 months earlier were successfully utilized for 3D guidance in adult patients. When it comes to younger pediatric patients, a much shorter interval might hinder reutilization of even the best quality 3D dataset. Previous report has shown successful usage of a CT scan performed 18 months earlier for an FI-guided diagnostic catheterization in a 3.5-year-old patient [29]. However, in three out of 4 registry patients with a major mismatch between the fluoroscopy and 3D roadmap, somatic growth after acquisition of the scan was blamed. Based on the aforementioned single-center experience supported with this registry’s initial data, it seems worthwhile to segment previous CT or MRI with FI software even in the youngest patients, with awareness of the need for critical evaluation of the accurateness of the 3D overlay.
All currently commercially available FI platforms depend on a rigid registration, which does not compensate for periodic and non-periodic movements of the heart and large vessels. Therefore, accurate initial alignment does not guarantee a reliable 3D roadmap throughout the entire study. One in 10 registry patients required intra-procedural readjustment of the 3D roadmap, most commonly due to distortion of the anatomy after introduction of a stiff equipment. All but two of these patients underwent sent implantation, which, by design, requires introduction of a stiffer wire, long sheath, and balloon/stent assembly. When significant misalignment was noted, in most patients repeat angiography was sufficient to correct the position of the 3D roadmap. Any further utility of 3D guidance in cases where misalignment requires angiography before balloon inflation or device deployment is debatable. Such an injection could alone be used to guide the remainder of the intervention, eliminating further use of the 3D roadmap or, as reported in 15 out of 17 patients, used for realignment and hence continuing its utility throughout the intervention.
In 20% of patients, catheterization was performed using only 3D guidance, with contrast being injected after the intervention. This was most frequently reported for stenosis of RVOT or PA and CoA treatment and more often for balloon dilation than for stent implantation. This percentage is less than reported in single-center experience, probably reflecting a learning curve and overall various operator confidence with the 3D roadmap in different participating centers [34]. Considering the limitations of current 3D guidance modalities, it seems unlikely for all interventions to be performed with only 3D guidance. For PA, RVOT, and CoA interventions, non-invasive imaging is often performed to make or confirm the diagnosis and to assess indications for treatment. It is interesting therefore that these initial registry data suggest that PA dilation or RVOT and CoA stent implantation may be performed with only the prior imaging roadmap.
Overall, FI was judged useful in most patients (98.3%), which is consistent with previous single-center experiences [11, 18, 27, 35, 40]. The initial data from the registry show reproducibility of the encouraging single-center outcomes across centers worldwide. With growing data, the optimal registration protocols for specific patients’ sizes, anatomies, and types of interventions could be established. Unified protocols and the addition of other groups with different imaging techniques (2D imaging, 3DRA) to the study are necessary to objectively explore all benefits of FI with direct 2D–3D registration.
Only a single vendor product was used in all patients included in the registry. Quality of pre-catheter imaging, accurateness of the alignment, and the overall usefulness of FI were judged subjectively. Patients in whom FI was performed were selected independently by the operators at each center, leading to potential selection bias. All operators had previous experience with 3D guided catheterizations, particularly with 3DRA, and were familiar with the limitations of rigid registration. Despite this, variation in the experience of operators less experienced with 3D overlay may have influenced scoring of the alignment and overall usefulness of FI.
Conclusions
Fusion with 2D–3D registration of pre-catheterization 3D imaging with fluoroscopy is a safe and effective method for guidance of cardiac catheterization in selected congenital heart diseases. Initial data from the registry show that fusion imaging is most useful during treatment of CoA, RVOT stenting prior to PPVI, or PA stenosis. In selected patients, fusion of CT or MRI datasets enables percutaneous interventions with no prior contrast application.