Summary
The prognostic significance of the infarct-related artery (IRA) in ST-segment elevation myocardial infarction (STEMI) remains controversial. In this study, although right coronary artery involvement was associated with higher in-hospital mortality than left anterior descending artery involvement, the IRA did not show a consistent independent association with short- or long-term outcomes. In contrast, left ventricular ejection fraction and serum albumin independently predicted in-hospital mortality. Left ventricular function remained the strongest determinant of long-term all-cause mortality, while multivessel disease showed only a borderline association. Major adverse cardiovascular events were independently associated with male sex, ventricular function, renal function, and hemoglobin levels. Overall, these findings suggest that prognostic evaluation in STEMI should focus more on functional and systemic factors than on the anatomical location of the IRA.
Introduction
ST-segment elevation myocardial infarction (STEMI) most commonly results from acute thrombotic coronary occlusion following the rupture of an atherosclerotic plaque [1]. Primary percutaneous coronary intervention (PCI) is the recommended mainstay reperfusion strategy for patients with STEMI [2]. In patients with STEMI, every minute of delay to primary PCI remains critically important [3]. Nevertheless, despite advances in interventional techniques and medical therapies, STEMI remains associated with substantial morbidity and mortality [4].
In STEMI, the infarct-related artery (IRA), has traditionally been considered an important determinant of clinical outcome [5, 6]. Previous studies have reported that STEMI involving the left anterior descending artery (LAD) is associated with worse clinical outcomes compared with infarctions related to the right coronary artery (RCA) or the left circumflex artery (LCx) [7, 8]. This disparity has been attributed to more extensive myocardial injury, lower left ventricular ejection fraction (LVEF) at presentation, and a higher incidence of heart failure in anterior myocardial infarctions.
With the widespread adoption of primary PCI in routine clinical practice, more recent studies have demonstrated that short-term outcomes are largely comparable across different IRAs. Nevertheless, data regarding the prognostic role of the IRA in terms of long-term mortality and major adverse cardiovascular events (MACE) remain inconsistent [9, 10]. Moreover, the number of studies comprehensively evaluating this relationship across both in-hospital and long-term outcomes within cohorts with extended follow-up remains limited.
Therefore, in the present study, STEMI patients were stratified into LAD, RCA, and LCx groups according to the infarct-related coronary artery. We investigated the association between the IRA and in-hospital mortality as well as long-term clinical outcomes in patients undergoing primary PCI.
Aim
The primary aim of this study was to evaluate the impact of the IRA on in-hospital and long-term mortality and clinical outcomes, and to determine whether it constitutes an independent risk predictor.
Material and methods
Study design and population
Consecutive patients who were retrospectively diagnosed with STEMI and underwent PCI after admission to our clinic between January 1, 2015, and January 31, 2016, were included in the study. Patients were retrospectively followed for a mean duration of 103 months (93–109 months). Based on the coronary artery responsible for STEMI, patients were categorized into three groups: LAD, RCA, and LCx. Clinical data, including baseline characteristics, laboratory findings, imaging parameters, and follow-up outcomes, were obtained from the hospital’s digital health records and social security databases.
Exclusion criteria
Patients who were managed medically or surgically without coronary stent implantation or drug-coated balloon intervention; those who received thrombolytic therapy before or during PCI; patients with malignancy; those undergoing chemotherapy or radiotherapy; patients with chronic kidney disease or chronic liver disease; and cases demonstrating occlusion myocardial infarction (OMI) without ST-segment elevation on electrocardiography, a concept that has been increasingly emphasized in the recent literature and is considered a potential paradigm shift [11], were excluded from the study.
Definitions
The diagnosis of STEMI was established based on clinical evaluation in accordance with the ESC 2023 Guidelines for Acute Coronary Syndromes, defined by newly developed persistent ST-segment elevation following the onset of ischemic symptoms, in conjunction with elevated cardiac troponin levels [2]. MACE in our STEMI population included nonfatal recurrent myocardial infarction (MI), cerebrovascular events (CVE), and cardiovascular death occurring during both the in-hospital and long-term follow-up periods after PCI [12]. Multivessel disease was defined as the presence of ≥ 2 diseased coronary vessels.
Study endpoints
The primary endpoint of the study was in-hospital mortality. The secondary endpoints were long-term all-cause mortality and long-term MACE.
Statistical analysis
Statistical analyses were performed using SPSS Statistics (IBM Corp., Armonk, NY, USA). The distribution of continuous variables was assessed using the Kolmogorov–Smirnov test. Non-normally distributed data were presented as median (interquartile range, IQR), and group comparisons were conducted using the Mann–Whitney U or Kruskal–Wallis tests, as appropriate. Categorical variables were expressed as counts and percentages and compared using the c2 test or Fisher’s exact test, when appropriate.
Logistic regression analysis was performed to assess in-hospital mortality, while Cox proportional hazards regression analyses were applied for long-term all-cause mortality and long-term MACE. Clinically relevant covariates, including age and sex, were considered for inclusion, and the IRA, with LAD as the reference category, was evaluated in the regression analyses. Composite risk scores, such as the TIMI risk score, were not included in the final multivariable models because of their composite nature and potential overlap with individual clinical and laboratory variables. Odds ratios (ORs) for logistic regression models and hazard ratios (HRs) for Cox regression models, along with their 95% confidence intervals, were reported. Long-term survival was estimated using the Kaplan–Meier method, and comparisons between groups were performed using the log-rank test. In all analyses, a two-sided p-value < 0.05 was considered statistically significant.
Results
Demographic and laboratory findings
In this study, a total of 693 patients were evaluated, and baseline demographic characteristics did not differ significantly among the groups. Sex distribution (p = 0.98), age (p = 0.99), and smoking status (p = 0.09) were comparable. LVEF was significantly lower in the LAD group (p < 0.001). Diabetes mellitus was more frequently observed in the LAD group (p = 0.037), whereas hypertension rates were similar across groups (p = 0.71). A history of prior coronary artery bypass grafting (CABG) was more common in the RCA group (p = 0.003).
Laboratory evaluation revealed significant intergroup differences in white blood cell count (WBC) (p = 0.032) and neutrophil levels (p = 0.040). High-density lipoprotein (HDL) (p = 0.017), albumin (p = 0.007), and total protein (p < 0.001) levels also showed significant differences. Other biochemical parameters did not demonstrate a significant distinction/difference. Follow-up duration was comparable among the groups (p = 0.13) (Table I).
Table I
Baseline demographic, clinical, and laboratory characteristics of the study population
| Variables | Total n = 693 | LAD n = 304 | LCx n = 116 | RCA n = 273 | P-value |
|---|---|---|---|---|---|
| Gender (female), n (%) | 202 (29.1) | 89 (29.3) | 33 (28.4) | 80 (29.3) | 0.98 |
| Age [years] | 62.00 (51.00–73.00) | 62.50 (49.75–73.00) | 61.00 (53.00–72.25) | 63.00 (51.00–73.00) | 0.99 |
| Smoking, n (%) | 356 (51.4) | 154 (50.7) | 70 (60.3) | 132 (48.4) | 0.09 |
| LVEF (%) | 45.00 (35.00–53.00) | 35.00 (32.00–45.00) | 50.00 (45.00–55.00) | 50.00 (45.00–55.00) | < 0.001 |
| HT, n (%) | 243 (35.1) | 107 (35.2) | 37 (31.9) | 99 (36.3) | 0.71 |
| DM, n (%) | 206 (29.7) | 105 (34.5) | 27 (23.3) | 74 (27.1) | 0.037 |
| Dyslipidemia, n (%) | 38 (5.5) | 17 (5.6) | 8 (6.9) | 13 (4.8) | 0.70 |
| CKD, n (%) | 7 (1.0) | 3 (1.0) | 0 (0.0) | 4 (1.5) | 0.54* |
| Previous MI, n (%) | 32 (4.6) | 12 (3.9) | 4 (3.4) | 16 (5.9) | 0.47 |
| Previous PCI, n (%) | 46 (6.6) | 20 (6.6) | 12 (10.3) | 14 (5.1) | 0.17 |
| Previous CABG, n (%) | 16 (2.3) | 1 (0.3) | 5 (4.3) | 10 (3.7) | 0.003* |
| WBC [× 103/µl] | 12.42 (9.98–15.57) | 12.58 (10.30–15.90) | 11.90 (9.56–14.52) | 12.72 (10.50–16.52) | 0.032 |
| Lymphocyte [× 103/µl] | 1.64 (1.14–2.45) | 1.60 (1.15–2.42) | 1.59 (1.08–2.42) | 1.81 (1.25–2.66) | 0.28 |
| Neutrophil [× 103/µl] | 9.73 (7.46–12.70) | 9.99 (7.62–13.10) | 9.21 (7.25–11.97) | 10.05 (8.02–13.90) | 0.040 |
| Hemoglobin [g/l] | 13.90 (12.50–15.03) | 13.96 (12.63–15.30) | 13.71 (12.30–14.88) | 14.36 (12.62–15.05) | 0.07 |
| Platelet [103/µl] | 247 (209–296) | 247 (210–303) | 244 (196–288) | 252 (224–303) | 0.07 |
| Glucose [mg/dl] median (Q1–Q3) | 137 (112–193) | 141 (116–213) | 136 (112–177) | 127 (108–176) | 0.05 |
| Creatinine [mg/dl] median (Q1–Q3) | 0.83 (0.72–1.01) | 0.84 (0.73–1.03) | 0.82 (0.70–0.99) | 0.81 (0.71–0.96) | 0.11 |
| CRP [mg/dl] median (Q1–Q3) | 0.58 (0.29–1.19) | 0.48 (0.29–1.05) | 0.61 (0.28–1.19) | 0.66 (0.44–1.90) | 0.17 |
| Total cholesterol [mg/dl] | 182 (153–210) | 184.00 (153–217) | 177 (153–205) | 183(153–211) | 0.09 |
| HDL cholesterol [mg/dl] | 34 (29–40) | 35 (30–41) | 33 (28–38) | 32 (28–39) | 0.017 |
| LDL cholesterol [mg/dl] | 118 (95–143) | 120 (97–146) | 115 (92–137) | 121(97–140) | 0.125 |
| Triglyceride [mg/dl] median (Q1–Q3) | 126 (85–191) | 117 (81–201) | 127(86–183) | 133 (90–197) | 0.65 |
| Albumin [g/dl] | 3.59 (3.30–3.87) | 3.60 (3.30–3.90) | 3.50 (3.30–3.70) | 3.60 (3.30–3.82) | 0.007 |
| Uric acid [mg/dl] | 6.00 (4.70–8.00) | 6.20 (4.70–7.97) | 5.80 (4.65–7.95) | 6.10 (5.00–7.95) | 0.54 |
| Total protein [g/dl] | 6.80 (6.30–7.20) | 6.90 (6.40–7.30) | 6.60 (6.10–7.00) | 6.90 (6.40–7.30) | < 0.001 |
| Follow-up duration [months] | 103 (93–109) | 103 (94–109) | 102 (90–108) | 101 (92–108) | 0.13 |
* Fisher’s exact test was used. LVEF – left ventricular ejection fraction, HT – hypertension, DM – diabetes mellitus, CKD – chronic kidney disease, MI – myocardial infarction, PCI – percutaneous coronary intervention, CABG – coronary artery bypass grafting, WBC – white blood cell count, CRP – C-reactive protein, HDL – high-density lipoprotein, LDL – low-density lipoprotein. Statistical significance is considered at a p-value of less than 0.05.
Periprocedural characteristics
Infarct location differed significantly among the groups (p < 0.001). No significant difference was observed between the groups in terms of stent type (DES/BMS) (p = 0.10). In contrast, multivessel disease was more frequently observed particularly in LCx and RCA lesions (p = 0.035). Door-to-balloon time was slightly longer in the LCx group compared with the other groups (p = 0.009).
Prior history of chronic heart failure (CHF) was more prevalent in the LAD group (p < 0.001). Rates of ventricular tachycardia/ventricular fibrillation (VT/VF) (p = 0.69) and recurrent myocardial infarction (p = 0.16) were similar among the groups. In-hospital mortality occurred in (n = 53, 7.6%) patients overall and was significantly higher in the LAD group (n = 32, 10.5%) compared with the LCx (n = 6, 5.2%) and RCA groups (n = 15, 5.5%) (p = 0.041). At 1-year follow-up, mortality was observed in (n = 102, 14.7%) patients overall, including (n = 46, 15.1%) in the LAD group, (n = 21, 18.1%) in the LCx group, and (n = 35, 12.8%) in the RCA group, with no significant difference among groups (p = 0.39). Similarly, long-term mortality occurred in (n = 167, 24.1%) patients overall and did not differ significantly between the LAD (n = 78, 25.7%), LCx (n = 26, 22.4%), and RCA (n = 63, 23.1%) groups (p = 0.69). Presentation with cardiac arrest was more frequent in the LAD group (p = 0.016), whereas atrioventricular (AV) block was most commonly observed in the RCA group (p < 0.001). Length of hospital stay was longer in the LAD group (p < 0.001). The no-reflow phenomenon was observed at a lower rate, particularly in the LCx group (p = 0.021). MACE rates did not differ significantly among the groups (p = 0.10) (Table II).
Table II
Periprocedural features of the study population
| Variables | Total n = 693 | LAD n = 304 | LCx n = 116 | RCA n = 273 | P-value |
|---|---|---|---|---|---|
| Infarct location, n (%) | < 0.001 | ||||
| Anterior MI | 292 (42.1) | 285 (93.8) | 3 (2.6) | 4 (1.5) | |
| Inferior MI | 350 (50.5) | 4 (1.3) | 86 (74.1) | 260 (95.2) | |
| Other | 51 (7.4) | 15 (4.9) | 27 (23.3) | 9 (3.3) | |
| Stent type (DES/BMS) | 486 (70.1) | 221 (72.7) | 72 (62.1) | 193 (70.7) | 0.10 |
| Presence of multivessel disease | 406 (58.4) | 101 (51.8) | 37 (64.9) | 99 (64.3) | 0.035 |
| Door-to-balloon time [min] | 44.10 (43.24–44.95) | 43.90 (42.71–45.08) | 46.97 (44.34–49.60) | 43.10 (41.80–44.41) | 0.009 |
| Cardiogenic shock, n (%) | 50 (7.2) | 28 (9.2) | 8 (6.9) | 14 (5.1) | 0.17 |
| Pulmonary edema, n (%) | 31 (4.5) | 17 (5.6) | 8 (6.9) | 6 (2.2) | 0.06 |
| Chronic heart failure, n (%) | 79 (11.4) | 66 (21.7) | 7 (6.0) | 6 (2.2) | < 0.001 |
| Killip > 2 | 78 (11.3) | 31 (10.2) | 15 (12.9) | 32 (11.7) | 0.70 |
| VT, VF | 81 (11.7) | 38 (12.5) | 11 (9.5) | 32 (11.7) | 0.69 |
| Recurrent MI, n (%) | 127 (18.3) | 46 (15.1) | 24 (20.7) | 57 (20.9) | 0.16 |
| CVE, n (%) | 8 (1.2) | 5 (1.6) | 2 (1.7) | 1 (0.4) | 0.29 |
| In-hospital mortality, n (%) | 53 (7.6) | 32 (10.5) | 6 (5.2) | 15 (5.5) | 0.041 |
| 1-year mortality, n (%) | 102 (14.7) | 46 (15.1) | 21 (18.1) | 35 (12.8) | 0.39 |
| Long-term mortality, n (%) | 167 (24.1) | 78 (25.7) | 26 (22.4) | 63 (23.1) | 0.69 |
| Hospitalization due to heart failure, n (%) | 39 (5.6) | 16 (5.3) | 6 (5.2) | 17 (6.2) | 0.86 |
| Heart rate [beats/min] | 80.00 (72.00–92.00) | 84.00 (75.00–95.25) | 80.00 (72.75–90.25) | 80.00 (70.00–88.00) | < 0.001 |
| Arrest on admission, n (%) | 55 (7.9) | 33 (10.9) | 10 (8.6) | 12 (4.4) | 0.016 |
| TIMI, median (IQR) | 4.00 (2.00–6.00) | 5.00 (3.00–7.00) | 3.00 (2.00–5.00) | 3.00 (2.00–5.00) | < 0.001 |
| AV complete block, n (%) | 28 (4.0) | 3 (1.0) | 4 (3.4) | 21 (7.7) | < 0.001 |
| Length of hospital stay [days] median (IQR) | 4.00 (3.00–5.00) | 4.50 (4.00–6.00) | 4.00 (3.00–5.00) | 4.00 (3.00–5.00) | < 0.001 |
| Cardiopulmonary resuscitation (CPR), n (%) | 49 (7.1) | 29 (9.5) | 7 (6.0) | 13 (4.8) | 0.07 |
| Bleeding | |||||
| Major | 4 (0.6) | 1 (0.3) | 0 (0.0) | 3 (1.1) | 0.40* |
| Minor | 10 (1.4) | 5 (1.6) | 1 (0.9) | 4 (1.5) | N/A* |
| MI, time groups | 0.21 | ||||
| 0–6 | 33 (10.9) | 8 (6.9) | 21 (7.7) | ||
| 6–12 | 84 (27.6) | 33 (28.4) | 73 (26.7) | ||
| 12–18 | 122 (40.1) | 53 (45.7) | 137 (50.2) | ||
| 18–24 | 65 (21.4) | 22 (19.0) | 42 (15.4) | ||
| No-reflow, n (%) | 105 (15.2) | 49 (16.1) | 8 (6.9) | 48 (17.6) | 0.021 |
| MACE, n (%) | 174 (25.1) | 86 (28.3) | 21 (18.1) | 67 (24.5) | 0.10 |
* Fisher’s exact test was used. MI – myocardial infarction, DES – drug-eluting stent, BMS – bare-metal stent, VT – ventricular tachycardia, VF – ventricular fibrillation, CVE – cerebrovascular event, TIMI – thrombolysis in myocardial infarction, AV – atrioventricular, MACE – major adverse cardiovascular events. Statistical significance is considered at a p-value of less than 0.05.
Regression analyses
In the univariable logistic regression analysis, in-hospital mortality was associated with advanced age, lower LVEF, higher TIMI score, diabetes mellitus, smoking status, and increased C-reactive protein and uric acid levels. Multivessel disease showed a borderline association with in-hospital mortality (p = 0.07). Given the limited number of in-hospital deaths, a parsimonious multivariable logistic regression model was constructed using clinically relevant covariates selected a priori to avoid overfitting and collinearity. In this model, lower LVEF remained independently associated with in-hospital mortality (OR = 0.85, 95% CI: 0.81–0.89; p < 0.001). A lower serum albumin level was also identified as an independent predictor (OR = 0.37, 95% CI: 0.18–0.78; p = 0.009). Compared with LAD involvement, RCA as the IRA was independently associated with higher in-hospital mortality (OR = 2.67, 95% CI: 1.01–7.00; p = 0.046), whereas LCx involvement was not. Other variables, including age, sex, and multivessel disease, did not retain statistical significance after adjustment (Table III).
Table III
Univariate and multivariate logistic regression analyses for in-hospital mortality
[i] A parsimonious multivariable logistic regression model was constructed using clinically relevant covariates selected a priori, given the limited number of in-hospital deaths. Odds ratios (ORs) with 95% confidence intervals (CIs) are shown. LVEF – left ventricular ejection fraction, TIMI – thrombolysis in myocardial infarction, DM – diabetes mellitus, HT – hypertension, CRP – C-reactive protein, RCA – right coronary artery, LAD – left anterior descending artery, LCx – left circumflex artery, DES – drug-eluting stent. Statistical significance is considered at a p-value of less than 0.05.
In the univariable Cox regression analysis for long-term all-cause mortality, advanced age, lower LVEF, higher TIMI score, elevated C-reactive protein (CRP) and creatinine levels, lower hemoglobin levels, and the presence of multivessel disease were significantly associated with mortality. Multivariable Cox regression analysis was performed to identify independent predictors, considering clinically relevant covariates and potential collinearity among related variables. In the adjusted model, age (HR = 1.02, 95% CI: 1.00–1.04; p = 0.008), male sex (HR = 1.98, 95% CI: 1.15–3.41; p = 0.014), lower LVEF (HR = 0.95, 95% CI: 0.92–0.97; p < 0.001), higher serum creatinine level (HR = 1.38, 95% CI: 1.13–1.68; p = 0.001), and lower hemoglobin level (HR = 0.77, 95% CI: 0.66–0.91; p = 0.002) emerged as independent predictors of long-term mortality. Although multivessel disease showed a strong association in univariable analysis, it demonstrated only a borderline association after multivariable adjustment (p = 0.05). IRA subgroups were not independently associated with long-term mortality (Table IV).
Table IV
Cox regression analysis for long-term all-cause mortality
[i] The multivariable Cox model was constructed using clinically relevant covariates selected a priori to avoid overfitting and collinearity. HRs with 95% CIs are shown. LVEF – left ventricular ejection fraction, TIMI – thrombolysis in myocardial infarction, DM – diabetes mellitus, HT – hypertension, CRP – C-reactive protein, RCA – right coronary artery, LAD – left anterior descending artery, LCx – left circumflex artery, DES – drug-eluting stent. Statistical significance is considered at a p-value of less than 0.05.
For long-term MACE, univariable Cox regression analysis identified advanced age, lower LVEF, higher TIMI score, elevated CRP levels, increased creatinine levels, lower hemoglobin levels, and multivessel disease as significant correlates. In the multivariable Cox regression model, constructed using clinically relevant covariates to identify independent predictors, male sex (HR = 1.75, 95% CI: 1.11–2.77; p = 0.015), lower LVEF (HR = 0.97, 95% CI: 0.96–0.99; p = 0.020), higher serum creatinine level (HR = 1.38, 95% CI: 1.13–1.68; p = 0.001), and lower hemoglobin level (HR = 0.84, 95% CI: 0.74–0.95; p = 0.005) remained independently associated with MACE. Multivessel disease showed a borderline association after adjustment (p = 0.07), whereas IRA subgroups were not identified as independent predictors (Table V).
Table V
Cox regression analysis for long-term MACE
[i] The multivariable Cox model was constructed using clinically relevant covariates selected a priori to avoid overfitting and collinearity. HRs with 95% CIs are shown. LVEF – left ventricular ejection fraction, TIMI – thrombolysis in myocardial infarction, DM – diabetes mellitus, HT – hypertension, CRP – C-reactive protein, RCA – right coronary artery, LAD – left anterior descending artery, LCx – left circumflex artery, DES – drug-eluting stent. Statistical significance is considered at a p-value of less than 0.05.
Kaplan–Meier survival analysis
When the Kaplan–Meier curves constructed for long-term all-cause mortality were examined, no significant difference in survival was observed among the LAD, RCA, and LCx groups (log-rank c2 = 0.227, df = 2, p = 0.893). The estimated mean survival times were approximately 115.4, 115.9, and 115.8 months for the LAD, RCA, and LCx groups, respectively, with largely overlapping 95% confidence intervals. The median survival was calculated as 120 months (95% CI: 118.4–121.6) in the LAD group and 124 months (95% CI: 120.3–127.7) in the RCA group. In the LCx group, the median survival could not be estimated because the proportion of surviving patients did not fall below 50%. Overall survival distributions were comparable across the three IRA groups (Figure 1 A).
Figure 1
Kaplan-Meier curves for long-term all-cause mortality (A) and MACE (B) according to the culprit artery
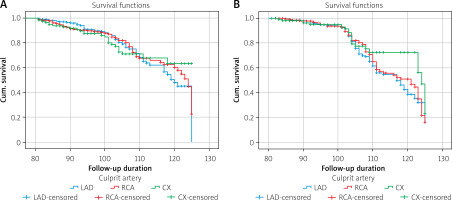
When the Kaplan–Meier curves constructed for long-term MACE were examined, no significant difference in survival was observed among the IRA groups (log-rank c2 = 3.497, df = 2, p = 0.174). The mean survival times were calculated as 114.5 months (95% CI: 112.9–116.1) in the LAD group, 115.4 months (95% CI: 113.6–117.1) in the RCA group, and 118.2 months (95% CI: 115.5–120.9) in the LCx group. Median survival was 117 months (95% CI: 111.3–122.7) in the LAD group, 121 months (95% CI: 112.2–129.8) in the RCA group, and 124 months (95% CI: 122.5–125.5) in the LCx group. Overall, the survival curves demonstrated a similar pattern across all three groups (Figure 1 B).
Discussion
In this study, we evaluated the impact of the infarct-related coronary artery (LAD, RCA, and LCx) on in-hospital and long-term clinical outcomes in STEMI patients treated with primary PCI. Although RCA involvement was associated with higher in-hospital mortality compared with LAD, the IRA did not show a consistent independent effect across short- and long-term outcomes after multivariable adjustment. In contrast, LVEF and serum albumin levels were identified as independent determinants of in-hospital mortality, while LVEF emerged as an independent predictor of long-term mortality, whereas multivessel disease showed only a borderline association after multivariable adjustment. Regarding long-term MACE, multivessel disease showed a borderline association after adjustment, whereas male sex, LVEF, renal function, and hemoglobin levels were identified as independent predictors.
In our study, although the IRA was not identified as an independent predictor of in-hospital or long-term mortality in adjusted analyses, this finding should not be interpreted as evidence that infarct location is clinically irrelevant; rather, it should be understood within a pathophysiological context. In particular, STEMI involving the LAD is well known to be associated with larger infarct size, more pronounced left ventricular dysfunction, and a poorer prognosis [9, 13–16]. In our cohort, LAD involvement was associated with lower LVEF, and once LVEF was incorporated into the multivariable models, the direct association between the IRA and mortality was no longer observed. The change in direction observed for the IRA after multivariable adjustment likely reflects confounding by left ventricular function, suggesting that the prognostic impact of infarct location is largely mediated through LVEF rather than representing a direct effect. These findings support a mediating framework in which the prognostic impact of infarct location operates primarily through its effect on left ventricular systolic function. Accordingly, infarct location should not be regarded as an isolated anatomical variable but rather as a determinant of the extent of myocardial injury and subsequent impairment of LVEF, which in turn influences both short- and long-term clinical outcomes. These findings are consistent with the results of the JAMIR subanalysis, which reported that LAD infarctions are associated with a larger infarct size and a more complicated clinical course [10]. However, while an increase in early mortality was reported in that study, this difference did not reach statistical significance in our cohort. Similarly, Entezarjou et al. demonstrated that LAD infarctions were associated with an increased risk of early mortality, but that this effect diminished over the later phases of follow-up [9]. In a subanalysis of the EXAMINATION-EXTEND study, early mortality was reported to be higher in the LAD-STEMI group, whereas no significant difference related to the IRA was observed during long-term follow-up [14]. These findings suggest that LAD-related STEMI is associated with a more severe clinical presentation, particularly in the acute phase; however, the prognostic role of the IRA appears to be limited with respect to long-term outcomes following primary PCI.
In anterior STEMI, a larger extent of myocardial damage and more pronounced microvascular dysfunction have been demonstrated in cardiac magnetic resonance (CMR) studies [15, 16]. This structural injury is known to be associated with lower LVEF and an increased risk of complications in the early phase. In our study, LVEF remained an independent predictor of long-term mortality after multivariable adjustment, highlighting the central role of ventricular function in long-term prognosis.
In our study, serum albumin levels were found to be significantly associated with in-hospital mortality (OR = 0.37, p = 0.009). Our findings are consistent with prior evidence demonstrating the prognostic relevance of serum albumin in the acute STEMI setting. In a contemporary cohort of patients with STEMI, Bicciré et al. reported that lower admission serum albumin levels were significantly associated with worse in-hospital outcomes, independent of established inflammatory markers such as C-reactive protein and markers of myocardial injury [17]. In a similar study, Oduncu et al. demonstrated that hypoalbuminemia at hospital admission was associated with long-term mortality and advanced heart failure in patients with STEMI undergoing primary PCI [18]. In addition, several studies have suggested that albumin-based indices may be useful for risk stratification in STEMI [19]. Collectively, these findings support the concept that serum albumin reflects a broader pathophysiological state encompassing systemic inflammation, oxidative stress, and increased vascular permeability, rather than acting as a direct determinant of mortality. These observations further indicate that prognosis in STEMI cannot be explained solely by coronary anatomy. Instead, the patient’s systemic condition and physiological reserve play a substantial role in early clinical outcomes. Taken together, serum albumin level may serve as an additional risk marker for in-hospital mortality, independent of the IRA.
Multivessel disease demonstrated borderline associations with long-term all-cause mortality and MACE after multivariable adjustment. Although these associations did not reach conventional statistical significance, they suggest that chronic atherosclerotic burden may still contribute to long-term clinical outcomes. Previous studies have shown that multivessel disease in STEMI patients is associated with a higher risk of recurrent adverse events [20]. Moreover, evidence suggests that more comprehensive revascularization strategies may improve clinical outcomes. These findings support the concept that long-term prognosis is more closely related to the overall burden of coronary artery disease rather than a single culprit lesion. Accordingly, the 2023 ESC Guidelines for Acute Coronary Syndromes recommend complete revascularization during the index procedure or within 45 days in stable STEMI patients with multivessel disease, with a Class I recommendation [2].
In our study, the TIMI risk score was higher in patients with LAD-related STEMI, which is consistent with the more severe clinical presentation typically observed in anterior infarctions. This finding is expected, as LAD involvement is frequently accompanied by lower LVEF, greater hemodynamic compromise, and a higher incidence of early complications [21]. Although the TIMI score demonstrated prognostic value in univariable analyses, it was not included in the final multivariable models because it is a composite score that overlaps with several individual clinical and laboratory variables already considered in the analysis. This approach allowed us to focus on the independent prognostic contribution of fundamental clinical and functional parameters rather than on a summary risk index. These observations are consistent with previous reports indicating that the TIMI score was primarily developed for short-term risk assessment in the acute phase of STEMI [22].
Our findings indicate that risk stratification in patients with STEMI should not be based solely on the type of the IRA. Functional and systemic indicators, such as LVEF and serum albumin levels, are particularly important in the early phase. In the long term, coronary atherosclerotic burden, even when not independently predictive after full adjustment, together with myocardial dysfunction, should be taken into consideration. Closer follow-up is especially warranted in patients with reduced ejection fraction and multivessel disease. Aggressive control of cardiovascular risk factors is essential. Planning revascularization strategies in accordance with current guidelines may contribute to a reduction in long-term clinical events.
The main limitations of this study are its single-center and retrospective design. Therefore, record-related biases inherent to retrospective analyses and the potential impact of unmeasured confounding variables cannot be completely excluded. Because of the retrospective nature of the study, detailed data on in-hospital and long-term medical therapies were not consistently available and therefore could not be comprehensively incorporated into the analyses. Assessment of LVEF and myocardial damage was based on echocardiography, and confirmation with CMR imaging was not performed. In addition, medical therapies were recorded according to discharge prescriptions, and long-term medication adherence could not be evaluated.
The unequal distribution of certain clinical variables among the LAD, RCA, and LCx groups may have limited the statistical power, particularly in subgroup analyses. Furthermore, the lack of a detailed assessment of coronary segments, dominance, and lesion location within the LAD/RCA/LCx territories represents another limitation. The exclusion of patients who received thrombolytic therapy, were managed medically, or were referred for urgent CABG may have resulted in the underrepresentation of LAD-related STEMI cases with more severe clinical presentations, thereby limiting the generalizability of our findings. Finally, the absence of detailed information on revascularization strategies in patients with multivessel disease, including whether culprit-only or complete (index or staged) revascularization was performed, should be acknowledged. Therefore, the association between multivessel disease and long-term mortality as well as MACE may reflect both residual coronary disease and the overall systemic burden of atherosclerosis.
Conclusions
In STEMI patients undergoing primary PCI, the IRA was not a dominant determinant of short- or long-term outcomes, suggesting that its prognostic relevance is largely reflected through the extent of myocardial injury and left ventricular systolic function. In contrast, LVEF and serum albumin emerged as key determinants of in-hospital mortality, while left ventricular function remained the strongest predictor of long-term mortality. Long-term adverse outcomes were associated with a combination of myocardial dysfunction, comorbidity burden, and systemic factors, highlighting that prognosis in STEMI is driven more by global cardiac and systemic status than by culprit vessel anatomy alone.







