Transcatheter closure of the patent ductus arteriosus (PDA) is a typical method of treatment of hemodynamically significant PDAs. Among the elderly, the incidence of PDA is lower than in children. Moreover, adult PDAs often exhibit multiple pathological changes not typically observed in the pediatric population, including calcification, aneurysmal dilatation, tortuosity, and shortening of the duct. Recently we presented 33 patients from our center older than 55 years old; the oldest one was 85 years old [1]. In those cases, various devices were used – mainly different types of Amplatzer Duct Occluders and Amplatzer Muscular Ventricular Septal Defect (VSD) Occluders.
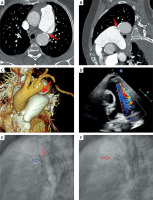
We present a case of an 80-year-old woman, in whom a PDA was closed using a KONAR-MF occluder, which is primarily designed for percutaneous VSD closure. She presented at the Cardiological Emergency Department with exacerbated dyspnea, decreased exercise tolerance, and frequent atrial fibrillation episodes. The echocardiographic examination revealed the following: mildly decreased left ventricular ejection fraction (LVEF) of 50%; enlarged left ventricle with an end-diastolic diameter (LVEDD) of 62 mm, and an end-diastolic volume (LVEDV) of 145 ml; an enlarged left atrium with a diameter of 44 mm and an area of 28 cm2, dilated pulmonary artery to 40 mm; right ventricular systolic pressure (RVSP) 47 mm Hg; right ventricular size of 38 mm in the 4-chamber view; moderate mitral regurgitation; pericardial effusion of up to 12 mm and PDA with left-to-right shunt. Her NT-pro-BNP concentration was 4289 pg/ml. The computed tomography angiography showed blood flow from the aorta to the main pulmonary artery with a calcified aortic bulge around the ductal ostium (Figures 1 A–D). The patient was diagnosed with PDA 2 years earlier but refused interventional treatment. In the course of ambulatory serial echocardiographic assessments, gradual left ventricular enlargement was observed, from initially LVEDD 52 mm and LVEDV 122 ml (74 ml/m2) to LVEDD 62 mm and LVEDV 145 ml (88 ml/m2) on admission. Due to the significance of the shunt, frequent heart failure decompensations and poorly tolerated atrial fibrillation episodes, the patient consented to transcatheter PDA closure.
In shallow sedation the right femoral artery was punctured, a JL 5-French catheter was introduced in the PDA area, and contrast was injected. PDA type A with the diameter of 4.5 mm and numerous calcifications of the duct ampulla were visualized. The catheter was exchanged and the second catheter was placed in the pulmonary trunk via the PDA. The pulmonary artery pressure was 42/15 mm Hg, mean 30 mm Hg. Subsequently the KONAR-MF VSD Occluder [2, 3] 8/6 mm was introduced. The distal disc was opened in the pulmonary trunk (Figure 1 E), then the device was withdrawn to the PDA and the proximal disc was opened in the aorta (Figure 1 F). The stability of the device was demonstrated through aortography. The procedure and post-procedure period were uneventful. The patient was discharged 7 days after the procedure, free of heart failure symptoms. The NT-pro-BNP concentration on discharge was 828 pg/ml, and the pericardial effusion had decreased to 6–7 mm.
Figure 1
Arrows indicate the PDA. A, B – Two-dimensional computed angiography reveals blood flow from descending aorta, through the PDA, into the pulmonary trunk. Non-contrasted blood from the descending aorta flows into the contrasted pulmonary trunk. C – Three-dimensional computed angiography reconstruction. D – Transthoracic echocardiographic visualization of the shunt. E – Catheter introduced from aorta through PDA (red arrow) to pulmonary artery – close to the pulmonary ostium of the PDA. Contrast infusion flows into the pulmonary trunk (blue arrow) (lateral projection). F – Fluoroscopy image showing the KONAR occluder implanted in the PDA (red arrow)
Eight months post-procedurally, the patient was reassessed in the Cardiology Outpatient Clinic. The patient reported no symptoms of heart failure and no symptomatic atrial fibrillation paroxysms. The 24-hour electrocardiography monitoring was normal. The echocardiography assessment revealed normal left ventricular function with LVEF of 55%, LVEDD of 46 mm, left atrial diameter of 41 mm, and an area of 18 cm2; the mitral regurgitation decreased to mild, and the pericardial effusion decreased to 3 mm. The heart failure and rhythm control pharmacotherapy remained unchanged throughout the entire 2-year observational period.
To date, rare cases of PDA shunts have been closed with KONAR-MF occluders [2, 3] mainly in children. Our experience shows that it may also be effective in elderly patients. The device, in contrast to most PDA occluders (such as ADO I), has 2 discs, which potentially prevents embolization and dislocation of the implant into the aorta in the event of slightly increased pulmonary pressure. Due to multifunctional properties and retention screws on both sides, it can be used in different locations effectively (it may be inserted from either the venous or arterial side), and the delivery system makes it possible to safely close severely calcified ducts in elderly patients [3]. Other two-disc implants that could be considered in this case include the Amplatzer Muscular VSD Occluder and Amplatzer Duct Occluder II. Transcatheter PDA closure in adults was reported to alleviate heart failure symptoms [1, 4], prevent further complications, and enable reverse cardiac remodeling [4].







