Summary
This meta-analysis evaluated the safety of performing transcatheter aortic valve implantation (TAVI) in centers without on-site cardiac surgery. Across eight observational studies including 22 203 patients, outcomes in non-surgical centers were comparable to those with surgical backup. In-hospital and 30-day mortality did not differ significantly, stroke rates were similar, and emergent cardiac surgery was rare. Pooled mortality in non-cardiac surgery centers was 3% in-hospital and 4% at 30 days. Sensitivity analyses confirmed robustness. The findings indicate that appropriately selected non-surgical centers can deliver TAVI safely, supporting reconsideration of current guideline restrictions to improve access to care.
Introduction
Aortic stenosis (AS) is the most common valvular disease requiring intervention [1, 2], affecting 3–5% of individuals over 75 years [3]. With aging populations and improved diagnostics, its prevalence is expected to rise [4]. Untreated AS carries an unfavorable prognosis, with mortality approaching 20% at one year and 45% at 4 years [3, 5]. The increasing prevalence and high mortality when untreated are placing growing pressure on the demand for aortic valve interventions.
Transcatheter aortic valve implantation (TAVI) is now established as a standard therapeutic option for aortic stenosis [6, 7]. Initially developed for patients at high surgical risk, it is now applied to a broader spectrum of individuals with severe AS [1, 8]. Progress in device design, procedural strategies, operator experience, and advances in pre-procedural planning and imaging have collectively enhanced safety and reduced complication rates [9].
International guidelines recommend that TAVI be performed exclusively in hospitals with on-site cardiac surgery (CS) services to ensure immediate support for rare but life-threatening complications that require emergent cardiac surgery (ECS) [1, 8, 10]. Nevertheless, the improvement in procedural safety and outcomes has reduced the need for ECS to approximately 0.5–1% of cases [6, 11]. Also, when needed, mortality remains high, with index event survival of 54% and 1-year survival rates below 22% [12]. The combination of a low ECS incidence and its unfavorable outcomes raises questions about the need for routine on-site surgical backup [7, 13].
Emerging real-world data have begun to support the safety and feasibility of TAVI in carefully selected non-surgical centers [13]. Large-scale registry analyses and observational studies suggest that, with experienced teams and adequate infrastructure, outcomes are comparable to those in surgical centers [9, 14–16].
While current findings are encouraging, existing evidence remains fragmented across studies with heterogeneous designs and contexts. This meta-analysis aims to clarify contemporary outcomes by systematically assessing the safety, efficacy, and procedural results of TAVI performed in centers without immediate surgical backup compared with conventional surgical sites.
Methods
Search for studies and data extraction
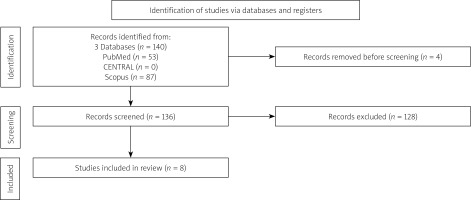
This meta-analysis followed the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines [17]. A systematic search of three electronic databases – PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), and Scopus – was performed up to November 2025, identifying studies describing results of TAVI without on-site CS, or comparing outcomes of TAVI performed in hospitals with and without on-site CS facilities. No language restrictions were applied.
The search strategy combined the terms (“TAVI” or “TAVR” or “transcatheter aortic valve replacement” or “transcatheter aortic valve implantation”) and (“without on-site cardiac surgery”). Two investigators (AA and RV) independently screened the titles and abstracts of all identified records. If a study appeared potentially eligible, its full text was retrieved and reviewed in detail to determine final inclusion. Data were then independently extracted from the eligible studies. Discrepancies were resolved by consensus, and when necessary, a third reviewer (LP) acted as adjudicator. Studies were eligible if they (1) compared TAVI outcomes in hospitals with and without on-site cardiac surgery (two-arm studies) or (2) specifically reported outcomes of TAVI procedures in centers without on-site CS (single-arm studies).
Data extraction was performed using a predefined data collection form that included study design, patient demographics, procedural characteristics, and clinical outcomes. The protocol was registered in the PROSPERO database (CRD420251044095).
We included comparative studies with two groups (TAVI performed with and without immediate on-site CS support) and descriptive single-arm studies reporting outcomes for centers without on-site cardiac surgery. The risk of bias of the included studies was independently assessed by two reviewers (AA and RV) using the Cochrane ROBINS-I tool for non-randomized studies [18]. Any disagreements were resolved through discussion or adjudication by the third reviewer (LP).
Clinical endpoints
The primary outcome was 30-day all-cause mortality. Secondary outcomes included procedural death, the need for emergent cardiac surgery (ECS), and stroke. The original endpoint definitions from each study were used for consistency.
Statistical analysis
All statistical analyses were performed using Stata Statistical Software: Release 18 (StataCorp LLC, College Station, TX). Categorical outcomes were reported as proportions, along with risk ratios (RR) and corresponding 95% confidence intervals (CI). In contrast, continuous outcomes were reported as mean ± standard deviation or median with interquartile range (IQR), as appropriate. A random-effects model was used for the primary analysis to account for between-study variability. Statistical heterogeneity was assessed using the I2 statistic, with 25%, 50%, and 75% considered low, moderate, and high heterogeneity, respectively. A sensitivity analysis was conducted using a fixed-effects model to assess the consistency of the results. Publication bias assessment was not performed due to the limited number of eligible studies.
Two separate meta-analyses were performed: one including only two-arm comparative studies, using risk ratios (RR) for binary outcomes to compare TAVI procedures performed with and without on-site CS; and another including all available studies (single-arm and comparative) to pool event rates (proportions) of outcomes without on-site CS. A meta-analysis of proportions was conducted using a random-effects model with a Freeman-Tukey double arcsine transformation to stabilize variances; the results were then back-transformed for interpretation.
Results
A total of 140 studies were identified through an initial database search. After removing duplicates, 136 studies were screened. Following the application of inclusion and exclusion criteria, we included eight studies (Figure 1) with observational, retrospective designs (3 cohort studies and 5 single-arm studies), covering a total of 22,203 patients (19,373 with on-site CS [87%] and 2830 without [13%]). Baseline patient characteristics for each study are presented in Table I.
Table 1
Baseline characteristics and inclusion criteria of studies included in the meta-analysis comparing transcatheter aortic valve implantation (TAVI) performed in centers with and without on-site cardiac surgery backup. Data are reported separately for surgical and non-surgical centers when available. Continuous variables are presented as mean ± standard deviation or median (interquartile range), and categorical variables are presented as numbers (percentages)
| Parameter | Eggebrecht et al. | Egger et al. | Eggebrecht et al. | Garrido et al. | Gaffor et al. | González et al. | Barashi et al. | Almeida et al. | |
|---|---|---|---|---|---|---|---|---|---|
| Year | 2016 | 2018 | 2014 | 2019 | 2015 | 2025 | 2025 | 2025 | |
| N | 17919 | 1822 | 1432 | 384 | 97 | 100 | 149 | 300 | |
| Surgical center (n) | 16587 | 1532 | 1254 | Not reported | Not reported | Not reported | Not reported | Not reported | |
| Non-surgery center (n) | 1332 | 290 | 178 | 384 | 97 | 100 | 149 | 300 | |
| Age [years]* | CS | 81.1 ±6.1 | 83.0 (79.0–86.0) | 81.6 ±6.2 | – | – | – | – | – |
| No CS | 82.1 ±5.8 | 84.0 (80.0–87.0) | 82.6 ±6.3 | 82.2 (77.2–87.2) | 82.0 ±6.0 | 82.4 ±5.3 | 80.5 ±6.4 | 82.0 ±5.0 | |
| Female, n | 9847 | 1089 | 827 | 202 | 47 | 50 | 75 | 54 | |
| EuroSCORE, % * | CS | 21.0 ±15.4 (a) | 14.2 (9.0–22.2) (a) | 21.0 ±14.0 (a) | – | – | – | – | – |
| No CS | 23.2 ±15.8 (a) | 20.9 (12.8–30.3) (a) | 20.0 ±11.0 (a) | 14.3 ±5.3 (a) | 21.8 ±14.4 (a) | 4.32 ±5.1 (b) | 3.16 ±2.27 (b) | 2.3 (1.6–4.0) (b) | |
| Inclusion criteria | Transfemoral TAVI in 2013 and 2014 German Quality Assurance Registry Aortic Valve Replacement of the Federal Joint Committee | Transfemoral TAVI between January 2011 and November 2016 from the Austrian TAVI Registry | Patients undergoing TAVI transfemoral or transapical at 27 hospitals in Germany between January 2009 and June 2010. | Consecutive patients undergoing TAVI at 10 centers without on-site cardiac surgery. located in 10 different provinces in 5 regions across Spain between May 2010 and May 2018 | Consecutive patients undergoing transfemoral TAVI at a hybrid operating room between 2005 and October 2012 | First 100 patients treated with TAVI in a tertiary referral center without an on-site cardiac surgery department between April 2022 and January 2024 | Consecutive patients treated at the valve clinic of Meir Medical Center (Kfar Saba, Israel) who underwent TAVI between November 2019 and December 2023 | First consecutive 300 patients undergoing TAVI between 2020 and 2024 | |
| Elective, % (n) | Total | 84 (15016) | – | 84 (1197) | – | – | – | – | – |
| CS | – | – | 83 (1034) | ||||||
| No CS | – | – | 92 (163) | 96 (368) | – | – | – | 83 (248) | |
| Self-expandable valve, (n) | Total | – | 73.7 (1343) | 81 (1164) | – | – | – | – | – |
| CS | – | 68.9 (1055) | 83 (1033) | ||||||
| No CS | – | 99.7 (289) | 74 (131) | 100 (384) | 94 (91) | 100 (100) | 71 (106) | 95 (286) | |
| Transcatheter transfemoral access, % (n) | Total | 100 (17919) | 100(1822) | – | – | – | – | – | – |
| CS | 100 (16587) | 100 (1532) | |||||||
| No CS | 100 (1332) | 100 (290) | 100 (384) | 100 (97) | 95 (95) | 100 (149) | 99 (299) | ||
| Left ventricular ejection fraction < 30%, % (n) | Total | 10 (1835) | 9.1(166) | 12 (169) | – | – | – | – | – |
| CS | 10 (1687) | 8.8 (135) | 12 (152) | ||||||
| No CS | 11 (148) | 10.7 (31) | 9.6 (17) | 4.9 (19) | 25 (24) | – | – | – | |
| Low flow low gradient, % (n) | Total | – | – | 11 (158) | – | – | – | – | – |
| CS | – | – | 12 (148) | ||||||
| No CS | – | – | 5.6 (10) | 3.6 (14) | – | – | – | 10 (31) | |
| Bicuspid aortic valve, % (n) | Total | – | – | 2.7 (39) | – | – | – | – | – |
| CS | – | – | 2.5 (31) | ||||||
| No CS | – | – | 4.5 (8) | 1.0 (4) | – | 3.0 (3) | – | 6.0 (18) | |
| NYHA III-IV, % (n) | Total | 85 (15283) | – | 88 (1257) | – | – | – | – | – |
| CS | 85 (14079) | – | 89 (1103) | ||||||
| No CS | 90 (1204) | – | 87 (154) | 67 (256) | 85 (82) | – | 45.6 (68) | 51 (153) | |
| Previous PCI, % (n) | Total | 30 (5313) | 32.3 (589) | 34 (493) | – | – | – | – | – |
| CS | 29 (4856) | 29.9 (458) | 36 (444) | ||||||
| No CS | 34 (457) | 45.3 (131) | 28 (49) | 34 (129) | – | – | 33.5 (50) | 14 (42) | |
| Previous open-heart surgery, % (n) | Total | 18 (3131) | – | 22 (318) | – | – | – | – | – |
| CS | 17 (2893) | – | 23 (285) | ||||||
| No CS | 18 (238) | – | 19 (33) | 9.1 (35) | 26 (25) | 13 (13) | 6.0 (9) | – | |
| Previous permanent pacemaker, % (n) | Total | 11 (2045) | 9.2 (167) | 13 (181) | – | – | – | – | – |
| CS | 11 (1868) | 8.4 (129) | 13 (158) | ||||||
| No CS | 13 (177) | 13 (38) | 13 (23) | – | 27 (26) | 12 (12) | 7.4 (10) | 15 (46) | |
AS – aortic stenosis, CS – on-site cardiac surgery, No CS – no on-site cardiac surgery, EuroSCORE – European System for Cardiac Operative Risk Evaluation, LVEF – left ventricular ejection fraction, NYHA – New York Heart Association, PCI – percutaneous coronary intervention, TAVI – transcatheter aortic valve implantation.
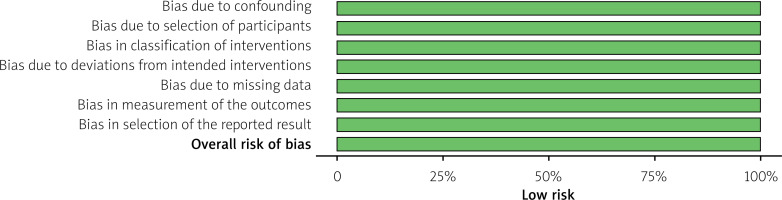
Risk of bias was evaluated using the ROBINS-I tool. All eight included studies demonstrated a consistently low risk of bias across all assessed domains, including confounding, participant selection, intervention classification, deviations from intended interventions, missing data, outcome measurement, and selective reporting (Figures 2 and 3). The overall judgment for each study was rated as low risk, supporting the internal validity of the pooled estimates.
Figure 2
Risk of bias assessment of included trials using the ROBINS-I tool. Green indicates low risk of bias

Figure 3
Risk of bias assessment of included trials using the ROBINS-I tool. Green indicates low risk of bias
The meta-analysis of the three cohort studies showed no significant difference in in-hospital mortality between TAVI performed with or without CS backup (RR = 1.1, 95% CI: 0.6–1.9, p = 0.8). There was moderate heterogeneity (I2 = 69%, t2 = 0.15, p = 0.07), with Egger et al. (2018) reporting an increased risk (RR = 2.3, 95% CI: 1.1–4.9), while both Eggebrecht cohorts showed non-significant protective effects (RR = 0.89, 95% CI: 0.7–1.2 and RR = 0.8, 95% CI: 0.5–1.5, respectively) (Figure 4).
Figure 4
Forest plots of comparative meta-analyses evaluating TAVI outcomes in centers without on-site cardiac surgery versus centers with on-site cardiac surgery backup. Effect estimates are presented as risk ratios with 95% confidence intervals for in-hospital mortality, 30-day mortality, emergent cardiac surgery, and stroke, using random-effects models

For 30-day mortality, the analysis included two studies (Eggebrecht et al. and Egger et al.), yielding a pooled RR of 1.16 (95% CI: 0.51–2.64, p = 0.7). Significant heterogeneity was present (I2 = 78%, t2 = 0.27, p = 0.03), with Egger et al. finding significantly increased risk (RR = 1.7, 95% CI: 1.1–2.8) while Eggebrecht et al. did not find a significant protective effect (RR = 0.75, 95% CI: 0.4–1.4) (Figure 4).
Regarding ECS conversion, two studies yielded a pooled RR of 0.8 (95% CI: 0.2–2.4, p = 0.7), indicating no significant difference between groups. Moderate heterogeneity was observed (I2 = 60%, t2 = 0.42, p = 0.1), with the first Eggebrecht cohort showing a non-significant protective effect (RR = 0.4, 95% CI: 0.2–1.2) and the second showing a non-significant increased risk (RR = 1.4, 95% CI: 0.5–4.1) (Figure 4).
Stroke analysis included three studies, with a pooled RR of 1.1 (95% CI: 0.8–1.5, p = 0.6). No heterogeneity was observed among the studies (I2 = 0.00%, t2 = 0.00, p = 0.4), with consistent results across all cohorts (Figure 4).
A pooled analysis of eight studies was conducted to characterize outcomes specifically in facilities lacking on-site CS backup. The pooled in-hospital mortality rate was 3.0% (95% CI: 1.0–4.0%), with individual study estimates ranging from 0.0% to 10.0%. Substantial heterogeneity was observed (I2 = 84%; p < 0.01). For 30-day mortality, the pooled rate was 4.0% (95% CI: 2.0–6.0%) across seven studies, while heterogeneity remained high (I2 = 80%; p < 0.01).
The requirement for ECS was exceptionally rare, with a pooled rate of 0.0% (95% CI: 0.0–1%). Of the seven studies reporting this outcome, five (Gafoor et al. and all 2025 studies) observed no cases of ECS, leading to a lack of statistical heterogeneity for this specific endpoint (I2 = 0%; p = 0.73). The pooled stroke rate across the available data was 2.0% (95% CI: 1.0–2.0%). Results were relatively consistent across centers (I2 = 71.2%), with most studies reporting incidences between 1.0% and 3.0%, further defining the procedural safety profile in this setting (Figure 5).
Figure 5
Forest plots of pooled proportions of TAVI outcomes in centers without on-site cardiac surgery backup. Summary estimates with 95% confidence intervals are shown for in-hospital mortality, 30-day mortality, emergent cardiac surgery, and stroke, calculated using random-effects meta-analytic models.

A sensitivity analysis was conducted using the propensity score-matched population reported in the Florian et al. (2018) study [16], as significant baseline differences were observed between the intervention and control groups (Figure 6). The meta-analysis of three studies showed no statistically significant difference in in-hospital mortality between TAVI performed with and without on-site CS (RR = 0.9, 95% CI: 0.6–1.3, p = 0.5). The analysis demonstrated no heterogeneity (I2 = 0%, t2 = 0.000), suggesting consistent findings across studies (Figure 2). For 30-day mortality, a pooled analysis of two studies revealed no significant difference between groups (RR = 0.9, 95% CI: 0.6–1.4, p = 0.7). Again, there was no evidence of heterogeneity (I2 = 0%, t2 = 0.00), indicating consistent results between the studies, with both contributing relatively equal weight to the analysis (Eggebrecht et al. [15], 51%; Egger et al. [16], 49%) (Figure 6). Notably, the heterogeneity observed across outcomes virtually disappeared when analyzing the propensity score-matched populations, further supporting the safety of TAVI without on-site CS.
Figure 6
Sensitivity analysis using forest plots of comparative meta-analyses evaluating TAVI outcomes in centers without on-site cardiac surgery versus centers with on-site cardiac surgery backup, restricted to propensity score-matched populations. Effect estimates are presented as risk ratios with 95% confidence intervals for in-hospital mortality, 30-day mortality, emergent cardiac surgery, and stroke

Discussion
We conducted this meta-analysis to address the growing need for TAVI, driven by the increasing prevalence and high mortality of severe AS, as well as disparities in access resulting from the centralization of TAVI in CS centers, considering emerging evidence supporting the safety and feasibility of TAVI without on-site surgical backup.
These results provide insight into the potential role of performing TAVI in centers without on-site CS backup. Across eight observational studies published between 2014 and 2025, including 22,203 patients, 13% underwent TAVI in hospitals without on-site surgical support. In carefully selected programs, TAVI without immediate CS backup achieved outcomes comparable to those observed in centers with on-site surgical availability. This interpretation aligns with recent discussions that focus on establishing safe conditions for implementing TAVI without on-site CS [19].
The pooled analysis of in-hospital mortality showed no significant difference between TAVI performed with or without CS backup (RR = 1.09, 95% CI: 0.6–1.9, p = 0.8). When analyzing propensity score-matched populations, this estimate remained non-significant and showed improved consistency (RR = 0.9, 95% CI: 0.6–1.3, p = 0.5; I2 = 0%). Individual studies reported in-hospital mortality rates ranging from 0% to 10%, with a pooled in-hospital mortality rate of 4% (95% CI: 3–5%) in centers without CS, closely aligning with contemporary TAVI registries and clinical trial benchmarks, even considering the long study period.
Similarly, the analysis of 30-day mortality revealed no significant difference between groups (RR = 1.16, 95% CI: 0.5–2.6, p = 0.7), with consistent 30-day mortality rates ranging from 4% to 7% across studies and no heterogeneity observed in propensity-matched populations (I2 = 0%). This consistency reinforces the procedural safety of TAVI without surgical backup when performed within structured and experienced heart team programs, provided it is adequately planned and executed, and achieves outcomes within accepted safety benchmarks.
ECS remains a major concern when TAVI is performed without on-site surgical backup. In this meta-analysis, ECS was rare, with a pooled rate of 0% (95% CI: 0–1%), indicating a very low but not absent risk. The incidence did not differ between centers with and without cardiac surgery (RR = 0.8; 95% CI: 0.2–2.4). These findings are consistent with large contemporary registries and surgical bailout cohorts, in which ECS rates typically range between 0.5% and 0.8% [11, 12]. Moderate heterogeneity was observed (I2 = 60%), likely reflecting differences in procedural protocols, operator thresholds for conversion, and patient selection. Non-surgical centers may adopt a more conservative approach, excluding patients with high-risk anatomical features such as severe left ventricular outflow tract (LVOT) calcification or borderline annular sizing, which likely contributes to the low observed ECS rates. In this context, TAVI without on-site surgical backup appears safe when performed in carefully selected patients within structured and experienced programs.
The declining need for surgical conversion also reflects advances in endovascular bailout strategies. Covered stents, snare-based retrieval, and valve-in-valve implantation are now routinely used to manage complications that previously required surgery [20]. These developments further support the feasibility of TAVI in selected non-surgical centers.
Importantly, stroke incidence was consistent across studies (RR = 1.08, 95% CI: 0.79–1.46, p = 0.64), with no heterogeneity (I2 = 0%, p = 0.35). As expected, this finding underscores that neurological events in TAVI are predominantly influenced by patient and procedural factors rather than the availability of on-site surgery.
There is significant heterogeneity across outcomes, ranging from moderate to substantial (I2 = 60–98%), which represents a limitation of this meta-analysis. This likely reflects variability in the routine, study design, period between studies, procedural protocols, patient risk profiles, valve generations, procedural techniques, and local resource availability at different centers. Notably, despite this heterogeneity, no significant differences in key outcomes were observed, reinforcing the consistency of safety signals in TAVI without CS backup. Additionally, when focusing on propensity-matched analyses, heterogeneity decreased significantly, while the results remained consistent, thereby strengthening the robustness of the primary findings.
To further test the robustness of our findings, we conducted a sensitivity analysis using an expected events model derived from the published literature, which accounted for the anticipated incidence of adverse outcomes. Notably, the results of this analysis were consistent with our primary findings, reinforcing the validity of our conclusions and addressing potential concerns regarding underreporting or selection bias in retrospective series.
The 2025 ESC/EACTS guidelines recommend that TAVI be performed in Heart Valve Centers, with on-site cardiac surgery [2]. The guidelines also introduce the Heart Valve Network concept, emphasizing integrated regional organization of care with structured referral pathways to specialist centers [2]. Our findings support a network-based model, in which selected non-surgical centers operate within structured systems of care with formal access to surgical expertise and backup when needed. In this context, surgical backup remains essential. However, its value lies in timely access to surgical expertise through organized referral pathways and formal collaboration, rather than mandatory, permanent on-site availability in all cases.
The improvement of the safety profile, supported by our meta-analysis, is closely linked to advancements in device technology, procedural planning, periprocedural imaging, and operator expertise. Notably, studies from 2014 to 2025 were characterized by substantial shifts towards lower-risk patient profiles, refinements in procedural techniques, and the introduction of newer-generation prostheses with improved safety profiles. These factors likely contribute to the observed heterogeneity in outcomes. Also, the advancements in the TAVI procedure have significantly improved its outcomes, reducing procedural complications and the need for ECS. As such, the feasibility of safely performing TAVI without CS backup is increasingly supported by evidence, offering a pragmatic solution for selected centers, particularly in regions with limited surgical capacity or geographic barriers.
While this meta-analysis provides valuable insights, several limitations must be acknowledged. First, all included studies were observational and retrospective, inherently prone to selection bias, residual confounding, and reporting variability. The lack of randomized controlled trials limits the ability to establish causality between the presence of CS backup and clinical outcomes. Differences in patient baseline characteristics, TAVI device generations, operator expertise, procedural techniques, and perioperative management protocols were not uniformly reported, which may have influenced the results. Additionally, the heterogeneity observed highlights variability in study populations, definitions of procedural complications, thresholds for ECS activation, and institutional policies. Substantial heterogeneity (I2 > 60% in multiple outcomes) suggests that the observed differences across studies cannot be attributed solely to chance. Although pooled analyses showed no significant differences in outcomes, the heterogeneity may limit the generalizability of these findings. Finally, publication bias cannot be excluded. Future prospective studies, larger multicenter registries, and randomized clinical trials are needed to validate these findings, identify optimal patient selection criteria, and establish programmatic standards to safely expand TAVI access in this setting.
Conclusions
Our findings suggest that TAVI can be safely performed in carefully selected centers without on-site surgical backup when implemented under strict programmatic and organizational criteria. Rather than challenging current guideline recommendations, these results support the Heart Valve Network model, in which non-surgical centers operate within structured systems of care with defined expertise, protocols, and access to surgical support.
Differences in patient baseline characteristics, device generations, operator experience, and procedural and periprocedural management across studies may have influenced outcomes. Randomized controlled trials and prospective registries are therefore needed to confirm these observations and to inform future recommendations as TAVI programs continue to expand.