Summary
The authors concluded that a high Fibrosis-4 (FIB-4) index is associated with a higher incidence of contrast-induced nephropathy, hospitalizations, and deaths in patients with acute coronary syndrome (ACS). A FIB-4 index ≥ 3.25 is an independent predictor of short- and long-term mortality. FIB-4 may therefore serve as a simple, non-invasive marker integrating hepatic and systemic factors that contribute to poor prognosis in ACS.
Introduction
Acute coronary syndromes (ACS) remain one of the leading causes of hospitalization and mortality worldwide. Despite significant advances in interventional treatment and pharmacotherapy, ACS patients remain at high risk of complications and increased short- and long-term mortality. One of the important complications in this group of patients is contrast-induced nephropathy (CIN), which may occur after diagnostic or therapeutic procedures involving contrast agents. The occurrence of CIN is associated with prolonged hospitalization, increased morbidity, and higher mortality [1]. In recent years, increasing attention has been paid to the use of indices derived from routine laboratory tests for the assessment of cardiovascular risk. Numerous hematological and biochemical markers that reflect chronic inflammation and may have prognostic value in ACS have been described in the literature, including the neutrophil-to-lymphocyte ratio (NLR) [2], platelet-to-lymphocyte ratio (PLR) [3], monocyte-to-high-density lipoprotein-cholesterol ratio (MHR) [4], and red cell distribution width (RDW) [5, 6]. These indices are simple to calculate, inexpensive, and widely available, making them attractive tools in clinical practice. RDW, in particular, has recently gained considerable attention. Elevated RDW in patients with non-ST-elevation myocardial infarction (NSTEMI) has been shown to be an independent predictor of long-term mortality [5], and a meta-analysis confirmed its prognostic value in patients with coronary artery disease undergoing percutaneous coronary intervention (PCI) [6]. The Fibrosis-4 (FIB-4) index, calculated from patient age, platelet count, and aminotransferase (AST, ALT) activity, was originally developed for the non-invasive assessment of liver fibrosis but has also been applied in patients with non-alcoholic fatty liver disease (NAFLD) and chronic hepatitis C. Growing evidence suggests that FIB-4 is associated with the risk of cardiovascular events and mortality [7, 8], which may be explained by chronic inflammation, endothelial dysfunction, and concomitant multiorgan damage. Some studies have also shown that combining FIB-4 with other inflammatory markers, such as NLR, may improve prognostic performance in coronary artery disease [7].
Aim
The aim of the present study was to evaluate the association between the FIB-4 index and the incidence of CIN, as well as 30-day and 365-day mortality, in patients hospitalized for ACS.
Material and methods
Study population
This retrospective, single-center study analyzed patients hospitalized due to ACS who underwent coronary angiography and/or percutaneous coronary intervention (PCI) between January 1, 2015 and December 31, 2018. Data were compiled from three independent sources: the national ORPKI Registry, medical documentation from the Regional Hospital in Kielce, and follow-up information on hospitalizations and mortality provided by the Świętokrzyskie branch of the National Health Fund (NFZ).
From hospital medical records, the following baseline laboratory data were obtained: complete blood count, renal and liver function tests (urea, creatinine, alanine and aspartate aminotransferase), electrolytes, glucose, C-reactive protein (CRP), lipid profile, and cardiac troponin. Additional angiographic and procedural information was extracted from the ORPKI Registry database.
Definitions and procedures
CIN was defined as a relative increase in serum creatinine of at least 25% from baseline or an absolute rise of ≥ 0.5 mg/dl (44 µmol/l) within 48 h after contrast administration [9]. All laboratory values were obtained at baseline, defined as the pre-procedural measurement. Contrast agent volume was recorded in milliliters (ml) and, when applicable, normalized to body weight (ml/kg). The estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease (MDRD) formula, consistent with standard practice at our institution. The FIB-4 index was calculated using the standard formula: FIB-4 = (age [years] × AST [U/l])/(platelet count [109/l] × √ALT [U/l]).
All coronary procedures were performed using non-ionic, iso-osmolar contrast media – iomeprol (350 mg/ml), iodixanol (270 mg/ml), or iopromide (370 mg/ml). Clinical management followed contemporary European Society of Cardiology (ESC) guidelines [10, 11].
The choice of vascular access (radial or femoral) was determined individually by the interventional cardiologist, taking into account the patient’s anatomy, hemodynamic status, and urgency of the procedure. In emergency cases, such as cardiogenic shock, the approach was selected at the operator’s discretion. The type of vascular access may therefore differ between patient groups, and this was beyond the control of the investigators. Generally, radial access is associated with reduced periprocedural and post-procedural mortality.
Statistical analysis
Continuous variables were reported as means with standard deviations (SD) if normally distributed, or as medians with interquartile ranges (IQR) otherwise. Categorical variables were presented as absolute numbers and percentages. Comparisons between groups were made using the c2 test or Fisher’s exact test for categorical variables, and the Kruskal-Wallis test was applied for continuous, non-normally distributed variables, while one-way ANOVA was used for variables following a normal distribution. Univariable and multivariable logistic regression models were used to identify factors associated with 30-day and 365-day mortality. The multivariable Cox proportional hazard regression was used to evaluate the effect of variables on survival time, with results reported as hazard ratios (HR) and 95% confidence intervals (CI). Multivariable analyses employed stepwise forward selection methods. Candidate covariates included demographic variables (age, gender), comorbidities (diabetes, hypertension, dyslipidemia, chronic obstructive pulmonary disease [COPD]), laboratory parameters (hemoglobin, hematocrit, red and white blood cell count, platelets, troponin, total cholesterol, high-density lipoprotein [HDL], low-density lipoprotein [LDL], non-HDL cholesterol, triglycerides, ALT, AST, sodium, potassium, baseline eGFR), procedural variables (contrast volume, contrast volume per kg), and clinical scores (FIB-4, triglyceride-glucose [TyG] index).
Variables were coded as binary or categorical, and continuous variables were standardized for interpretability (e.g., platelets per 25 G/l, troponin per 100 ng/l, total cholesterol per 10 mg/dl). Univariable Cox proportional hazards analyses were performed for all candidate variables. Variables with p < 0.005 in univariable analyses were considered for multivariable modeling. Forward selection added variables stepwise based on statistical significance and HR, with a maximum of one variable per 10 events to reduce overfitting. Collinearity was minimized through dummy coding of categorical variables and iterative selection.
Age was not included in multivariable models due to collinearity with FIB-4. All predictors were measured at baseline. Model discrimination was evaluated using the area under the curve (AUC). Kaplan-Meier survival curves were constructed, and the log-rank test was used to compare survival across FIB-4 groups. Statistical significance was defined as a two-sided p-value < 0.05. All analyses were performed using Python (version 3.12).
Results
A total of 1465 patients met the inclusion criteria and were analyzed. Based on the FIB-4 index, participants were categorized into three strata: < 1.45, 1.45–3.25, and ≥ 3.25.
Marked differences were found among these groups in demographic and clinical profiles. Patients with higher FIB-4 values tended to be older and of lower body weight. Significant variability was also observed in the prevalence of dyslipidemia, smoking habits, and baseline laboratory results, including urea, glucose, hemoglobin, troponin T, and liver enzyme levels. Procedural parameters, such as contrast volume, as well as renal function indices (eGFR and serum creatinine), also differed significantly between the groups.
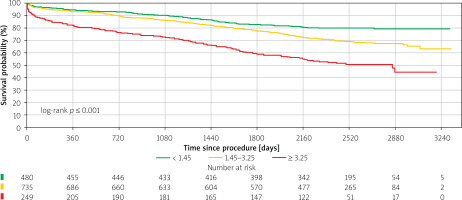
Individuals with FIB-4 ≥ 3.25 represented the most clinically vulnerable subgroup, characterized by the oldest median age, the highest incidence of contrast-induced nephropathy (10.0%), and increased mortality at both 30 days (8.0%) and 1 year (18.1%). The same group also exhibited the highest composite rates of death or rehospitalization at 30 days (9.6%) and 1 year (30.5%). An overview of the study cohort is provided in Table I, while Figure 1 displays Kaplan-Meier curves showing significant separation of survival trajectories across FIB-4 categories. In multivariable models, factors associated with 30-day mortality included a history of stroke (HR > 6), occurrence of CIN (HR > 6), membership of the high FIB-4 index group (HR > 3.5), and hs-TnT levels, with a 3% increase in mortality for each 100 ng/l increase. The discriminative ability of the full model achieved a concordance index of 0.772, compared to 0.637 for the model not including FIB-4 > 3.25, yielding a Dc-index of 0.135 (Table II). For 1-year mortality, multivariable analysis identified male gender (HR = 1.97), high FIB-4 (≥ 3.25, HR = 2.21), and CIN (HR = 2.76) as independent risk factors. Protective factors included higher body weight (HR = 0.98/kg), higher hematocrit (HR = 0.94/%), and baseline eGFR (HR 0.98/ml/min/1.73 m2). The full Cox model achieved a concordance index (c-index) of 0.759, compared to 0.746 for the model not including FIB-4 > 3.25 (Dc-index = 0.013). Continuous residual FIB-4 remained an independent predictor of 1-year mortality (HR 1.08 per unit, 95% CI: 1.04–1.13, p < 0.001), confirming that its prognostic value is not solely driven by age or platelet count (Table III).
Table I
Descriptive characteristics according to FIB-4 index groups
[i] ALT – alanine aminotransferase, AST – aspartate aminotransferase, CABG – coronary artery bypass grafting, CIN – contrast-induced nephropathy, eGFR – estimated glomerular filtration rate, FIB-4 – Fibrosis-4 index, HDL – high-density lipoprotein, LDL – low-density lipoprotein, PCI – percutaneous coronary intervention, TyG index – triglyceride-glucose index, Dyslipidemia was defined as total cholesterol ≥ 200 mg/dl and/or triglycerides ≥ 150 mg/dl, COPD – chronic obstructive pulmonary disease.
Table II
Factors affecting death in 30 days
[i] ALT – alanine aminotransferase, AST – aspartate aminotransferase, CABG – coronary artery bypass grafting, CIN – contrast-induced nephropathy, eGFR – estimated glomerular filtration rate, FIB-4 – Fibrosis-4 index, HDL – high-density lipoprotein, LDL – low-density lipoprotein, PCI – percutaneous coronary intervention, TyG index – triglyceride-glucose index, Dyslipidemia was defined as total cholesterol ≥ 200 mg/dl and/or triglycerides ≥ 150 mg/dl, COPD – chronic obstructive pulmonary disease.
Table III
Factors affecting death in 365 days
[i] ALT – alanine aminotransferase, AST – aspartate aminotransferase, CABG – coronary artery bypass grafting, CIN – contrast-induced nephropathy, eGFR – estimated glomerular filtration rate, FIB-4 – Fibrosis-4 index, HDL – high-density lipoprotein, LDL – low-density lipoprotein, PCI – percutaneous coronary intervention, TyG index – triglyceride-glucose index, Dyslipidemia was defined as total cholesterol ≥ 200 mg/dl and/or triglycerides ≥ 150 mg/dl, COPD – chronic obstructive pulmonary disease.
Discussion
According to the most recent report from the ORPKI registry, the total number of coronary angiography procedures in Poland increased by approximately 3% in 2023 compared with 2022, reaching over 157,000 interventions nationwide [12]. Of these, 91,381 were PCI procedures, representing a slight annual increase of 0.5% [13].
The growing number of invasive procedures underscores the clinical relevance of post-procedural complications such as CIN. CIN has been recognized for decades as a significant adverse event following PCI, associated with prolonged hospitalization and increased mortality [14–16]. Patients with ACS are at particularly high risk, as urgent procedures often preclude adequate pre-procedural hydration and optimization [14]. Identifying reliable predictors of CIN and adverse outcomes in this population therefore remains of major clinical interest [17, 18]. Similar mechanisms of renal injury related to contrast exposure and hemodynamic instability have also been reported in other high-risk populations, such as patients undergoing mechanical thrombectomy for acute ischemic stroke [19].
Anemia, advanced age, and renal dysfunction have been consistently identified as risk factors for CIN and poor outcomes after PCI [20–22]. In our cohort of 1,465 patients with ACS, low hemoglobin concentration, older age, and male sex were independently associated with higher mortality. These findings are in line with prior evidence indicating that reduced hematocrit is an important determinant of CIN and adverse prognosis [21, 22].
Beyond traditional risk factors, recent research has highlighted the prognostic potential of novel biomarkers such as the FIB-4 index. Originally developed to estimate hepatic fibrosis in patients with viral hepatitis, FIB-4 has emerged as a broader marker of systemic inflammation and metabolic stress. In large ACS populations, elevated FIB-4 values have been associated with higher in-hospital mortality, greater incidence of heart failure, and increased risk of major adverse cardiovascular events (MACE) [23, 24]. Similarly, in our study, higher FIB-4 was linked to increased rates of CIN, rehospitalization, and both short- and long-term mortality.
Our results are consistent with prior analyses suggesting that elevated FIB-4 reflects not only hepatic injury but also vascular dysfunction, endothelial activation, and systemic inflammation – all of which may predispose to kidney injury and adverse cardiovascular outcomes. He et al. demonstrated that FIB-4 independently predicted CIN in over 5,600 ACS patients undergoing coronary interventions [25], while Liu et al. confirmed its long-term prognostic value in more than 4,000 patients with ischemic heart disease [26]. These observations, together with our findings, indicate that FIB-4 may represent an integrated marker of patient frailty and multisystem dysfunction rather than a liver-specific index.
The apparent inverse association between baseline creatinine and 1-year mortality in our model likely reflects multicollinearity with eGFR rather than a true protective effect. This finding emphasizes the importance of cautious model interpretation when correlated renal indices are included simultaneously.
Altogether, the emerging evidence supports the potential utility of FIB-4 in refining risk stratification among ACS patients undergoing invasive procedures. Nevertheless, given the retrospective and single-center nature of our analysis, these results should be considered hypothesis-generating rather than confirmatory.
Several limitations should be acknowledged. First, this was a retrospective, single-center study, which inherently limits the ability to infer causality and may introduce unmeasured confounding. Second, data on the etiology of potential chronic liver disease (e.g., alcohol-related vs. non-alcoholic fatty liver disease) were not available. FIB-4 was therefore analyzed as a surrogate of systemic and hepatic fibrosis rather than as a direct measure of liver pathology. Third, information on baseline frailty and the chronic use of nephroprotective or nephrotoxic medications – including SGLT2 inhibitors, NSAIDs, or ACE inhibitors – was lacking. These factors may have influenced both renal outcomes and mortality. Fourth, liver enzymes (AST and ALT) were not routinely available in all ACS patients, possibly leading to selection bias toward individuals with more complete laboratory data. Fifth, detailed angiographic characteristics (culprit lesion, presence of multivessel disease, staged PCI) were not captured, precluding procedural-level risk assessment. In summary, the predictive value of FIB-4 should be interpreted with caution and confirmed in prospective multicenter studies. The FIB-4 index takes age into account, which is why it was excluded from multivariate models due to collinearity; the impact of FIB-4 is not adjusted for age.
Conclusions
In patients with acute coronary syndromes undergoing PCI, elevated FIB-4 index values were associated with a higher incidence of contrast-induced nephropathy, increased hospitalization rates, and greater short- and long-term mortality. FIB-4 may therefore serve as a simple, non-invasive marker integrating hepatic and systemic factors that contribute to poor prognosis in ACS.
However, given the retrospective nature of the analysis and incomplete data on liver disease, frailty, and medication use, these findings should be interpreted with caution. The observed association does not establish causality but supports further exploration of FIB-4 as an adjunctive risk indicator in future prospective studies.